Advanced Palladium-Catalyzed Synthesis of 1,5-Diene Derivatives for Commercial Scale-Up
Introduction to Novel 1,5-Diene Synthetic Methodology
The development of efficient synthetic routes for 1,5-diene structural motifs remains a pivotal challenge in modern organic synthesis, particularly given their ubiquity in bioactive natural products and functional materials. Patent CN111484388B introduces a transformative approach to constructing these valuable scaffolds through a palladium-catalyzed coupling reaction that bypasses traditional limitations. This technology leverages aryl-substituted propynes and allyl boron esters as primary building blocks, utilizing a specialized catalytic system comprising a palladium precursor, a monophosphorus ligand, and crucial acidic and alcoholic additives. By operating under mild thermal conditions in organic solvents, this method achieves high atom economy and exceptional yields, positioning it as a superior alternative for the production of high-purity pharmaceutical intermediates. The strategic integration of these readily available starting materials allows for the direct assembly of complex diene frameworks without the need for pre-functionalized leaving groups on the allylic component.
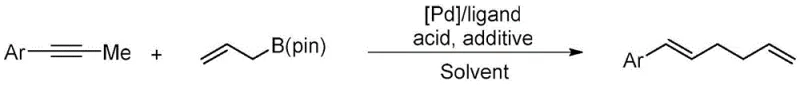
For research directors and process chemists, the significance of this invention lies in its operational simplicity and robustness. The protocol eliminates the cumbersome preparation of allylic carbonates or halides, which are typically required in conventional transition-metal-catalyzed allylic substitutions. Instead, the direct use of stable allyl boron pinacol esters streamlines the supply chain and reduces waste generation. The reaction proceeds efficiently with a catalyst loading as low as 2.5 mol%, demonstrating that high turnover numbers are achievable without excessive metal consumption. This advancement not only accelerates the discovery phase for new drug candidates containing 1,5-diene backbones but also provides a scalable pathway for the commercial manufacturing of key intermediates used in the synthesis of terpenes, fragrances, and advanced polymeric materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,5-dienes has relied heavily on transition-metal-catalyzed allyl-allyl coupling reactions, which often necessitate the use of allylic substrates bearing specific leaving groups such as carbonates, halides, acetates, or amines. While these methods can offer good selectivity, they suffer from inherent drawbacks regarding atom economy and step efficiency. The requirement to pre-install a leaving group on the allylic starting material adds synthetic steps, increases raw material costs, and generates stoichiometric amounts of salt or organic waste byproducts. Furthermore, the handling of reactive allylic halides or unstable carbonates can pose safety risks and complicate storage logistics in a manufacturing environment. From a green chemistry perspective, the displacement of these leaving groups represents a loss of molecular mass that does not contribute to the final product, thereby reducing the overall efficiency of the process and increasing the environmental footprint of the manufacturing operation.
The Novel Approach
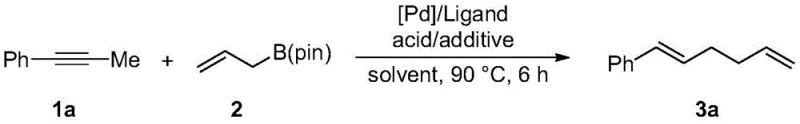
In stark contrast, the methodology disclosed in patent CN111484388B utilizes a direct coupling strategy that merges arylpropynes with allyl boron esters under palladium catalysis. This novel route circumvents the need for allylic leaving groups entirely, relying instead on the activation of the boron-carbon bond in the presence of specific acid and alcohol additives. The reaction conditions are remarkably mild, typically proceeding at temperatures around 90 °C in solvents like 1,4-dioxane, which are standard in industrial settings. The inclusion of n-butanol and 1-adamantanecarboxylic acid as additives is critical, as they facilitate the catalytic cycle and ensure high conversion rates, often exceeding 80% for a wide range of substrates. This approach not only simplifies the reaction setup but also enhances the sustainability profile of the synthesis by improving atom economy and reducing the number of purification steps required to isolate the final 1,5-diene product.
Mechanistic Insights into Pd-Catalyzed Hydro-Allylation
The catalytic cycle underpinning this transformation involves a sophisticated interplay between the palladium center, the phosphine ligand, and the unique additive system. The reaction initiates with the oxidative addition or transmetallation of the allyl boron species to the palladium catalyst, facilitated by the presence of the carboxylic acid additive which likely activates the boron ester. Subsequent coordination and insertion of the arylpropyne alkyne into the palladium-allyl bond generates a vinyl-palladium intermediate. The critical role of the alcohol additive, specifically n-butanol, is believed to assist in the protonolysis of this organometallic intermediate, releasing the final 1,5-diene product and regenerating the active palladium species. This mechanism avoids the formation of stable off-cycle complexes that often plague palladium-catalyzed reactions, ensuring a continuous and efficient turnover. The use of bulky ligands like tricyclohexylphosphine further stabilizes the active catalyst and prevents the formation of palladium black, maintaining catalytic activity over extended reaction times.
From an impurity control perspective, this mechanism offers distinct advantages over traditional cross-coupling methods. The specificity of the protonolysis step minimizes the formation of homocoupling byproducts or isomerized alkene side products that are common in base-mediated processes. The mild acidic conditions prevent the degradation of sensitive functional groups on the aromatic ring, such as esters or halides, which might otherwise undergo hydrolysis or elimination under harsher basic conditions typical of Suzuki-Miyaura couplings. Consequently, the crude reaction mixture is cleaner, simplifying downstream processing and reducing the load on purification columns. This high level of chemoselectivity is paramount for pharmaceutical applications where strict limits on genotoxic impurities and heavy metal residues must be maintained throughout the synthesis of complex active pharmaceutical ingredients.
How to Synthesize 1,5-Diene Derivatives Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of scaling, making it accessible for both laboratory discovery and pilot plant operations. The standardized protocol involves the sequential addition of the palladium catalyst, ligand, acid additive, and alcohol additive into a reaction vessel under an inert atmosphere, followed by the introduction of the substrates and solvent. The reaction is then heated to a moderate temperature, typically 90 °C, and stirred for a defined period, usually around 6 hours, to ensure complete conversion. Following the reaction, the solvent is removed under reduced pressure, and the crude residue is purified via standard column chromatography using silica gel and a gradient of ethyl acetate in petroleum ether. For detailed standard operating procedures and specific stoichiometric ratios tailored to different substrate classes, please refer to the comprehensive guide below.
- Prepare the reaction mixture by weighing Pd2(dba)3 (2.5 mol%), PCy3 (10 mol%), 1-adamantanecarboxylic acid (10 mol%), and n-butanol (2.0 equiv) into a reaction flask.
- Add the arylpropyne substrate (0.2 mmol), allylboron pinacol ester (0.4 mmol), and dioxane solvent (1.0 mL) under inert atmosphere.
- Seal the vessel, heat to 90 °C for 6 hours, then remove solvent and purify via column chromatography to isolate the pure 1,5-diene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers significant strategic advantages in terms of cost structure and logistical reliability. The primary raw materials, arylpropynes and allyl boron pinacol esters, are commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of single-source dependency. Unlike specialized allylic electrophiles which may require custom synthesis and have limited shelf-life, allyl boron esters are stable, easy to handle, and can be stocked in bulk without significant degradation. This stability translates directly into reduced inventory costs and simplified warehouse management, allowing for just-in-time manufacturing strategies that optimize working capital. Furthermore, the elimination of leaving group precursors reduces the overall bill of materials, as fewer reagents are required to prepare the starting materials, leading to substantial cost savings in the early stages of the supply chain.
- Cost Reduction in Manufacturing: The streamlined nature of this reaction directly impacts the cost of goods sold by minimizing unit operations. By avoiding the separate synthesis of allylic carbonates or halides, manufacturers save on reagents, solvents, and labor associated with those preparatory steps. Additionally, the high atom economy means that a greater proportion of the input mass ends up in the final product, reducing waste disposal costs. The use of a recyclable catalyst system further enhances economic efficiency, as the potential for catalyst recovery lowers the effective cost of the precious metal input. These factors combine to create a leaner, more cost-effective manufacturing process that improves margin potential for high-volume fine chemical production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality, which is critical for maintaining uninterrupted supply to downstream customers. The tolerance for various functional groups means that a single platform technology can be used to produce a diverse library of intermediates, reducing the need for multiple specialized production lines. This flexibility allows manufacturers to respond rapidly to changes in market demand or customer specifications without significant retooling. Moreover, the use of common solvents like dioxane and standard workup procedures ensures that the process can be easily transferred between different manufacturing sites, enhancing supply chain resilience against regional disruptions or capacity constraints.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is straightforward due to the absence of hazardous reagents and the use of moderate temperatures. The reaction does not require cryogenic conditions or high-pressure equipment, lowering the capital expenditure required for scale-up. From an environmental standpoint, the improved atom economy and reduced waste generation align with increasingly stringent global regulations on chemical manufacturing. The ability to produce high-purity intermediates with minimal byproduct formation simplifies wastewater treatment and reduces the environmental footprint of the facility, supporting corporate sustainability goals and ensuring long-term regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 1,5-diene synthesis technology. These answers are derived directly from the experimental data and optimization studies presented in the patent literature, providing a reliable basis for process evaluation. Understanding these nuances is essential for R&D teams assessing the feasibility of integrating this route into their existing pipelines and for procurement teams evaluating the long-term viability of the supply source.
Q: What are the critical additives for high yield in this 1,5-diene synthesis?
A: The patent identifies 1-adamantanecarboxylic acid (10 mol%) and n-butanol (2.0 equivalents) as essential additives. Without the alcohol additive or acid, yields drop significantly, indicating their role in facilitating the catalytic turnover and protonolysis steps.
Q: Can this method tolerate electron-withdrawing groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent functional group tolerance. Substrates with electron-withdrawing groups such as trifluoromethyl (yield 84%) and ester groups (yield 90%) react efficiently, making it suitable for synthesizing diverse pharmaceutical intermediates.
Q: Is the catalyst system commercially viable for large-scale production?
A: The process utilizes commercially available palladium precursors like Pd2(dba)3 and standard monodentate phosphine ligands. The mild reaction temperature of 90 °C and the use of common solvents like dioxane support its feasibility for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Diene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and commercialization. Our team of expert process chemists has extensively evaluated the palladium-catalyzed coupling of arylpropynes and allyl boron esters, confirming its potential for robust commercial scale-up. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,5-diene intermediate delivered meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of novel diene scaffolds or reliable supply of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce overall manufacturing costs.
