Revolutionizing Agrochemical Intermediate Production: Advanced Microchannel Synthesis of O-(3-Chloro-2-Propenyl) Hydroxylamine
The global demand for high-efficiency herbicides, particularly cyclohexenone oxime derivatives such as Clethodim and Tralkoxydim, has necessitated a rigorous re-evaluation of key intermediate supply chains. At the forefront of this chemical evolution is O-(3-chloro-2-propenyl) hydroxylamine, a critical alkoxyamine compound that serves as the foundational building block for these potent ACCase inhibitor herbicides. Recent advancements documented in patent CN112851544A introduce a transformative synthetic methodology that leverages continuous flow microchannel technology to overcome the longstanding inefficiencies of batch processing. This innovation not only addresses the purity requirements demanded by R&D Directors but also aligns with the sustainability goals of modern supply chain leadership. By integrating methyl isobutyl ketone as a dual-function reagent, this process eliminates the need for excessive solvent consumption and hazardous drying agents, marking a significant leap forward in green chemistry for agrochemical manufacturing.
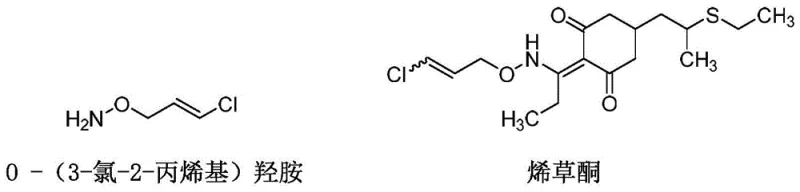
Historically, the industrial production of O-(3-chloro-2-propenyl) hydroxylamine has been plagued by significant operational bottlenecks and environmental liabilities inherent to conventional batch methodologies. Traditional routes, such as the ethyl acetate method, suffer from severe solvent recovery challenges where the post-reaction mixture contains substantial amounts of byproduct ethanol, rendering the solvent difficult to recycle efficiently. Furthermore, these legacy processes often rely on the addition of stoichiometric amounts of solid drying agents like anhydrous magnesium sulfate or sodium sulfate, which generates considerable volumes of solid hazardous waste that require costly disposal protocols. Another prevalent route involving ethanol and acetonitrile necessitates the introduction of large quantities of hydrogen chloride gas, creating severe corrosion risks for reactor vessels and demanding expensive anti-corrosion materials that drive up capital expenditure. These conventional approaches also frequently employ relatively expensive phase transfer catalysts, which add to the raw material cost burden without guaranteeing consistent impurity profiles suitable for high-end pharmaceutical or agrochemical applications.
In stark contrast, the novel approach disclosed in the referenced patent utilizes a continuous flow strategy that fundamentally reengineers the reaction environment to maximize efficiency and minimize waste. The core innovation lies in the strategic deployment of methyl isobutyl ketone, which acts simultaneously as the solvent medium and the amino protective group, thereby maintaining its molecular integrity throughout the synthesis cycle. This dual functionality allows for the direct recovery and reuse of the solvent after a simple azeotropic dehydration step, effectively closing the material loop and drastically reducing raw material consumption. By transitioning from static kettle reactors to dynamic microchannel systems, the process achieves superior control over reaction parameters such as temperature and residence time, which is critical for managing the exothermic nature of the alkylation steps. This technological shift not only enhances the overall yield to levels exceeding 94% but also ensures a purity profile greater than 99%, meeting the stringent specifications required for downstream herbicide synthesis without the need for extensive purification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on traditional batch synthesis routes for O-(3-chloro-2-propenyl) hydroxylamine presents multifaceted challenges that impact both the economic viability and the environmental compliance of manufacturing facilities. In the ethyl acetate route, for instance, the formation of acetic acid and sodium chloride as stoichiometric byproducts creates a highly corrosive environment that accelerates equipment degradation and increases maintenance downtime. The separation of the organic phase from the aqueous layer is often complicated by emulsion formation, leading to product loss and reduced overall throughput. Moreover, the necessity to use large volumes of organic solvents for extraction, followed by energy-intensive distillation for recovery, results in a substantial carbon footprint that is increasingly untenable under modern environmental regulations. The use of solid drying agents further exacerbates the issue by introducing a solid waste stream that cannot be easily treated or recycled, forcing manufacturers to incur additional costs for hazardous waste management and disposal services.
The Novel Approach
The novel microchannel-based synthesis route offers a comprehensive solution to these entrenched problems by redesigning the chemical process at the molecular and engineering levels. By utilizing methyl isobutyl ketone as a recyclable protecting group, the process eliminates the generation of acetic acid and salt byproducts, thereby neutralizing the corrosion risk and simplifying the downstream workup procedure. The continuous flow nature of the microchannel reactor ensures that reactants are mixed at the molecular level almost instantaneously, which suppresses side reactions and minimizes the formation of impurities that are common in poorly mixed batch tanks. This precision control allows for the operation at optimal temperatures between 50°C and 90°C without the risk of thermal runaway, ensuring consistent product quality across large production campaigns. Furthermore, the implementation of azeotropic dehydration removes the need for solid desiccants, resulting in a cleaner process stream that significantly reduces the volume of three wastes (wastewater, waste gas, and solid waste) generated per kilogram of product.
Mechanistic Insights into MIBK-Catalyzed Continuous Flow Synthesis
The chemical elegance of this new process is rooted in the reversible protection of the hydroxylamine functionality using methyl isobutyl ketone, which stabilizes the reactive nitrogen species during the harsh alkylation conditions. In the first stage of the reaction, free hydroxylamine, generated in situ from hydroxylamine salts via ion exchange resin treatment, reacts with methyl isobutyl ketone to form the corresponding ketoxime. This protection step is crucial because it prevents the self-decomposition of hydroxylamine and directs the nucleophilic attack specifically towards the desired allylic position on the 1,3-dichloropropene molecule. The use of a microchannel reactor for this step ensures that the equilibrium is rapidly established under controlled thermal conditions, typically between 20°C and 100°C, preventing the accumulation of unstable intermediates that could lead to safety incidents.
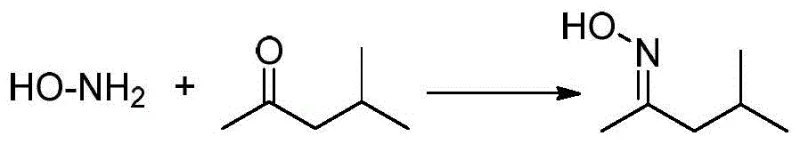
Following the protection step, the reaction mixture flows directly into a second-stage microchannel reactor where the etherification takes place in the presence of an alkaline solution. The ketoxime intermediate undergoes nucleophilic substitution with 1,3-dichloropropene, facilitated by the basic environment which deprotonates the oxime hydroxyl group to enhance its nucleophilicity. The precise mixing capabilities of the microchannel system ensure that the local concentration of the base and the alkylating agent remains uniform, preventing localized hot spots that could cause polymerization or degradation of the sensitive chloro-allyl moiety. After the etherification is complete, the protecting group is removed under mild acidic conditions, regenerating the methyl isobutyl ketone for recovery and releasing the final O-(3-chloro-2-propenyl) hydroxylamine product. This mechanistic pathway not only maximizes atom economy but also ensures that the impurity profile is tightly controlled, as the reversible nature of the protection step allows for error correction during the synthesis.
How to Synthesize O-(3-Chloro-2-Propenyl) Hydroxylamine Efficiently
The implementation of this advanced synthetic route requires a systematic approach to process engineering that prioritizes safety, efficiency, and reproducibility. The procedure begins with the preparation of a high-purity hydroxylamine solution by passing an aqueous solution of hydroxylamine salt through a column of alkaline ion exchange resin, which effectively removes chloride or sulfate ions that could interfere with the reaction or contaminate the wastewater. This purified hydroxylamine solution is then preheated and pumped into the microchannel system alongside methyl isobutyl ketone, where the initial condensation reaction occurs under strictly controlled thermal gradients. The detailed standardized synthesis steps, including specific flow rates, residence times, and quenching protocols, are outlined in the technical guide below to ensure that operators can replicate the high yields and purity levels demonstrated in the patent examples.
- Preparation of Hydroxylamine Solution: Treat hydroxylamine salt aqueous solution with alkaline ion exchange resin to obtain free hydroxylamine solution, effectively reducing salt content in wastewater.
- Protection and Etherification in Microchannel: Pump preheated hydroxylamine and methyl isobutyl ketone into a first-stage microchannel reactor to form ketoxime, then feed directly into a second-stage reactor with 1,3-dichloropropene and alkali.
- Workup and Solvent Recovery: Adjust pH to weak acidity, separate phases, extract water phase with methyl isobutyl ketone, perform azeotropic dehydration, and recover the solvent to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microchannel synthesis technology translates into tangible strategic advantages that extend far beyond simple unit cost metrics. The elimination of expensive phase transfer catalysts and the reduction in solvent consumption directly contribute to a lower variable cost structure, making the supply of this critical intermediate more resilient against fluctuations in raw material pricing. Furthermore, the continuous nature of the process allows for a smaller physical footprint compared to traditional batch plants, enabling decentralized production capabilities that can reduce logistics costs and lead times for regional manufacturing hubs. The robustness of the process also means fewer batch failures and less variability in quality, which reduces the risk of production stoppages downstream at the herbicide formulation plants.
- Cost Reduction in Manufacturing: The innovative use of methyl isobutyl ketone as a recyclable solvent and protecting agent eliminates the need for purchasing large volumes of disposable solvents like ethyl acetate. By recovering and reusing the ketone through azeotropic distillation, the process significantly lowers the raw material intensity per kilogram of product. Additionally, the removal of solid drying agents from the workflow eradicates the cost associated with purchasing, handling, and disposing of these consumables. The continuous flow system also reduces energy consumption by optimizing heat exchange efficiency, leading to substantial utility savings over the lifecycle of the production facility.
- Enhanced Supply Chain Reliability: The transition to a continuous manufacturing model inherently improves supply continuity by removing the start-stop cycles associated with batch processing. This allows for a steady, on-demand production rate that can be scaled up or down more flexibly to match market demand without the long turnaround times required for cleaning and setting up batch reactors. The simplified workup procedure, which avoids complex extractions and filtrations, reduces the overall cycle time from raw material intake to finished goods, thereby shortening the lead time for high-purity agrochemical intermediates. This agility ensures that customers can maintain leaner inventory levels while still securing a reliable supply of critical materials.
- Scalability and Environmental Compliance: Scaling this process is achieved primarily by running the microchannel reactors for longer durations or numbering up the reactor units, rather than increasing the vessel size, which mitigates the safety risks associated with scaling exothermic reactions. The process generates significantly less wastewater with lower salt content due to the use of ion exchange resins instead of neutralization with strong acids and bases. The absence of solid waste from drying agents simplifies environmental compliance and reduces the regulatory burden on the manufacturing site. This green chemistry profile aligns with the increasing pressure from stakeholders to adopt sustainable manufacturing practices, enhancing the brand value of the supply chain partners involved.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of O-(3-chloro-2-propenyl) hydroxylamine using this advanced methodology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, ensuring that the information provided is accurate and relevant for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing supply chain or development pipeline.
Q: What are the primary advantages of using methyl isobutyl ketone in this synthesis?
A: Methyl isobutyl ketone serves a dual function as both the amino protective agent and the reaction solvent. Its molecular structure remains unchanged before and after the reaction, allowing for efficient recovery and reuse, which overcomes the defects of traditional processes that consume large amounts of ethyl acetate and generate acetic acid byproducts.
Q: How does the microchannel reactor improve the production of O-(3-chloro-2-propenyl) hydroxylamine?
A: The microchannel reactor replaces traditional kettle-type or tubular reactors, providing superior heat and mass transfer capabilities. This enhances reaction control, improves product yield and quality, and facilitates safer handling of exothermic reactions compared to batch processes.
Q: Does this process generate significant solid waste?
A: No, the process utilizes an azeotropic dehydration method instead of chemical drying agents like anhydrous sodium sulfate. This eliminates the generation of solid waste associated with drying agents, contributing to a more environmentally friendly production profile.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-(3-Chloro-2-Propenyl) Hydroxylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of next-generation herbicides depends on the availability of high-quality intermediates produced through sustainable and scalable processes. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials regardless of market volatility. Our state-of-the-art facilities are equipped with the necessary infrastructure to implement continuous flow technologies, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for agrochemical synthesis. We are committed to being a long-term partner who not only supplies chemicals but also provides technical solutions that enhance the efficiency of your entire value chain.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your production costs and secure your supply of critical herbicide intermediates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us help you navigate the complexities of modern chemical manufacturing with confidence and precision.
