Advanced Alpha-Ketothioamide Synthesis: Scalable S8 Technology for Pharma Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing sulfur-containing heterocycles and functional motifs, particularly alpha-ketothioamides, which serve as pivotal scaffolds in drug discovery. A significant technological breakthrough in this domain is documented in patent CN110511193B, which discloses a novel, efficient, and environmentally benign synthetic route for alpha-ketothioamide compounds. Unlike traditional approaches that rely on hazardous reagents or extreme conditions, this innovation utilizes elemental sulfur (S8) as the primary sulfur source, reacting it with alpha-azidoacetophenone derivatives and various amines. The process operates under remarkably mild conditions, typically at room temperature, and achieves completion within a short timeframe of 0.5 to 4 hours. For R&D directors and procurement strategists, this patent represents a paradigm shift towards greener chemistry, offering a pathway to high-purity intermediates while simultaneously addressing the critical industry pain points of waste management and raw material costs. The versatility of this method allows for a broad substrate scope, accommodating various substituents on both the ketone and amine components, thereby ensuring its applicability across diverse medicinal chemistry programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamides and alpha-ketothioamides has been plagued by significant operational and environmental challenges that hinder large-scale adoption. The most ubiquitous traditional method involves the use of Lawesson's reagent or other phosphorus-based thionating agents. While effective in small-scale laboratory settings, these reagents are notoriously expensive, moisture-sensitive, and generate substantial amounts of phosphorus-containing waste, which complicates downstream purification and imposes heavy burdens on wastewater treatment facilities. Furthermore, alternative routes such as the Willger-Kindler reaction, which utilizes aryl methyl ketones, amines, and sulfur, often require harsh reaction conditions, including high temperatures and prolonged reaction times, leading to poor energy efficiency and potential safety hazards in a plant environment. These conventional methods frequently suffer from limited substrate tolerance, where sensitive functional groups may degrade under the aggressive conditions required for C=O to C=S conversion. Consequently, manufacturers face inflated production costs due to expensive reagents, complex waste disposal protocols, and lower overall yields caused by side reactions, creating a compelling need for a more sustainable and economically viable alternative.
The Novel Approach
The methodology outlined in patent CN110511193B offers a transformative solution by leveraging the unique reactivity of alpha-azidoacetophenones with elemental sulfur. This approach fundamentally alters the synthetic landscape by replacing costly and toxic thionating agents with abundant, inexpensive, and odorless elemental sulfur (S8). The reaction mechanism proceeds through a fascinating C-N bond cleavage pathway rather than direct oxygen-sulfur exchange, allowing for the construction of the alpha-ketothioamide core with exceptional atom economy. As illustrated in the general reaction scheme below, the process involves the simple mixing of an alpha-azidoacetophenone compound, an amine (primary or secondary), and elemental sulfur in a common organic solvent.
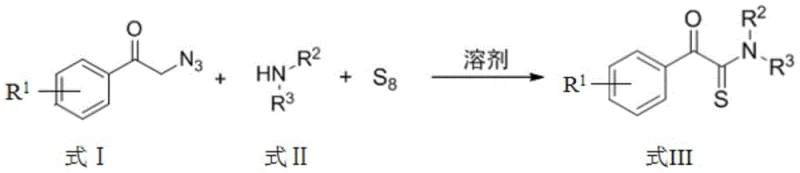
The operational simplicity of this new route cannot be overstated; it eliminates the need for specialized equipment capable of withstanding high pressure or temperature, as the reaction proceeds efficiently at room temperature. The compatibility with a wide range of solvents, including THF, DMF, DMSO, and toluene, provides process chemists with the flexibility to optimize solubility and crystallization parameters for specific substrates. Moreover, the workup procedure is straightforward, involving standard extraction with ethyl acetate and brine followed by silica gel chromatography, which facilitates the isolation of high-purity products without the need for complex metal scavenging or phosphorus removal steps. This streamlined workflow not only accelerates the timeline from bench to pilot plant but also significantly reduces the operational expenditure associated with solvent recovery and waste neutralization, positioning this technology as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Elemental Sulfur-Mediated C-N Bond Cleavage
For the technical leadership evaluating the feasibility of this process, understanding the underlying mechanistic pathway is crucial for troubleshooting and optimization. The reaction initiates with the tautomerization of the substrate, alpha-azidoacetophenone, into its electron-rich enol form. This nucleophilic enol species then attacks the electrophilic elemental sulfur ring (S8), triggering the opening of the sulfur chain and the formation of a sulfur-intermediate complex. A critical proton transfer (PT) step follows, stabilizing the intermediate and setting the stage for the extrusion of nitrogen gas and sulfur fragments. The mechanism culminates in the formation of a key alpha-ketothioacyl azide intermediate, which subsequently undergoes nucleophilic attack by the amine component to yield the final alpha-ketothioamide product. This intricate dance of bond breaking and forming, specifically the cleavage of the C-N bond of the azido group and the concurrent formation of the C=S bond, is what grants this reaction its unique selectivity and efficiency.
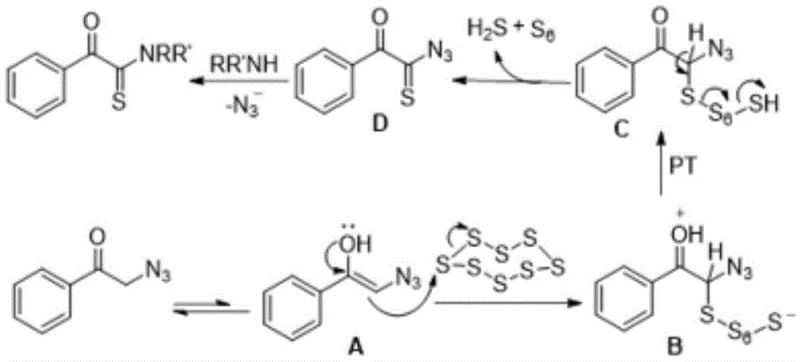
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy thermal processes. Because the reaction proceeds through well-defined ionic or polar intermediates at mild temperatures, the formation of random polymeric sulfur byproducts or decomposition products is minimized. The specific stoichiometry recommended in the patent, typically utilizing a molar ratio of 1:(1.2-5):(1.5-4) for the ketone, amine, and sulfur respectively, ensures that the sulfur source is in slight excess to drive the reaction to completion without overwhelming the system with unreacted sulfur that could complicate purification. The release of H2S and S6 as byproducts is managed within the solvent system, and the use of saturated brine during the workup effectively salts out water-soluble impurities and breaks emulsions, ensuring a clean phase separation. This level of mechanistic control translates directly to a cleaner crude profile, reducing the load on the purification columns and enhancing the overall recovery of the target API intermediate.
How to Synthesize Alpha-Ketothioamides Efficiently
Implementing this synthesis in a production environment requires adherence to precise operational parameters to maximize yield and safety. The process begins with the careful selection of the amine substrate, where both primary amines (such as benzylamine or cyclohexylamine) and secondary amines (such as morpholine or piperidine) have demonstrated excellent reactivity. The choice of solvent plays a pivotal role; while polar aprotic solvents like DMF and DMSO offer superior solubility for the sulfur and reactants, ethers like THF or hydrocarbons like toluene may be preferred for easier downstream removal. The reaction mixture is stirred at ambient temperature, monitoring progress via TLC or HPLC until the starting azido compound is fully consumed, typically within 0.5 to 4 hours depending on the electronic nature of the substituents. Detailed standardized synthesis steps follow below.
- Mix alpha-azidoacetophenone, amine compound, and elemental sulfur (S8) in a suitable solvent such as THF, DMF, or DMSO.
- Stir the reaction mixture at room temperature for 0.5 to 4 hours to allow for C-N bond cleavage and thionation.
- Quench the reaction, extract with ethyl acetate and saturated brine, dry the organic layer, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this elemental sulfur-based methodology offers profound strategic benefits that extend beyond mere technical novelty. The primary driver for adoption is the drastic simplification of the raw material supply chain. Elemental sulfur is a commodity chemical produced in massive quantities globally as a byproduct of oil and gas refining, ensuring a stable, long-term supply with minimal price volatility compared to specialized organophosphorus reagents. By eliminating the dependency on imported, high-cost thionating agents, manufacturers can insulate their production costs from market fluctuations and geopolitical supply disruptions. Furthermore, the mild reaction conditions translate directly into energy savings; operating at room temperature removes the need for steam heating or cryogenic cooling, significantly lowering the utility burden on the manufacturing facility. This energy efficiency, combined with the reduced reaction time, increases the throughput capacity of existing reactor trains, allowing for greater production volume without capital investment in new hardware.
- Cost Reduction in Manufacturing: The economic argument for this process is robust, driven primarily by the substitution of expensive reagents with commodity sulfur. Traditional methods often require stoichiometric or excess amounts of costly reagents like Lawesson's reagent, which also necessitate expensive quenching and waste treatment protocols to handle phosphorus residues. In contrast, the elemental sulfur method generates inorganic sulfur byproducts that are easier and cheaper to dispose of or recycle. The simplified workup procedure, which avoids complex metal scavenging or multi-step extractions required to remove phosphorus impurities, further reduces labor and material costs. Additionally, the high yields reported in the patent examples (often exceeding 85-90%) mean less raw material is wasted per kilogram of product, directly improving the gross margin for the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Alpha-azidoacetophenones can be prepared from readily available acetophenones via established bromination and azidation sequences, ensuring that the upstream supply chain is secure. The robustness of the reaction conditions means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or mixing rates, which is a common risk with sensitive reagents. This reliability ensures consistent delivery schedules to downstream customers, reducing the risk of stockouts that can halt clinical trials or commercial drug production. The ability to use a variety of common solvents also provides flexibility; if one solvent faces a shortage, the process can often be adapted to another without compromising yield, adding another layer of security to the supply chain.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental hurdles, but this methodology is inherently designed for scalability. The absence of highly exothermic steps or hazardous gas evolution (beyond manageable H2S which is trapped in the solvent/workup) makes the scale-up from grams to metric tons predictable and safe. From an environmental compliance standpoint, the "green" nature of using elemental sulfur aligns perfectly with increasingly stringent global regulations on chemical manufacturing. Reducing phosphorus waste and avoiding heavy metal catalysts simplifies the permitting process for new production lines and lowers the cost of environmental compliance audits. This sustainability profile is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies who prioritize green chemistry in their vendor selection criteria.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific pipelines, we have compiled answers to common questions regarding the practical application of this patent. These insights address concerns about substrate compatibility, safety profiles, and purification strategies, providing a clear roadmap for implementation. Understanding these nuances is essential for project managers planning the integration of this synthesis into existing GMP manufacturing suites.
Q: What are the advantages of using elemental sulfur over Lawesson's reagent?
A: Elemental sulfur is significantly cheaper, non-toxic, and avoids the generation of phosphorus-containing waste, making the process more environmentally friendly and cost-effective compared to traditional thionating agents.
Q: What is the typical reaction time for this synthesis?
A: The reaction proceeds rapidly under mild conditions, typically completing within 0.5 to 4 hours at room temperature, which drastically reduces energy consumption compared to high-temperature methods.
Q: Is this method scalable for industrial production?
A: Yes, the process uses readily available raw materials and simple workup procedures (extraction and chromatography), making it highly suitable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketothioamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our technical team has extensively analyzed the methodology described in CN110511193B and is fully equipped to translate this laboratory-scale innovation into a robust, GMP-compliant manufacturing process. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are designed to handle sulfur chemistry safely, with dedicated scrubbing systems and containment protocols that exceed industry standards. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of alpha-ketothioamide meets the exacting requirements of modern drug development, minimizing the risk of downstream failures.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this elemental sulfur route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities. Let us partner with you to accelerate your timeline to market with a reliable, cost-effective, and sustainable supply of high-purity alpha-ketothioamide intermediates.
