Revolutionizing Chiral Intermediate Manufacturing with Advanced Phase Transfer Catalysis Technology
The landscape of chiral synthesis is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective manufacturing processes in the fine chemical industry. Patent CN107721858B introduces a groundbreaking phase-transfer catalyzed method for the asymmetric α-benzoylation of β-ketoesters, addressing critical bottlenecks in the production of optically active α-oxo-β-ketoester compounds. These structural units are indispensable precursors in the synthesis of complex natural products, chiral pharmaceuticals, and advanced agrochemical intermediates. Unlike traditional methods that rely on stoichiometric chiral oxidants or expensive metal complexes, this innovation leverages cheap and readily available cinchona alkaloid quaternary ammonium salts to achieve high levels of stereocontrol. The technical breakthrough lies in the ability to perform this transformation under mild conditions using benzoyl peroxide as the oxidant, thereby simplifying the operational workflow and enhancing safety profiles for industrial applications. This patent represents a pivotal shift towards greener chemistry, offering a robust platform for the reliable pharmaceutical intermediates supplier market to deliver high-value chiral building blocks with improved economic efficiency.
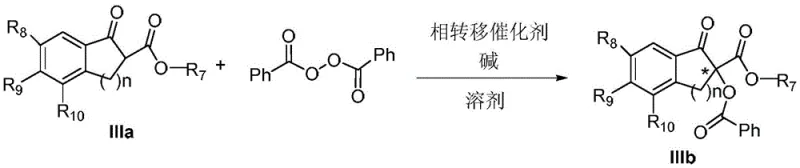
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral α-oxo-β-ketoesters has been plagued by significant technical and economic challenges that hinder large-scale commercialization. Early methodologies, such as those utilizing Davis reagents, were characterized by cumbersome operational procedures and harsh reaction conditions that posed safety risks in a production environment. Furthermore, these traditional routes often required chemical doses of chiral oxidants, leading to exorbitant raw material costs and generating substantial amounts of chemical waste that complicated downstream processing. Even more recent advancements involving metal complex catalysis, while offering improved selectivity, introduced new barriers related to the high cost of precious metal ligands and the persistent issue of heavy metal residue in the final product. For a procurement manager focused on cost reduction in pharmaceutical intermediates manufacturing, the necessity of extensive purification steps to remove trace metals like Titanium or Iron adds unacceptable layers of complexity and expense. Additionally, the sensitivity of many metal catalysts to moisture and oxygen often necessitates specialized equipment and inert atmosphere handling, further inflating the capital expenditure required for facility setup and maintenance.
The Novel Approach
The novel phase-transfer catalysis (PTC) strategy described in the patent data offers a compelling solution to these entrenched industry pain points by fundamentally rethinking the catalytic mechanism. By employing organocatalysts derived from cinchona alkaloids, this method completely eliminates the reliance on transition metals, thereby removing the risk of metal contamination and the associated regulatory hurdles for pharmaceutical grade materials. The reaction proceeds efficiently in a biphasic system where the catalyst facilitates the transport of reactive species across the interface, allowing for the use of inexpensive inorganic bases like potassium carbonate or sodium hydroxide. This shift not only drastically simplifies the reaction setup but also enables the use of benign solvents such as toluene or even solvent-free conditions in some variations. From a supply chain perspective, the robustness of this system means that production is less susceptible to fluctuations in the availability of rare earth metals or complex ligands. The ability to achieve high yields and excellent enantioselectivity under mild temperatures further underscores the practical viability of this approach for the commercial scale-up of complex polymer additives and fine chemical intermediates, ensuring a stable and continuous supply of critical materials.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Asymmetric Benzoylation
The core of this technological advancement lies in the sophisticated design of the chiral phase-transfer catalysts, which are derived from naturally occurring cinchona alkaloids modified into quaternary ammonium salts. These catalysts function by forming a chiral ion pair with the enolate intermediate generated from the β-ketoester substrate in the aqueous phase. The bulky chiral environment surrounding the nitrogen center of the catalyst dictates the facial selectivity of the subsequent attack by the benzoyl peroxide electrophile. This precise stereochemical control is achieved through a combination of steric hindrance and specific non-covalent interactions, such as hydrogen bonding and pi-stacking, between the catalyst scaffold and the substrate. The patent details a library of catalyst structures, including variations with different substituents on the quinoline and benzyl moieties, allowing for fine-tuning of the electronic and steric properties to match specific substrate requirements. Understanding this mechanistic nuance is crucial for R&D directors aiming to optimize the process for new derivatives, as slight modifications to the catalyst structure can lead to significant improvements in enantiomeric excess.
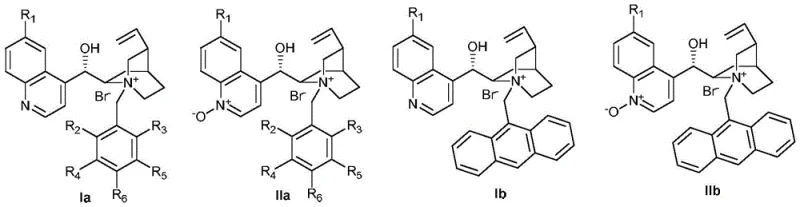
Beyond mere selectivity, the mechanism also provides inherent advantages in terms of impurity control and product purity. The biphasic nature of the reaction ensures that the ionic byproducts and excess base remain largely confined to the aqueous layer, while the organic product partitions into the organic solvent. This natural separation minimizes the formation of side products that often arise from over-oxidation or base-catalyzed decomposition in homogeneous systems. Furthermore, the mild reaction conditions prevent the thermal degradation of sensitive functional groups that might be present on complex substrate molecules. For quality assurance teams, this translates to a cleaner crude reaction profile, reducing the burden on purification columns and crystallization steps. The ability to consistently produce high-purity OLED material or pharmaceutical precursors with minimal impurity profiles is a direct result of this controlled mechanistic pathway. By avoiding the radical pathways often associated with metal-catalyzed oxidations, the PTC method ensures a more predictable and reproducible outcome, which is essential for maintaining batch-to-batch consistency in regulated industries.
How to Synthesize Chiral α-Benzoyl-β-Ketoesters Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the interplay between the organic and aqueous phases to maximize catalytic efficiency. The process begins with the dissolution of the β-ketoester substrate and the benzoyl peroxide oxidant in a suitable organic solvent such as toluene or trifluorotoluene, followed by the addition of the chiral phase-transfer catalyst. The reaction is initiated by the slow addition of an aqueous alkaline solution, with temperature control being a critical parameter to maintain high enantioselectivity, typically ranging from 0 to 20 degrees Celsius. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during operation.
- Prepare the reaction mixture by combining β-ketoester substrates with benzoyl peroxide and a specific cinchona alkaloid quaternary ammonium salt catalyst in an organic solvent.
- Introduce an aqueous alkaline solution, such as potassium carbonate or sodium hydroxide, to initiate the phase transfer catalytic cycle under controlled low-temperature conditions.
- Upon completion, separate the organic layer containing the product while retaining the catalyst in the aqueous phase for subsequent recycling and reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this phase-transfer catalyzed methodology represents a strategic opportunity to optimize operational expenditures and mitigate supply risks. The elimination of expensive transition metal catalysts and ligands directly translates to substantial cost savings in raw material procurement, as cinchona alkaloid derivatives are significantly more affordable and commercially available than specialized metal complexes. Moreover, the simplicity of the workup procedure, which often involves simple phase separation rather than complex chromatography or metal scavenging, reduces the consumption of solvents and silica gel, further driving down the cost of goods sold. This economic efficiency is compounded by the ability to operate under milder conditions, which lowers energy consumption for heating or cooling and reduces the wear and tear on reactor equipment. By adopting this technology, companies can achieve significant cost reduction in electronic chemical manufacturing and other high-value sectors without compromising on the quality or purity of the final product.
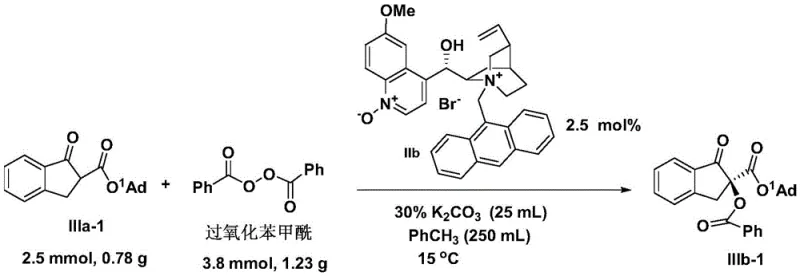
Enhanced supply chain reliability is another critical benefit derived from the recyclability of the catalyst system demonstrated in the patent data. Since the phase-transfer catalyst resides in the aqueous phase after the reaction is complete, it can be easily separated from the organic product layer and reused for subsequent batches with minimal loss of activity. This feature drastically reduces the dependency on continuous fresh catalyst supply, insulating the production process from potential market volatility or shortages of chiral starting materials. For supply chain planners, this means a more predictable production schedule and reduced lead time for high-purity pharmaceutical intermediates, as the need for lengthy catalyst synthesis or sourcing is minimized. Additionally, the reduced generation of hazardous waste and the absence of heavy metals simplify environmental compliance and waste disposal procedures, aligning with increasingly stringent global sustainability regulations. This holistic improvement in process efficiency ensures a robust and resilient supply chain capable of meeting the demanding requirements of international clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric benzoylation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and technical stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the primary advantages of this phase transfer catalysis method over traditional metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal complexes, utilizing cheap and readily available cinchona alkaloid derivatives instead. It offers easier product purification and significantly reduces heavy metal contamination risks in the final pharmaceutical intermediates.
Q: Is the catalyst system suitable for large-scale industrial production?
A: Yes, the patent demonstrates that the phase transfer catalyst remains in the aqueous layer after reaction, allowing for easy separation and multiple cycles of reuse without significant loss of catalytic activity or enantioselectivity, which is critical for cost-effective scale-up.
Q: What level of enantioselectivity can be achieved with this protocol?
A: The technology achieves high enantiomeric excess (ee) values, with specific examples demonstrating selectivity up to 96% ee, ensuring the production of high-purity chiral building blocks required for stringent regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Intermediates Supplier
The technical potential of this phase-transfer catalyzed asymmetric α-benzoylation method is immense, offering a pathway to produce high-value chiral building blocks with unprecedented efficiency and sustainability. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are successfully translated into industrial reality. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards. We understand the critical nature of chiral intermediates in drug development and are equipped to handle the complex synthesis requirements needed to bring your projects from concept to commercialization.
We invite you to collaborate with us to leverage this advanced technology for your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes and quality requirements. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can optimize your manufacturing process. By partnering with us, you gain access to a reliable supply of high-purity chiral intermediates that will strengthen your product pipeline and enhance your competitive edge in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
