Scaling High-Purity Optically Active 1-Amino-2-Vinylcyclopropanecarboxylate for Antiviral Drug Manufacturing
Scaling High-Purity Optically Active 1-Amino-2-Vinylcyclopropanecarboxylate for Antiviral Drug Manufacturing
The pharmaceutical industry continuously demands more efficient and stereoselective pathways for constructing complex molecular architectures, particularly for antiviral agents where chirality dictates biological activity. Patent CN102471239A introduces a groundbreaking methodology for the production of optically active 1-amino-2-vinylcyclopropanecarboxylic acid esters, which serve as critical building blocks in the synthesis of next-generation therapeutics. This technology leverages advanced asymmetric phase-transfer catalysis to achieve high enantioselectivity directly from achiral starting materials, bypassing the limitations of traditional resolution techniques. By utilizing specialized chiral quaternary ammonium salts, manufacturers can access high-value intermediates with superior optical purity, streamlining the supply chain for complex API production. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic advancements is crucial for optimizing procurement strategies and ensuring the consistent quality of raw materials entering the drug development pipeline.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
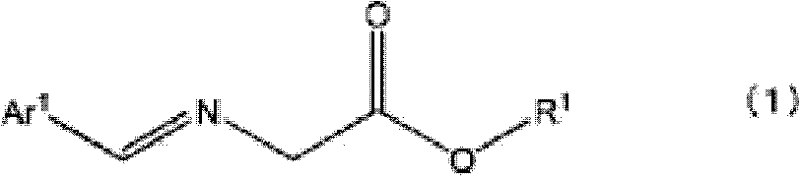
Historically, the manufacture of optically active 1-amino-2-vinylcyclopropanecarboxylates has relied heavily on enzymatic resolution of racemic mixtures, a process fraught with inherent inefficiencies and yield losses. Conventional protocols typically involve reacting N-benzylidene glycinate with (E)-1,4-dibromo-2-butene to generate a racemic product, followed by protection of the amino group and subsequent enzymatic splitting. A significant drawback of this legacy approach is the tendency for the unwanted enantiomer to racemize during the enzymatic resolution step, making its recovery and reuse technically challenging and economically unviable. Furthermore, the multi-step nature of protection and deprotection adds substantial operational complexity, increasing solvent consumption and waste generation. For procurement managers focused on cost reduction in API manufacturing, these inefficiencies translate into higher raw material costs and extended lead times, creating bottlenecks in the production of essential antiviral medications.
The Novel Approach
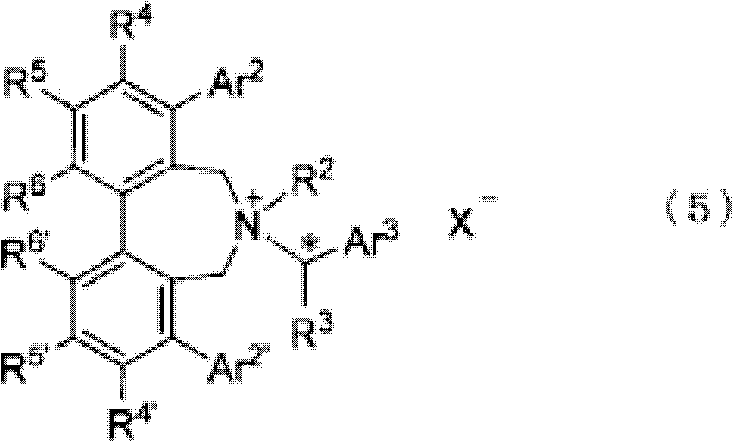
In stark contrast, the novel methodology disclosed in the patent employs a direct asymmetric synthesis strategy driven by chiral phase-transfer catalysis, fundamentally altering the economic and technical landscape of this intermediate's production. By reacting an N-(arylmethylene)glycine ester with a compound such as (E)-1,4-dihalo-2-butene in the presence of a base and a specifically designed optically active quaternary ammonium salt, the desired enantiomer is generated directly with high selectivity. This approach eliminates the need for enzymatic resolution and the associated racemization issues, thereby maximizing atom economy and overall yield. The use of robust chiral catalysts, such as those represented by Formula (5) or Formula (6) in the patent, allows for precise control over stereochemistry under mild biphasic conditions. This shift not only simplifies the synthetic route but also enhances the scalability of the process, offering a compelling value proposition for supply chain heads seeking more reliable and continuous manufacturing workflows.
Mechanistic Insights into Asymmetric Phase-Transfer Catalysis
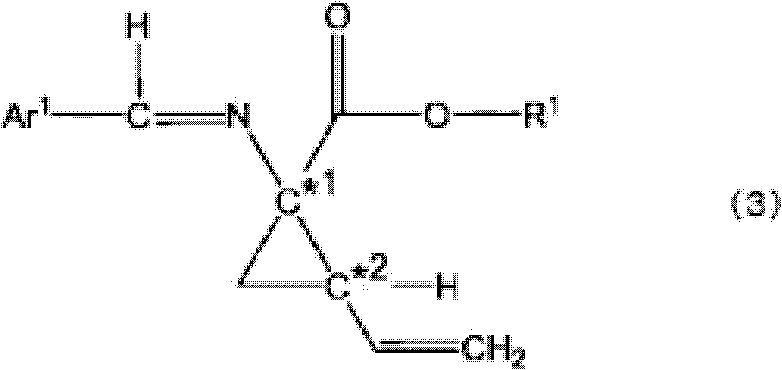
The core of this technological breakthrough lies in the sophisticated interaction between the chiral quaternary ammonium salt catalyst and the reactive intermediates within a biphasic solvent system. The catalyst, often a cinchona alkaloid derivative or a C2-symmetric spiro-type salt, functions by transporting the deprotonated glycine ester anion from the aqueous phase into the organic phase as a tight ion pair. Within the chiral environment created by the bulky substituents of the catalyst, the nucleophilic attack on the dihalo-butene electrophile is strictly controlled, favoring the formation of one specific enantiomer of the cyclopropane ring. This precise spatial arrangement ensures that the newly formed asymmetric centers at C*1 and C*2 possess the desired configuration, typically yielding products with optical purities ranging from 40% e.e. to over 95% e.e. directly from the reaction mixture. Understanding this mechanism is vital for R&D directors aiming to replicate or optimize these conditions, as subtle changes in catalyst structure or solvent composition can significantly impact the stereochemical outcome.
Furthermore, the process incorporates a unique impurity control mechanism that addresses the formation of diastereoisomers, which are common byproducts in cyclopropanation reactions. The patent details how any formed diastereoisomers (3c-d) can be effectively converted into a 7-membered ring compound (Formula 7) under the reaction conditions or via mild heating, effectively removing them from the product stream. This self-correcting feature simplifies the purification process, as the desired optically active compound (3) remains distinct and separable from the cyclized byproduct. Subsequent imine hydrolysis converts the protected intermediate into the final free amine (Formula 4) without compromising the established stereochemistry. This robust control over the impurity profile ensures that the final API intermediate meets stringent regulatory standards for purity, reducing the burden on downstream analytical and purification teams.
How to Synthesize Optically Active 1-Amino-2-Vinylcyclopropanecarboxylate Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly the choice of base, solvent system, and catalyst loading, to ensure optimal yield and enantioselectivity. The process typically begins with the preparation of the N-(arylmethylene)glycine ester, which reacts with the dihalo-butene derivative in a mixture of water and an organic solvent such as toluene or tert-butyl methyl ether. The addition of a strong base like potassium hydroxide initiates the formation of the reactive enolate, which is then captured by the chiral catalyst to facilitate the cyclopropanation. Detailed standard operating procedures for scaling this reaction from gram to kilogram quantities involve precise temperature control, often maintaining the reaction between 0°C and room temperature to minimize side reactions. For a comprehensive breakdown of the specific molar ratios, mixing sequences, and workup procedures required to achieve reproducible results, please refer to the standardized synthesis guide below.
- React N-(arylmethylene)glycine ester with (E)-1,4-dihalo-2-butene in the presence of a chiral quaternary ammonium salt and base.
- Perform imine hydrolysis on the resulting optically active 1-N-(arylmethylene)amino-2-vinylcyclopropanecarboxylate to remove the protecting group.
- Purify the final product by forming a salt with an achiral acid such as sulfuric acid or halobenzoic acid to enhance optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced catalytic process offers substantial strategic benefits for organizations focused on optimizing their chemical supply chains and reducing overall manufacturing expenditures. By shifting from enzymatic resolution to direct asymmetric synthesis, companies can eliminate the costs associated with expensive enzymes and the complex infrastructure required for biocatalytic processes. The use of commercially available and inexpensive reagents, such as potassium hydroxide and simple glycine esters, drastically lowers the raw material cost base. Additionally, the ability to recycle or use low loadings of the chiral catalyst contributes to significant cost savings over time, making the process economically attractive for large-scale production. For procurement managers, this translates into a more stable pricing model for critical intermediates, shielding the organization from volatility associated with specialized biocatalysts.
- Cost Reduction in Manufacturing: The elimination of enzymatic steps and the use of inexpensive inorganic bases significantly lower the operational expenditure per kilogram of product. The streamlined workflow reduces solvent usage and energy consumption associated with multiple protection and deprotection cycles, leading to a leaner and more cost-efficient manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: Relying on robust chemical catalysis rather than sensitive biological enzymes improves the consistency and reliability of supply. The starting materials are commodity chemicals with well-established global supply networks, reducing the risk of shortages and ensuring continuous production capability even during market fluctuations, which is critical for maintaining drug availability.
- Scalability and Environmental Compliance: The biphasic reaction system is inherently scalable, allowing for seamless transition from pilot plant to commercial production without major process redesigns. Furthermore, the reduction in waste generation and the avoidance of heavy metal catalysts align with green chemistry principles, simplifying environmental compliance and waste disposal logistics for sustainability-focused organizations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Whether you are evaluating the feasibility of in-house production or sourcing from external partners, these answers highlight the key performance indicators such as optical purity, yield, and purification methods that define the success of this manufacturing route.
Q: What are the advantages of this phase-transfer catalysis method over enzymatic resolution?
A: This method avoids the racemization of undesired isomers often seen in enzymatic resolution, allowing for direct asymmetric synthesis with high optical purity and simplified downstream processing.
Q: Can the optical purity be further improved after the initial reaction?
A: Yes, the optical purity can be significantly enhanced to over 99% e.e. by forming and recrystallizing salts with achiral acids like sulfuric acid or 4-chlorobenzoic acid.
Q: Is this process suitable for large-scale commercial production?
A: Absolutely. The process utilizes mild reaction conditions (0°C to room temperature), inexpensive bases like potassium hydroxide, and low catalyst loadings, making it highly scalable and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Amino-2-Vinylcyclopropanecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of antiviral therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-amino-2-vinylcyclopropanecarboxylate meets the highest industry standards. Our commitment to technical excellence allows us to navigate complex synthetic challenges, delivering materials that facilitate faster clinical progression and regulatory approval for our partners.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce time-to-market for your vital pharmaceutical products.
