Scalable Metal-Free Synthesis of Alpha-Carbonyl Amides for Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access critical building blocks, and the preparation method disclosed in patent CN109096139B represents a significant leap forward in the synthesis of alpha-carbonyl amide derivatives. This innovative technology utilizes a novel oxidative ring-opening strategy where pyridoimidazole amine compounds react with water under the influence of an oxidant to insert an oxygen atom, effectively cleaving the heterocyclic ring to form the desired amide structure. Unlike conventional methods that often rely on harsh conditions or toxic heavy metal catalysts, this approach operates under mild conditions ranging from room temperature to 60°C, offering a greener alternative for large-scale manufacturing. The process is particularly notable for its ability to utilize water molecules directly from crystalline hydrates or undried solvents as the oxygen source, which drastically simplifies the reaction setup and reduces the dependency on strictly anhydrous environments. By achieving high yields across a broad spectrum of substrates, including those with complex heterocyclic and substituted phenyl groups, this method addresses long-standing challenges in the production of high-purity pharmaceutical intermediates.
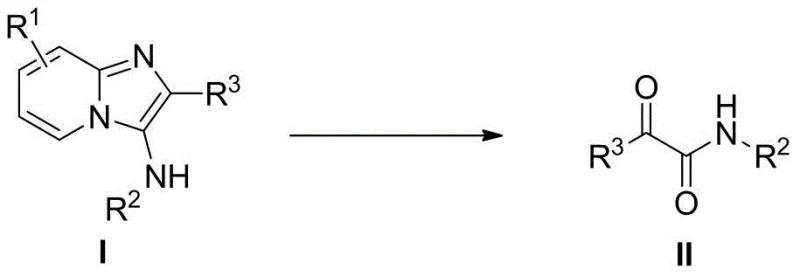
Traditional synthetic routes for alpha-carbonyl amides have historically been plagued by significant limitations that impact both cost and environmental compliance in industrial settings. Conventional strategies often involve small molecule coupling reactions or metal-catalyzed oxidations using copper or other transition metals, which necessitate rigorous purification steps to remove residual metal impurities to meet stringent regulatory standards for active pharmaceutical ingredients. These metal-catalyzed processes frequently require specialized ligands, inert atmospheres, and elevated temperatures, leading to increased energy consumption and operational complexity. Furthermore, the disposal of heavy metal waste streams poses a substantial environmental burden and increases the overall cost of goods sold due to waste treatment fees. In contrast, the novel approach detailed in the patent circumvents these issues by employing a metal-free catalytic system that relies on hypervalent iodine oxidants like PhI(OAc)2. This shift not only eliminates the risk of metal contamination but also streamlines the downstream processing, as the byproducts generated are easier to separate and the reaction conditions are far more forgiving regarding moisture and oxygen exposure.
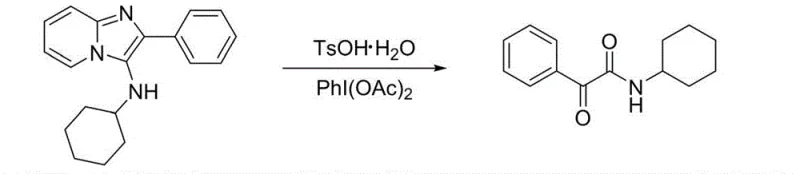
The mechanistic insights into this oxidative ring-opening reaction reveal a sophisticated yet elegant transformation driven by the unique reactivity of the pyridoimidazole scaffold. The reaction initiates with the activation of the imidazole ring by the hypervalent iodine oxidant, which facilitates the nucleophilic attack by water molecules introduced into the system. This attack leads to the insertion of an oxygen atom at the alpha-position relative to the carbonyl group, followed by the cleavage of the carbon-nitrogen bond within the heterocyclic ring. The presence of acids such as TsOH·H2O plays a dual role, acting both as a source of water and as a proton donor to facilitate the ring-opening event. This mechanism ensures that the oxygen atom in the final alpha-carbonyl structure is derived directly from water, highlighting the atom-economic nature of the process. The robustness of this mechanism is evidenced by its tolerance to various electronic effects on the aromatic rings, allowing for the successful synthesis of derivatives containing electron-withdrawing nitro groups and electron-donating methoxy groups without compromising the integrity of the reaction pathway. Additionally, the method effectively handles steric hindrance, as seen in the successful conversion of substrates with bulky tert-butyl and cyclohexyl amine groups, demonstrating the versatility required for diverse drug discovery programs.
How to Synthesize Alpha-Carbonyl Amide Derivatives Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must follow a streamlined protocol that leverages the mild conditions and broad substrate tolerance of the reaction. The process begins by charging the specific pyridoimidazole amine starting material into a standard reaction vessel equipped with a stirrer, followed by the addition of an organic solvent such as 1,2-dichloroethane or dichloromethane. A crucial aspect of this method is the flexible introduction of water, which can be achieved by adding crystalline hydrates like TsOH·H2O or simply by using undried solvents, removing the need for expensive drying agents or molecular sieves. Once the oxidant, typically iodobenzene diacetate, is added, the mixture is stirred at ambient temperatures for a short duration, usually between one to two hours, until thin-layer chromatography indicates complete consumption of the starting material. The workup procedure is equally straightforward, involving a simple separation of the organic phase followed by column chromatography to isolate the pure alpha-carbonyl amide product, often in yields exceeding 90 percent for optimized substrates. For detailed standardized operating procedures and safety guidelines, please refer to the technical documentation below.
- Charge the pyridoimidazole amine substrate into a reaction vessel with an organic solvent such as 1,2-dichloroethane.
- Introduce a water source, which can be crystalline hydrates like TsOH·H2O or undried solvents, along with the oxidant PhI(OAc)2.
- Stir the mixture at room temperature to 60°C for 1-2 hours, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free oxidative ring-opening technology offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of transition metal catalysts removes the necessity for expensive scavenging resins and complex filtration systems, resulting in substantial cost savings in raw materials and waste disposal. Furthermore, the ability to run reactions at room temperature significantly reduces energy consumption compared to traditional high-temperature reflux processes, contributing to a lower carbon footprint and reduced utility costs for the manufacturing facility. The simplified purification workflow also shortens the overall production cycle time, allowing for faster turnaround on custom synthesis orders and improved responsiveness to market demands. By utilizing readily available and inexpensive reagents like TsOH·H2O and common organic solvents, the supply chain becomes less vulnerable to fluctuations in the availability of specialized catalysts or ligands. This robustness ensures a more reliable supply of critical intermediates, mitigating the risk of production delays that can cascade through the entire pharmaceutical development pipeline.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts fundamentally alters the cost structure of producing alpha-carbonyl amides by eliminating the expensive downstream purification steps required to meet ppm-level metal specifications. Without the need for specialized metal scavengers or repeated recrystallizations to remove copper residues, the overall processing time is drastically reduced, leading to lower labor and overhead costs per kilogram of product. Additionally, the high atom economy of using water as an oxygen source minimizes the generation of stoichiometric byproducts, further reducing the volume of chemical waste that requires treatment. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediate, providing a clear economic advantage over legacy synthetic routes.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as hypervalent iodine oxidants and simple organic acids ensures that the supply chain is not dependent on single-source suppliers of exotic ligands or rare earth metals. This diversification of raw material sources enhances supply security, protecting manufacturing schedules from geopolitical disruptions or shortages of specialized reagents. The mild reaction conditions also allow for the use of standard glass-lined or stainless steel reactors without the need for specialized corrosion-resistant equipment, increasing the number of qualified contract manufacturing organizations capable of executing the process. This flexibility empowers procurement teams to negotiate better terms and secure capacity more easily across a broader network of potential partners.
- Scalability and Environmental Compliance: The inherent safety of operating at near-ambient temperatures and pressures makes this process highly scalable from gram-scale discovery to multi-ton commercial production without significant engineering hurdles. The absence of pyrophoric reagents or highly toxic gases simplifies the safety profile of the plant, reducing insurance premiums and regulatory compliance burdens associated with hazardous operations. Moreover, the generation of benign byproducts aligns with increasingly strict environmental regulations regarding industrial effluent, facilitating smoother permitting processes for new manufacturing lines. This alignment with green chemistry principles not only future-proofs the supply chain against tightening environmental laws but also enhances the brand reputation of the end-product as being sustainably manufactured.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative ring-opening technology in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent literature to ensure accuracy and reliability for decision-makers. Understanding these nuances is critical for evaluating the feasibility of integrating this method into existing production workflows or new drug development projects. For further clarification on specific substrate compatibility or custom scale-up requirements, our technical team is available to provide detailed assessments.
Q: What are the primary advantages of this metal-free oxidation method over traditional copper-catalyzed routes?
A: This method eliminates the need for transition metal catalysts like copper, thereby removing the costly and time-consuming heavy metal removal steps typically required in API manufacturing. It also utilizes water as the oxygen source, enhancing atom economy and reducing hazardous waste generation compared to traditional stoichiometric oxidants.
Q: Can this synthesis protocol accommodate diverse functional groups on the aromatic ring?
A: Yes, the process demonstrates excellent substrate universality, successfully tolerating electron-withdrawing groups such as nitro and chloro substituents, as well as electron-donating groups like methoxy. It is also compatible with heterocyclic systems including thiophene and furan derivatives without significant yield loss.
Q: Is the reaction condition sensitive to moisture or require strict anhydrous environments?
A: Unlike many sensitive organometallic reactions, this protocol does not require strict anhydrous or oxygen-free conditions. In fact, water is a necessary reagent for the oxygen insertion step, and it can be introduced via crystalline hydrates or even undried organic solvents, simplifying operational requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Carbonyl Amide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting advanced synthetic methodologies like the metal-free oxidative ring-opening described in CN109096139B to maintain a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity alpha-carbonyl amide derivatives that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. By leveraging our state-of-the-art facilities and deep expertise in process chemistry, we can help you optimize this novel route to maximize yield and minimize cost, securing a stable supply of critical intermediates for your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. Contact us today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this metal-free protocol. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and accelerate your time to market.
