Scalable Visible Light Catalysis for High-Purity Indole C3 Alkylated Derivatives Manufacturing
Introduction to Patent CN108727246B: A Breakthrough in Metal-Free Indole Functionalization
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly indole derivatives which serve as critical cores in numerous bioactive molecules. Patent CN108727246B introduces a transformative preparation method for substituted indole C3 alkylated derivatives that fundamentally shifts the paradigm from traditional transition-metal catalysis to visible-light-mediated organophotocatalysis. This innovation leverages inexpensive organic dyes, such as Eosin B and Eosin Y, acting as photosensitizers under mild LED white light irradiation to drive the alkylation reaction at room temperature. By replacing harsh acidic conditions or precious metal catalysts with abundant organic dyes and visible light, this technology addresses key pain points in modern process chemistry, including environmental impact, catalyst cost, and product purity. For R&D directors and procurement managers alike, this patent represents a significant opportunity to optimize synthetic routes for high-value pharmaceutical intermediates while adhering to stricter green chemistry principles.
The core innovation lies in the specific combination of reactants and conditions that enable efficient C3-selective alkylation without the need for inert atmospheres or extreme temperatures. The process utilizes substituted indoles and substituted N-methylaniline compounds as starting materials, reacting them in solvents like acetonitrile or aqueous acetonitrile mixtures. The reaction is monitored via TLC and typically proceeds to completion within 18 to 48 hours, yielding the desired C3 alkylated products with good efficiency. This approach not only simplifies the operational complexity of the synthesis but also mitigates the risks associated with heavy metal contamination, a critical factor for API intermediate manufacturing where residual metal limits are increasingly stringent. As we delve deeper into the technical specifics, it becomes clear that this methodology offers a robust platform for the commercial scale-up of complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of indoles at the C3 position has relied heavily on acid-catalyzed Friedel-Crafts type reactions or transition-metal-catalyzed cross-coupling strategies. While effective in many contexts, these conventional methods suffer from significant drawbacks that hinder their applicability in large-scale, cost-sensitive manufacturing environments. Acid-catalyzed processes often require strong protic or Lewis acids, which can lead to substrate decomposition, polymerization, or poor regioselectivity, necessitating complex purification steps to isolate the desired isomer. Furthermore, the use of strong acids generates corrosive waste streams that require specialized handling and disposal infrastructure, increasing the overall environmental footprint and operational costs of the facility.
Transition-metal catalysis, particularly using Ruthenium or Iridium complexes for photoredox applications, has offered milder alternatives but introduces its own set of challenges. These precious metal catalysts are exceptionally expensive, subject to volatile market pricing, and often difficult to remove completely from the final product. For pharmaceutical applications, ensuring that residual metal levels comply with ICH Q3D guidelines adds a layer of complexity and cost to the downstream processing, often requiring additional scavenging steps or chromatography. Moreover, the synthesis of these metal complexes themselves can be resource-intensive. The reliance on such scarce resources creates supply chain vulnerabilities, making the long-term sustainability of these processes questionable for high-volume commodity chemicals or intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN108727246B utilizes readily available organic dyes such as Eosin B, Eosin Y, Rhodamine B, and Rose Bengal as photocatalysts. These organic molecules are not only orders of magnitude cheaper than their transition-metal counterparts but are also non-toxic and environmentally benign. The reaction proceeds under the irradiation of a simple LED white light source, typically ranging from 5 to 30 Watts, with a preference for 15 to 20 Watts, which is easily scalable using standard photochemical reactor arrays. This shift to organophotocatalysis eliminates the need for expensive metal ligands and the associated purification burdens, directly translating to a cleaner crude product profile and reduced manufacturing costs.
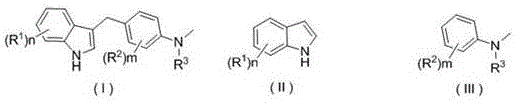
The operational simplicity of this novel approach is further enhanced by its tolerance to air and moisture, allowing reactions to be conducted under ambient atmospheric conditions rather than requiring rigorous inert gas purging. The solvent system is flexible, supporting pure organic solvents like acetonitrile and tetrahydrofuran, as well as greener aqueous mixtures where water and acetonitrile are combined in volume ratios ranging from 1:3 to 1:20. This flexibility allows process chemists to optimize solubility and reaction kinetics while minimizing organic solvent usage. The result is a highly efficient, selective, and economically viable pathway to substituted indole C3 alkylated derivatives that aligns perfectly with the goals of modern sustainable manufacturing.
Mechanistic Insights into Organic Dye Photocatalyzed C3 Alkylation
The mechanistic underpinning of this transformation involves a visible-light-induced single electron transfer (SET) process mediated by the excited state of the organic dye photosensitizer. Upon absorption of photons from the LED white light source, the organic dye (e.g., Eosin B) transitions from its ground state to an excited singlet state, which rapidly undergoes intersystem crossing to a longer-lived triplet excited state. This excited species acts as a potent oxidant or reductant, facilitating the generation of reactive radical intermediates from the N-methylaniline substrate. Specifically, the excited dye likely oxidizes the amine to form an aminium radical cation, which subsequently undergoes deprotonation to generate an alpha-amino radical. This nucleophilic radical species then attacks the electron-rich C3 position of the indole ring, forming a new carbon-carbon bond.
Following the radical addition, the resulting indolyl radical intermediate is oxidized by the oxidized form of the photocatalyst (or by oxygen in the air, given the reaction is run under aerobic conditions), regenerating the ground state catalyst and restoring aromaticity to the indole system. This catalytic cycle ensures that only a small loading of the dye, typically between 1.0 mol% and 10.0 mol%, is required to drive the reaction to completion. The mildness of this radical mechanism is crucial for impurity control; because the reaction occurs at room temperature without strong acids or bases, sensitive functional groups such as esters, halides, and ethers remain intact. This chemoselectivity minimizes the formation of side products like polymerized indoles or over-alkylated species, thereby simplifying the isolation process and improving the overall purity of the final intermediate.
Furthermore, the use of air as a terminal oxidant or co-catalyst in some variations of this mechanism eliminates the need for stoichiometric chemical oxidants, which often generate hazardous waste. The regeneration of the catalyst through interaction with molecular oxygen or the substrate radical cation ensures high turnover numbers and sustained catalytic activity throughout the 18 to 48-hour reaction window. Understanding this mechanism allows process engineers to fine-tune parameters such as light intensity, oxygen flow rate, and catalyst concentration to maximize space-time yield. For R&D teams, this mechanistic clarity provides a solid foundation for troubleshooting and optimizing the synthesis of specific analogues within the indole family, ensuring robust technology transfer from the laboratory to the pilot plant.
How to Synthesize Substituted Indole C3 Alkylated Derivatives Efficiently
To implement this technology effectively, manufacturers must adhere to the specific parameters outlined in the patent to ensure reproducibility and high yield. The process begins with the precise weighing of the substituted indole and the N-methylaniline derivative, typically employing a molar ratio of indole to aniline ranging from 1:1.0 to 1:10.0, with a preferred range of 1:4.0 to 1:6.0 to drive the reaction equilibrium forward. These reagents are dissolved in the chosen solvent system, such as acetonitrile or an acetonitrile-water mixture, within a transparent reaction vessel like a Schlenk tube or a quartz flask to allow maximum light penetration. The organic dye photocatalyst is then added at the optimized loading, usually between 1.5 mol% and 5.0 mol% for best results.
- Combine substituted indole and substituted N-methylaniline compounds in a suitable solvent such as acetonitrile or acetonitrile/water mixture.
- Add an organic dye photosensitizer like Eosin B or Eosin Y-2Na (1.0-10.0 mol%) to the reaction mixture under air atmosphere.
- Irradiate the mixture with a white LED light source (15-20 W) at room temperature for 18-48 hours, monitoring progress via TLC until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light-mediated synthesis offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The most immediate impact is seen in raw material costs; replacing precious metal catalysts like Ruthenium or Iridium complexes with commodity organic dyes such as Eosin B results in a drastic reduction in catalyst expenditure. These dyes are produced on a multi-ton scale for the textile and paper industries, ensuring a stable and abundant supply chain that is immune to the geopolitical volatility often associated with platinum group metals. This stability allows for more accurate long-term budgeting and reduces the risk of production stoppages due to catalyst shortages.
Additionally, the simplified workup procedure contributes significantly to cost reduction in fine chemical manufacturing. Since the process avoids heavy metals, the extensive and costly purification steps required to meet residual metal specifications are largely eliminated. This not only saves on chromatography media and scavenger resins but also reduces solvent consumption and waste disposal fees. The ability to run reactions at room temperature under air atmosphere further lowers energy costs by removing the need for cryogenic cooling or heating mantles, as well as the infrastructure for inert gas blanketing. These cumulative efficiencies translate into a lower cost of goods sold (COGS), providing a competitive edge in the marketplace for high-purity pharmaceutical intermediates.
From a supply chain reliability perspective, the scalability of LED photochemistry is well-established. Modern flow chemistry reactors and large-scale batch photoreactors equipped with high-power LED arrays can easily accommodate the 15 to 20 W per unit requirement described in the patent, allowing for seamless translation from gram-scale optimization to multi-kilogram production. The robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring consistent quality and delivery timelines for downstream customers. By integrating this technology, suppliers can offer reducing lead time for high-purity intermediates, as the streamlined process reduces the total manufacturing cycle time. Ultimately, this method supports a more agile and responsive supply chain capable of meeting the dynamic demands of the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light catalyzed indole alkylation technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN108727246B, providing clarity for stakeholders evaluating this process for potential adoption. Understanding these details is essential for assessing the feasibility of integrating this method into existing manufacturing workflows and for appreciating the specific advantages it holds over legacy synthetic routes.
Q: What are the primary advantages of using organic dye photocatalysis over transition metal catalysis for indole alkylation?
A: The primary advantages include the elimination of expensive and toxic transition metals like Ruthenium or Iridium, which simplifies downstream purification and reduces heavy metal residue risks in pharmaceutical products. Additionally, organic dyes such as Eosin B are significantly cheaper and more readily available, leading to substantial raw material cost reductions.
Q: Can this visible light-mediated method be scaled up for commercial production?
A: Yes, the method utilizes standard LED white light sources and operates at room temperature under air atmosphere, which are conditions highly favorable for scale-up. The absence of stringent inert gas requirements and cryogenic cooling further enhances the feasibility of large-scale manufacturing in standard reactor setups.
Q: What types of substituents are tolerated on the indole and aniline rings in this synthesis?
A: The protocol demonstrates excellent functional group tolerance, accommodating various substituents including alkyl groups, alkoxy groups, and halogens (Cl, Br, I) on both the indole and N-methylaniline moieties. This versatility allows for the synthesis of a diverse library of bioactive indole derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Indole C3 Alkylated Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible-light-mediated synthesis described in CN108727246B for the production of high-value indole intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this organophotocatalytic method are fully realized in a GMP-compliant manufacturing environment. We possess the specialized photochemical reactor infrastructure required to handle large-scale LED irradiation processes safely and efficiently, coupled with rigorous QC labs that enforce stringent purity specifications to guarantee product quality that exceeds industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced technology for their drug development pipelines. Whether you require custom synthesis of specific indole analogues or large-scale supply of key intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this metal-free, sustainable approach can enhance your supply chain resilience and reduce your overall manufacturing costs.
