Advanced Metal-Free Synthesis of 2-Methylthio-Maleic Diesters for Commercial Pharmaceutical Production
Introduction to Next-Generation Olefin Functionalization
The patent CN113072472A introduces a groundbreaking methodology for the synthesis of 2-methylmercapto-maleic diester compounds, a class of highly functionalized olefins that serve as critical building blocks in modern medicinal chemistry and agrochemical development. This innovative approach leverages a multi-component one-pot reaction strategy involving butynedioic acid diester, elemental iodine, and dimethyl sulfoxide (DMSO) to construct carbon-sulfur bonds with exceptional efficiency. Unlike traditional pathways that often rely on complex catalytic systems or harsh conditions, this protocol operates under mild thermal conditions without the necessity for additional transition metal catalysts, thereby addressing significant purity and environmental concerns prevalent in the industry. The technical breakthrough lies in the dual role of DMSO, which acts not only as a green solvent but also as the sulfur donor, decomposing in situ to facilitate the transformation. For R&D directors and process chemists, this represents a paradigm shift towards more sustainable and atom-economical synthetic routes that minimize waste generation while maximizing yield.
From a commercial perspective, the ability to produce high-purity pharmaceutical intermediates through such a streamlined process offers substantial advantages for supply chain stability and cost management. The elimination of heavy metal catalysts removes the need for expensive scavenging steps and rigorous testing for residual metals, which are critical bottlenecks in API manufacturing. Furthermore, the use of inexpensive and readily available reagents like iodine and DMSO ensures that the raw material costs remain low, supporting the economic viability of large-scale production. This synthesis method is particularly relevant for companies seeking a reliable pharma intermediate supplier who can deliver complex molecules with consistent quality and reduced lead times. By adopting this technology, manufacturers can enhance their competitive edge in the global market through improved process robustness and environmental compliance.
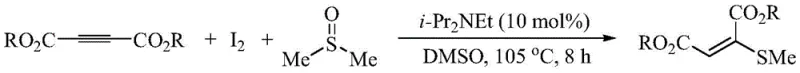
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-sulfur bonds around the butynedioic acid diester scaffold has been fraught with challenges related to stereoselectivity, reagent toxicity, and operational complexity. Previous methodologies, such as those reported by Sarrafi et al., utilized aryl thiophenols in aqueous solutions, which often resulted in limited substrate scope and difficulties in controlling the stereochemistry of the resulting fumaric acid diester products. Other approaches involving nucleophilic reactions with arylthiolates in chloroform solvents, as described by Griffith et al., introduced additional hazards due to the use of chlorinated solvents and required precise stoichiometric control to avoid over-reaction or side product formation. Furthermore, photocatalytic methods for constructing benzothiophene heterocycles, while innovative, often demand specialized equipment and energy-intensive light sources, complicating the scale-up process for industrial applications. These conventional routes frequently suffer from poor atom economy and generate significant amounts of chemical waste, posing environmental burdens and increasing disposal costs for manufacturing facilities.
The Novel Approach
In stark contrast, the novel method disclosed in the patent utilizes a simple yet highly effective combination of elemental iodine and DMSO to achieve the desired transformation in a single pot. This approach bypasses the need for pre-functionalized sulfur reagents or expensive metal catalysts, relying instead on the thermal decomposition of DMSO to generate the reactive methyl mercaptan species in situ. The reaction proceeds with high selectivity, predominantly yielding the Z-isomer (maleic derivative) rather than the E-isomer, which is a significant advantage for downstream synthetic applications requiring specific geometric configurations. The operational simplicity of mixing the reagents and heating to moderate temperatures (100-110°C) makes this process inherently safer and easier to control compared to photochemical or cryogenic alternatives. Additionally, the byproduct of the reaction is hydrogen iodide, which is neutralized by the amine base, further simplifying the workup procedure and enhancing the overall green chemistry profile of the synthesis.
Mechanistic Insights into Iodine-Mediated Thio-Functionalization
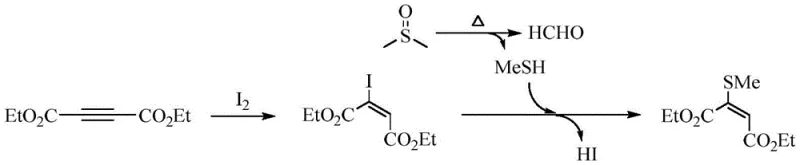
The mechanistic pathway of this transformation is a fascinating example of cascade reactivity where iodine acts as an electrophilic activator for the alkyne moiety. Initially, the elemental iodine undergoes an addition reaction with the electron-deficient triple bond of the butynedioic acid diester to form a transient 2-iodo-maleic acid diester intermediate. This iodination step is crucial as it activates the vinyl position for subsequent nucleophilic attack, effectively lowering the energy barrier for the substitution reaction. Simultaneously, the dimethyl sulfoxide solvent undergoes thermal decomposition at the reaction temperature of 105°C, releasing methyl mercaptan (MeSH) and formaldehyde. The generated methyl mercaptan then acts as a potent nucleophile, attacking the iodine-activated carbon center to displace the iodide ion and form the final carbon-sulfur bond. This concerted mechanism ensures high regioselectivity and minimizes the formation of bis-substituted byproducts, which are common pitfalls in direct thiolation reactions.
Understanding this mechanism is vital for impurity control and process optimization, as the stoichiometry of iodine plays a pivotal role in driving the reaction to completion without excess halogen contamination. Experimental data indicates that maintaining the iodine dosage at approximately 50% of the molar amount of the diester is optimal; deviations from this ratio can lead to incomplete conversion or the accumulation of iodinated intermediates. The presence of a tertiary amine base, specifically diisopropylethylamine (DIPEA), is also critical for scavenging the hydrogen iodide produced during the substitution step, thereby preventing acid-catalyzed degradation of the sensitive ester groups. This delicate balance of reagents ensures that the reaction proceeds cleanly to the target 2-methylthio-maleic diester with yields consistently exceeding 90%, demonstrating the robustness of the chemical design.
How to Synthesize Diethyl 2-(methylthio)maleate Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric ratios and thermal profiles to maximize yield and purity. The standard protocol involves charging a reaction vessel with the diacetylene diester substrate, followed by the addition of the amine catalyst and elemental iodine in DMSO solvent. The mixture is then heated to 105°C and maintained under stirring for 8 hours to ensure complete conversion of the starting material. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the exact endpoint, although the established time frame is generally sufficient for most substrates. Upon completion, the reaction mixture is quenched with water and extracted with ethyl acetate, followed by standard washing and drying procedures to isolate the crude product. For detailed standardized operating procedures and safety guidelines regarding this specific synthesis route, please refer to the technical guide below.
- Combine butynedioic acid diester, diisopropylethylamine (10 mol%), iodine (50 mol%), and excess dimethyl sulfoxide in a reaction flask.
- Stir the mixture at 105°C for approximately 8 hours under monitored conditions.
- Quench with water, extract with ethyl acetate, wash with brine, dry, and purify to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this metal-free synthesis route offers profound strategic benefits for procurement managers and supply chain leaders looking to optimize their sourcing strategies for complex organic intermediates. By eliminating the reliance on precious metal catalysts such as palladium or copper, the process drastically reduces the raw material costs associated with catalytic systems and the subsequent purification steps required to meet regulatory limits on heavy metals. This cost reduction in pharmaceutical intermediate manufacturing is achieved not only through cheaper reagents but also through simplified downstream processing, which lowers energy consumption and solvent usage. Furthermore, the use of DMSO as both solvent and reactant streamlines the material inventory, reducing the number of SKUs that need to be managed and stored, thereby enhancing operational efficiency. These factors collectively contribute to a more resilient supply chain that is less vulnerable to fluctuations in the prices of specialty catalysts or rare earth metals.
- Cost Reduction in Manufacturing: The exclusion of transition metal catalysts removes the significant expense associated with purchasing, recovering, or disposing of these materials, leading to substantial cost savings per kilogram of product. Additionally, the high atom economy of the reaction means that a larger proportion of the starting materials are incorporated into the final product, minimizing waste disposal fees and improving overall process efficiency. The mild reaction conditions also reduce energy costs compared to high-temperature or high-pressure alternatives, further contributing to the economic viability of the process. Qualitative analysis suggests that the simplified workup procedure reduces labor hours and solvent volumes, providing a leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including elemental iodine, DMSO, and diisopropylethylamine, are commodity chemicals with stable and abundant global supply chains. This availability ensures that production schedules are not disrupted by shortages of exotic reagents or specialized catalysts, guaranteeing consistent delivery timelines for customers. The robustness of the reaction conditions also implies a lower risk of batch failures due to sensitive parameters, enhancing the predictability of output volumes. Consequently, partners can rely on a steady flow of high-quality intermediates, reducing the need for excessive safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The one-pot nature of the reaction facilitates easy scale-up from gram to ton quantities without the need for complex reactor modifications or additional unit operations. The absence of toxic heavy metals aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the process attractive for green chemistry initiatives. Waste streams are simpler to treat due to the lack of metal contaminants, reducing the environmental impact and compliance burden on manufacturing sites. This scalability ensures that the technology can meet growing market demands for functionalized olefins in the pharmaceutical and agrochemical sectors without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-methylthio-maleic diesters, based on the detailed experimental data provided in the patent literature. These insights are designed to clarify the operational parameters and potential advantages for stakeholders evaluating this technology for integration into their supply chains. Understanding these nuances helps in making informed decisions about process adoption and vendor selection.
Q: Why is this metal-free synthesis preferred over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, significantly simplifying downstream purification and reducing heavy metal residue risks in pharmaceutical intermediates.
Q: What is the role of dimethyl sulfoxide (DMSO) in this reaction?
A: DMSO serves a dual function as both the benign reaction solvent and the sulfur source, decomposing thermally to generate methyl mercaptan in situ for the nucleophilic substitution.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction utilizes cheap, commercially available raw materials like iodine and DMSO under mild conditions (105°C), making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylthio-Maleic Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the development of next-generation therapeutics and crop protection agents. Our team of expert process chemists has extensively evaluated the metal-free synthesis of 2-methylthio-maleic diesters and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-purity intermediates regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications and complies with international regulatory standards. By leveraging this innovative technology, we can offer our partners a competitive advantage through superior product quality and reliable delivery performance.
We invite pharmaceutical and agrochemical companies to collaborate with us to explore how this advanced synthesis method can optimize your specific project needs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing processes, highlighting potential efficiencies and budget improvements. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample quantities for your evaluation. Together, we can accelerate your development timelines and bring valuable new products to market faster and more economically.
