Scalable Visible Light Catalyzed Dichlorination for High-Purity Pharmaceutical Intermediates
Introduction to Next-Generation Dichlorination Technology
The synthesis of vicinal dichlorides represents a critical transformation in the production of high-value pharmaceutical intermediates and agrochemical precursors. Traditional methods often suffer from harsh conditions and limited substrate scope, creating bottlenecks in supply chains for complex molecules. However, the groundbreaking methodology disclosed in patent CN111253254B introduces a paradigm shift by utilizing visible light photocatalysis to drive the dichlorination of aliphatic olefins. This innovative approach leverages the unique photochemical properties of copper chloride to generate chlorine radicals under exceptionally mild conditions, specifically at room temperature and in the presence of air. By replacing hazardous thermal processes with a green, energy-efficient photochemical protocol, this technology offers a robust pathway for manufacturing high-purity intermediates. The strategic use of 38W white LEDs provides the necessary photon flux to initiate Ligand-to-Metal Charge Transfer (LMCT), enabling the activation of inexpensive hydrochloric acid as a chlorine source. This not only simplifies the operational workflow but also drastically reduces the environmental footprint associated with halogenation reactions.
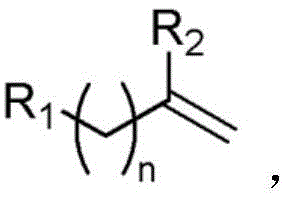
For procurement specialists and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic elegance of this patent is crucial. The process accommodates a broad spectrum of substrates, including those with C9-C15 aliphatic chains covalently linked to carbon-carbon double bonds, which are prevalent in lipid-mimetic drug candidates. The ability to perform these transformations without inert gas protection or cryogenic cooling translates directly into lower capital expenditure for reactor infrastructure. Moreover, the compatibility with diverse functional groups ensures that downstream synthetic steps remain viable, preserving the integrity of sensitive moieties often found in late-stage intermediates. As the industry moves towards sustainable manufacturing, adopting such visible-light-driven protocols positions companies at the forefront of green chemistry innovation, ensuring long-term regulatory compliance and market competitiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing bis-chloro compounds have been plagued by significant technical and economic drawbacks that hinder scalable production. For instance, methods reported by Babak Borhan utilize expensive chiral catalysts and large quantities of inorganic salts, which not only inflate raw material costs but also complicate waste stream management. Similarly, protocols involving potassium peroxymonosulfonate and ammonium chloride, as described by Rendy Kartika, rely on dangerous inorganic oxidants that pose severe safety hazards and environmental pollution risks. Other traditional routes, such as those employing triphosgene and pyridine described by James B. Hendrickson, require complex precursor synthesis like epoxy compounds, adding unnecessary steps and reducing overall atom economy. Furthermore, electrocatalytic methods using manganese catalysts often necessitate hazardous electrolytes like lithium perchlorate, introducing explosion risks and demanding specialized equipment. These conventional thermal reactions typically require high activation energy, leading to energy-intensive processes that are difficult to control and often result in poor selectivity and low yields.
The Novel Approach
In stark contrast, the visible light-catalyzed method presented in CN111253254B offers a streamlined, safe, and highly efficient alternative that addresses these legacy issues. By utilizing copper chloride, a transition metal salt with inherent visible light absorption capabilities, the process initiates chlorine radical formation through a photo-induced electron transfer mechanism rather than thermal decomposition. This allows the reaction to proceed at ambient temperatures (23-25°C), eliminating the need for heating and the associated energy costs. The use of commercial hydrochloric acid as the chlorine source is a game-changer, as it is inexpensive, readily available, and avoids the logistical nightmares of transporting toxic chlorine gas or unstable solid chlorinating agents. Additionally, the reaction operates effectively in an aerobic atmosphere, removing the requirement for costly inert gas purging systems. This novel approach not only enhances operational safety but also significantly simplifies the workup procedure, making it an ideal candidate for cost reduction in fine chemical manufacturing where efficiency and safety are paramount.
Mechanistic Insights into CuCl2-Catalyzed Photo-Chlorination
The core of this technological breakthrough lies in the sophisticated interplay between visible light energy and the electronic structure of the copper catalyst. Upon irradiation with 38W white LEDs, the copper(II) chloride complex undergoes a Ligand-to-Metal Charge Transfer (LMCT) excitation. This photo-excitation weakens the Cu-Cl bond, facilitating the homolytic cleavage that generates highly reactive chlorine radicals. These radicals then engage in a selective addition across the carbon-carbon double bond of the aliphatic olefin substrate, forming a carbon-centered radical intermediate. Crucially, the presence of molecular oxygen from the air serves as a terminal oxidant, regenerating the copper(II) species from the reduced copper(I) state formed during the radical propagation cycle. This catalytic turnover ensures that only a catalytic amount of copper (20 mol%) is required, maximizing atom efficiency. The mild nature of the radical generation prevents non-selective background reactions that often plague thermal radical processes, thereby ensuring high regioselectivity and minimizing the formation of poly-chlorinated byproducts.

From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material precursors and pharmaceutical intermediates. The room temperature conditions suppress thermal degradation pathways and rearrangement reactions that are common in heated systems. For example, in the conversion of N-allylphthalimide derivatives (as seen in Example 1), the reaction achieves an impressive 85% yield with minimal side products, demonstrating the system's tolerance to nitrogen-containing heterocycles. The radical addition is kinetically controlled by the photon flux rather than thermal energy, allowing for precise modulation of reaction rates. Furthermore, the use of acetonitrile as a polar aprotic solvent stabilizes the charged intermediates and facilitates the solubility of the inorganic catalyst, ensuring a homogeneous reaction environment. This level of control is essential for maintaining stringent purity specifications required by global regulatory bodies, reducing the burden on downstream purification units.
How to Synthesize Dichloride Addition Products Efficiently
Implementing this visible light catalytic protocol requires careful attention to reaction parameters to maximize yield and reproducibility. The standard procedure involves dissolving the aliphatic olefin substrate in acetonitrile, followed by the addition of copper(II) chloride and aqueous hydrochloric acid. The mixture is then subjected to continuous irradiation using high-power white LEDs while being stirred in an open vessel to allow air exchange. The reaction typically reaches completion within 72 hours, after which standard aqueous workup and chromatographic purification afford the target dichloride. This straightforward operational sequence minimizes the need for specialized training or complex equipment, making it accessible for both laboratory-scale optimization and pilot plant operations. The detailed standardized synthesis steps below outline the precise stoichiometry and conditions validated in the patent examples.
- Combine the aliphatic olefin substrate with 20 mol% CuCl2 catalyst and 2.5 equivalents of aqueous HCl in acetonitrile solvent within a reaction vessel.
- Irradiate the reaction mixture with 38W white LEDs while stirring at room temperature (23-25°C) in an air atmosphere for 72 hours to ensure complete conversion.
- Quench the reaction with saturated sodium sulfite solution, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this visible light catalytic technology presents a compelling value proposition centered on cost efficiency and operational resilience. The elimination of expensive noble metal catalysts such as iridium, ruthenium, or palladium directly translates to substantial raw material cost savings, as copper chloride is a commodity chemical with a stable global supply. Additionally, the avoidance of hazardous reagents like triphosgene or perchlorates reduces the costs associated with safety compliance, waste disposal, and insurance premiums. The ability to run reactions at room temperature using ambient air significantly lowers utility costs related to heating, cooling, and inert gas consumption. These factors collectively contribute to a more lean and agile manufacturing process, capable of responding quickly to market demands without the bottlenecks associated with complex thermal setups.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with earth-abundant copper chloride results in a drastic reduction in catalyst procurement costs. Since the catalyst loading is low (20 mol%) and the metal is inexpensive, the overall cost of goods sold (COGS) for the final intermediate is significantly optimized. Furthermore, the use of commercial hydrochloric acid eliminates the need for synthesizing or purchasing specialized chlorinating agents, streamlining the supply chain and reducing inventory holding costs. The energy efficiency of running reactions at room temperature with LED lighting further drives down operational expenditures, making the process economically superior to traditional thermal methods.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like CuCl2 and HCl ensures a stable and secure supply chain, mitigating the risks associated with the scarcity or price volatility of exotic reagents. The simplicity of the reaction setup, which does not require high-pressure reactors or cryogenic systems, allows for greater flexibility in manufacturing site selection and capacity expansion. This robustness ensures consistent delivery timelines for clients, as the production process is less susceptible to equipment failures or utility interruptions. The broad substrate scope also means that a single production line can be adapted for various intermediates, enhancing asset utilization and supply continuity.
- Scalability and Environmental Compliance: The green chemistry credentials of this method facilitate easier regulatory approval and environmental compliance, as it generates less hazardous waste and avoids toxic byproducts. The scalability is supported by the use of flow chemistry-compatible LED arrays, allowing for seamless transition from gram-scale to multi-ton production. The mild conditions reduce the risk of thermal runaway, a critical safety factor when scaling up exothermic halogenation reactions. This combination of safety, sustainability, and scalability makes the technology an ideal choice for commercial scale-up of complex polymer additives and fine chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible light catalytic dichlorination technology. These insights are derived directly from the experimental data and technical specifications outlined in patent CN111253254B, providing a clear understanding of its practical applications and benefits. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary cost advantages of this visible light catalytic method compared to traditional thermal chlorination?
A: This method eliminates the need for expensive noble metal catalysts like iridium or ruthenium, utilizing instead abundant and low-cost copper chloride. Furthermore, it operates at room temperature using air as the oxidant, significantly reducing energy consumption and safety costs associated with high-pressure or high-temperature reactors.
Q: Does this process support a wide range of functional groups for complex intermediate synthesis?
A: Yes, the patent demonstrates excellent substrate tolerance, successfully converting olefins containing esters, amides, sulfonamides, and various aromatic substituents including halogens and nitro groups without requiring protective group strategies.
Q: How does the use of hydrochloric acid as a chlorine source impact operational safety?
A: Using commercial hydrochloric acid avoids the handling risks associated with toxic gaseous chlorine or unstable solid chlorinating agents like DCDMH. The reaction proceeds under mild aerobic conditions, minimizing the risk of runaway exothermic reactions common in traditional radical chlorinations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dichloride Addition Products Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible light catalysis in modernizing the synthesis of critical chemical intermediates. Our team of expert chemists has extensively evaluated the methodology described in CN111253254B and confirmed its viability for producing high-quality dichloride derivatives. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch delivered meets the highest international standards.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this green protocol can optimize your budget. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering value through innovation and reliability in the supply of high-performance chemical intermediates.
