Advanced Synthesis of 3,5-Diiodo-2-Hydroxybenzoyl Chloride for Veterinary Drug Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic routes for critical intermediates that balance efficiency with safety and environmental compliance. Patent CN102206152A introduces a significant advancement in the chemical synthesis of 3,5-diiodo-2-hydroxybenzoyl chloride, a pivotal precursor for the broad-spectrum anthelmintic drug Closantel Sodium. This veterinary pharmaceutical is essential for treating parasitic infections in livestock, representing a high-value market segment where supply chain reliability is paramount. The disclosed method replaces traditional, hazardous chlorinating agents with bis(trichloromethyl) carbonate, offering a pathway that drastically reduces operational risks while maintaining high yields and purity standards. For R&D directors and procurement managers, understanding this technological shift is crucial for optimizing the cost reduction in pharmaceutical intermediates manufacturing and ensuring a sustainable supply of high-quality raw materials.
The Limitations of Conventional Methods vs. The Novel Approach
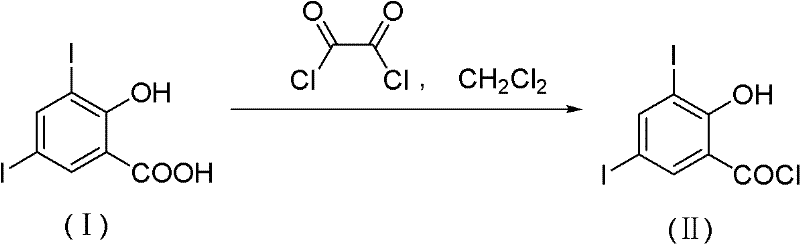
The Limitations of Conventional Methods
Historically, the synthesis of 3,5-dihalo-2-hydroxybenzoyl chlorides has relied heavily on aggressive chlorinating reagents such as oxalyl chloride or thionyl chloride. As illustrated in the reaction scheme below, the conventional route using oxalyl chloride often requires strict anhydrous conditions and generates toxic byproducts that complicate waste management. Oxalyl chloride is a volatile liquid with high toxicity and strong corrosiveness, posing severe safety hazards during transportation and storage due to its violent decomposition upon contact with moisture. Furthermore, alternative methods using thionyl chloride produce large volumes of suffocating sulfur dioxide gas, a pollutant strictly regulated by environmental agencies. The treatment of such acidic waste gases requires sophisticated scrubbing systems, increasing capital expenditure and operational complexity for manufacturers aiming for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
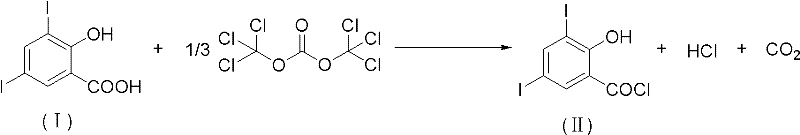
In contrast, the novel approach detailed in the patent utilizes bis(trichloromethyl) carbonate (also known as triphosgene) as a solid, stable substitute for liquid acid chlorides. This reagent acts as a safe source of phosgene equivalents, releasing the active chlorinating species in a controlled manner within the reaction medium. The reaction proceeds smoothly in the presence of catalytic amounts of organic amines, such as pyridine or DMF, within common organic solvents like toluene or dichloroethane. This methodology not only eliminates the handling risks associated with volatile liquids but also simplifies the post-reaction workup, as the byproducts are easier to manage than sulfur dioxide. The transition to this solid-state reagent technology represents a strategic improvement for any reliable veterinary drug intermediate supplier looking to enhance their production safety profile and reduce environmental liability while delivering high-purity pharmaceutical intermediates to the global market.
Mechanistic Insights into Amine-Catalyzed Acylation
The core of this synthesis lies in the nucleophilic acyl substitution mechanism facilitated by the organic amine catalyst. When bis(trichloromethyl) carbonate interacts with the carboxylic acid group of 3,5-diiodo-2-hydroxybenzoic acid, the amine catalyst activates the carbonyl carbon, making it more susceptible to nucleophilic attack. This activation lowers the energy barrier for the formation of the mixed anhydride intermediate, which subsequently collapses to release the desired acid chloride and carbon dioxide. The choice of catalyst, ranging from tertiary amines like triethylamine to heterocyclic amines like pyridine, plays a critical role in tuning the reaction rate and selectivity. For R&D teams, optimizing the molar ratio of the catalyst to the substrate is key to minimizing side reactions, such as the potential chlorination of the phenolic hydroxyl group, thereby ensuring the structural integrity of the final product.
Impurity control is another vital aspect of this mechanistic pathway. The mild reaction conditions, typically ranging from 40°C to 150°C, prevent thermal degradation of the sensitive diiodo-substituted aromatic ring. Unlike harsher methods that might lead to dehalogenation or polymerization, this catalytic system preserves the iodine atoms, which are crucial for the biological activity of the downstream anthelmintic drug. The use of solvents like toluene or xylene allows for easy removal of water formed during the reaction via azeotropic distillation, further driving the equilibrium towards product formation. This precise control over the reaction environment ensures that the impurity profile remains within tight specifications, a requirement for any high-purity API intermediate intended for veterinary use where residue limits are strictly enforced.
How to Synthesize 3,5-Diiodo-2-Hydroxybenzoyl Chloride Efficiently
The practical implementation of this synthesis involves a straightforward sequence of mixing, heating, and isolation steps that are amenable to standard chemical processing equipment. Operators begin by charging the reactor with the starting acid, the solid carbonate reagent, and the chosen solvent, followed by the addition of the catalytic amine. The mixture is then heated to the specified temperature range and monitored, often using thin-layer chromatography, until the starting material is fully consumed. This operational simplicity reduces the training burden on plant personnel and minimizes the risk of human error during batch execution. For detailed procedural specifics regarding stoichiometry and temperature ramps, please refer to the standardized guide below.
- Mix 3,5-diiodo-2-hydroxybenzoic acid with bis(trichloromethyl) carbonate and an organic amine catalyst in a suitable organic solvent such as toluene or dichloroethane.
- Heat the reaction mixture to a temperature between 40°C and 150°C and maintain stirring for 1 to 10 hours to ensure complete conversion.
- Remove the organic solvent via reduced pressure distillation, filter the resulting concentrate, and dry the solid product to obtain high-purity 3,5-diiodo-2-hydroxybenzoyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of bis(trichloromethyl) carbonate offers profound logistical and economic benefits over traditional chlorinating agents. The shift from hazardous liquids to a stable solid reagent fundamentally alters the storage and transportation requirements, eliminating the need for specialized corrosion-resistant tanks and reducing insurance costs associated with dangerous goods. This stability ensures a more consistent supply of raw materials, mitigating the risk of production stoppages due to reagent degradation or delivery delays. For procurement managers, this translates into a more resilient supply chain capable of meeting fluctuating market demands for veterinary pharmaceuticals without compromising on safety or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of complex gas scrubbing systems required for sulfur dioxide neutralization leads to significant capital and operational expenditure savings. Additionally, the high atom economy and yield of the reaction reduce the consumption of raw materials per unit of product, directly lowering the variable cost of goods sold. The simplified workup procedure, involving basic distillation and filtration, reduces energy consumption and labor hours, contributing to overall manufacturing efficiency and improved profit margins for producers of veterinary drug intermediates.
- Enhanced Supply Chain Reliability: Bis(trichloromethyl) carbonate is commercially available in large quantities and possesses a long shelf life, unlike oxalyl chloride which requires careful handling and has stricter transport regulations. This availability ensures that manufacturers can maintain adequate inventory levels to buffer against market volatility. The robustness of the process also means that production timelines are more predictable, reducing lead time for high-purity pharmaceutical intermediates and allowing for faster response to customer orders in the competitive animal health sector.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of organic solvents that can be recovered and recycled, aligning with green chemistry principles. The absence of toxic gas emissions simplifies environmental permitting and reduces the regulatory burden on manufacturing facilities. This environmental friendliness facilitates easier commercial scale-up, as the process can be expanded from pilot plants to multi-ton reactors without encountering the bottlenecks associated with waste gas treatment capacity, ensuring long-term sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 3,5-diiodo-2-hydroxybenzoyl chloride. These insights are derived directly from the patented technology and are intended to clarify the advantages of this method for potential partners and stakeholders. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your existing supply chain or development pipeline.
Q: Why is bis(trichloromethyl) carbonate preferred over oxalyl chloride for this synthesis?
A: Bis(trichloromethyl) carbonate is a solid reagent that is significantly safer to transport and store compared to the highly toxic and corrosive liquid oxalyl chloride. It eliminates the release of hazardous sulfur dioxide gas associated with thionyl chloride methods, simplifying waste treatment and improving workplace safety.
Q: What represents the typical purity profile for this intermediate?
A: The patented process consistently achieves high product purity, often exceeding 97% as determined by HPLC analysis. The use of specific organic amine catalysts helps minimize side reactions, ensuring a clean impurity profile suitable for downstream veterinary drug synthesis.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. The reaction conditions are moderate, and the work-up procedure involves simple distillation and filtration, avoiding complex purification steps. This makes it highly viable for commercial scale-up from pilot batches to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Diiodo-2-Hydroxybenzoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the production of effective veterinary medicines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3,5-diiodo-2-hydroxybenzoyl chloride meets the highest industry standards, providing you with a reliable foundation for your final drug formulation.
We invite you to collaborate with us to optimize your sourcing strategy for this essential intermediate. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced synthesis method can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us support your journey towards more efficient and sustainable pharmaceutical manufacturing.
