Advanced NHC Catalysis for Commercial Scale Chiral Sulfonamide Production
The global pharmaceutical industry constantly seeks robust and scalable methods for constructing complex chiral scaffolds essential for next-generation therapeutics. Patent CN112898318A introduces a novel N-heterocyclic carbene catalyzed pathway that fundamentally alters the synthesis landscape. This method specifically targets chiral polycyclic compounds containing sulfonamide frameworks which are pivotal in modern drug design and agrochemical applications. The process achieves excellent yields reaching up to 89 percent and exceptional enantioselectivity exceeding 99 percent ee. It represents a significant strategic shift from traditional transition metal catalysis which often leaves toxic residues. This innovation offers a cleaner and more efficient route for producing high-value intermediates. Consequently it addresses critical purity concerns faced by regulatory bodies worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic methods for similar polycyclic structures often rely heavily on stoichiometric or catalytic amounts of transition metals like palladium or rhodium. These precious metals are inherently expensive and subject to volatile market pricing which impacts budget stability. More critically they require rigorous and costly removal steps to meet stringent international safety standards for pharmaceutical ingredients. Residual metal contamination poses severe risks for final drug product approval and can lead to batch rejection. Furthermore traditional routes may suffer from poor atom economy and frequently necessitate harsh reaction conditions to drive conversion. High temperatures and extreme pressures increase energy consumption and introduce significant operational hazards for plant personnel. Side reactions often lead to complex impurity profiles that are chemically difficult to separate using standard chromatography. This increases the overall cost of goods sold and extends development timelines significantly for process chemistry teams.
The Novel Approach
The novel approach utilizes advanced organocatalysis via stable N-heterocyclic carbenes which act as efficient metal-free activators. This eliminates the need for heavy metals entirely thereby removing the burden of expensive scavenging processes. The reaction proceeds under remarkably mild conditions typically at room temperature which preserves sensitive functional groups. Substrate universality is extensively demonstrated across twenty-five diverse examples in the patent data showing broad applicability. The mechanism involves a unique cyclization cascade that constructs multiple bonds in a single operation. This ensures high stereocontrol without the need for external chiral ligands which are often costly to source. The process simplifies downstream processing significantly by reducing the number of unit operations required. Ultimately this leads to a greener manufacturing footprint and improved overall process mass intensity.
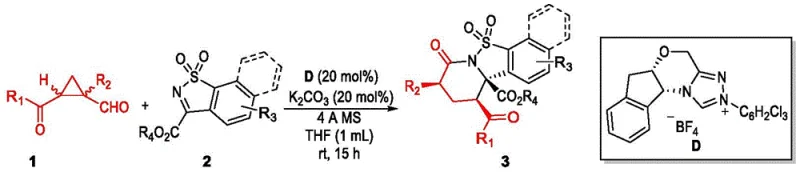
Mechanistic Insights into NHC-Catalyzed Asymmetric Cyclization
The catalytic cycle begins with the reversible formation of a key Breslow intermediate between the catalyst and the aldehyde. The N-heterocyclic carbene activates the 1-alkyl-1-cyclopropylcarbaldehyde substrate effectively through umpolung chemistry. This generates a highly nucleophilic homoenolate equivalent species ready for subsequent carbon-carbon bond formation. The substituted sulfimide acts as a precise electrophile in this carefully balanced system to accept the nucleophile. Ring expansion occurs through a coordinated sequence of bond breaking and making events involving the cyclopropane moiety. This forms the core polycyclic sulfonamide structure efficiently while maintaining the integrity of the stereocenters. The catalyst is regenerated at the end of the cycle allowing for turnover and minimal loading requirements. Understanding this flow is critical for optimizing reaction parameters during technology transfer to production scales.
Stereocontrol is achieved through highly organized transition states dictated by the bulky groups on the carbene catalyst. Severe steric repulsion guides the facial selectivity during the critical Mannich-type addition step. The Re-face attack is thermodynamically favored over the Si-face approach due to these spatial constraints. This results in the observed major enantiomer with optical purity exceeding ninety-nine percent enantiomeric excess. Impurity formation is minimized by this intrinsic selectivity which prevents the generation of unwanted diastereomers. The diastereomeric ratio is consistently high across various substrate combinations as evidenced by the data. This reduces the need for extensive and yield-lossing recrystallization steps during purification. Such robust control is essential for meeting the strict chiral specifications required by global regulatory agencies.
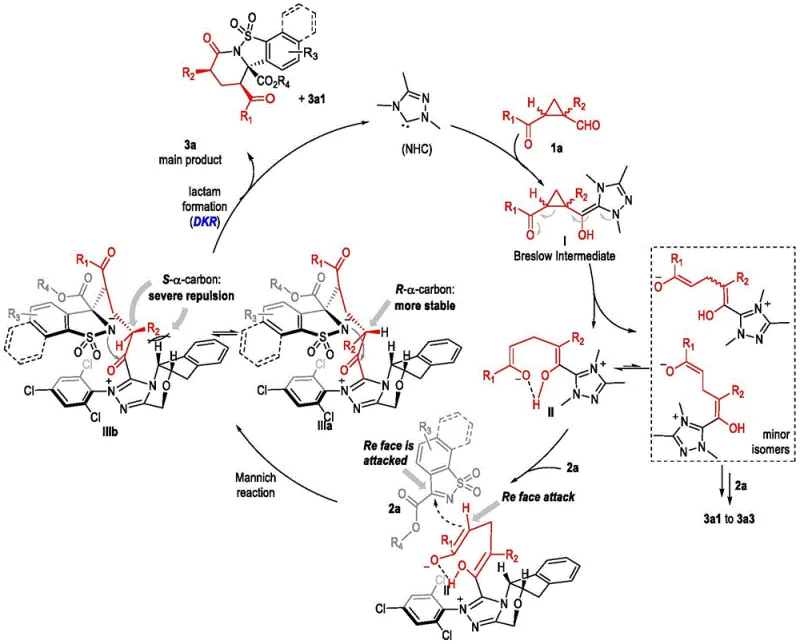
How to Synthesize Chiral Polycyclic Sulfonamide Efficiently
Efficient synthesis of these complex chiral molecules requires precise control over reaction parameters and environmental conditions. The patent outlines a standardized protocol designed for high reproducibility across different laboratory and plant settings. Operators must maintain strictly anhydrous conditions using molecular sieves to ensure catalyst stability and activity. Temperature control is vital for maintaining selectivity and preventing decomposition of the sensitive intermediates formed. The detailed standardized synthesis steps are provided in the guide below to assist process engineers in implementation. This ensures consistent quality across batches and minimizes variability in the final product specifications. Adhering to these guidelines allows for a smooth transition from bench scale to pilot plant operations.
- Prepare 1-alkyl-1-cyclopropylcarbaldehyde and substituted sulfimide substrates under anhydrous conditions.
- Mix substrates with N-heterocyclic carbene catalyst and potassium carbonate in THF solvent.
- Stir at room temperature for 15 hours and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
Procurement teams face increasing pressure to reduce costs while maintaining uncompromised quality standards for active ingredients. This technology offers substantial supply chain benefits by utilizing readily available starting materials and simple reagents. Raw materials such as substituted sulfonyl chlorides and aldehydes are commercially available from multiple global vendors. The process is inherently safer than traditional metal catalysis due to the absence of pyrophoric reagents or heavy metals. Waste generation is lower which simplifies disposal and reduces environmental compliance costs. These factors combine to create a more resilient and cost-effective sourcing strategy for long-term production needs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts directly reduces the raw material cost per kilogram of product. There is no need for specialized metal scavenging resins which are a significant expense in traditional processing. Lower raw material cost combined with reduced waste treatment fees contributes to a healthier margin profile. The mild reaction conditions also lower energy consumption compared to high-temperature alternatives. Operational expenditures are further optimized by the simplified workup procedure requiring fewer extraction steps. This economic advantage allows for competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on simple and commodity-grade substrates ensures a reliable supply of starting materials year-round. There is less dependency on a single source for specialized catalysts which often face availability bottlenecks. The robustness of the organocatalyst means it can be stored and handled without special inert atmosphere equipment. This reduces the risk of production delays caused by reagent degradation or shipping restrictions. Procurement managers can secure long-term contracts with greater confidence knowing the chemistry is not fragile. This stability is crucial for maintaining continuous manufacturing lines for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reaction demonstrates excellent scalability potential due to its exothermic profile being manageable at room temperature. Green chemistry principles are adhered to by avoiding toxic heavy metals and volatile organic solvents where possible. Less waste is generated which aligns with corporate sustainability goals and regulatory expectations. The process mass intensity is favorable compared to multi-step metal-catalyzed routes. Environmental compliance is easier to achieve reducing the administrative burden on EHS teams. This makes the technology attractive for companies aiming to reduce their carbon footprint in chemical manufacturing.
Frequently Asked Questions (FAQ)
Technical questions often arise during the technology transfer phase from R&D to commercial production units. We address common concerns here to facilitate smoother decision-making for stakeholders. Purity and yield are key topics discussed by quality assurance and control departments regularly. Scalability is another frequent question raised by engineering teams assessing plant fit. The answers below are based on patent data and practical chemical engineering principles. This transparency helps build trust between the supplier and the manufacturing partner. Clear communication resolves potential bottlenecks before they impact the project timeline.
Q: What is the primary advantage of using NHC catalysis over transition metals?
A: NHC catalysis eliminates the need for expensive and toxic heavy metals, reducing purification costs and environmental impact while maintaining high enantioselectivity.
Q: Is this synthesis process suitable for large-scale manufacturing?
A: Yes, the reaction operates under mild room temperature conditions with simple reagents, making it highly scalable and safe for industrial production.
Q: What level of enantioselectivity can be achieved with this method?
A: The method consistently achieves excellent enantioselectivity exceeding 99 percent ee across a wide range of substrate derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Polycyclic Compound Supplier
Partnering with NINGBO INNO PHARMCHEM provides access to a reliable chiral polycyclic compound supplier with deep technical expertise. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT annual commercial production volumes. Our facilities are equipped to handle complex organocatalytic reactions with stringent purity specifications required for global markets. Rigorous QC labs ensure every batch meets the highest standards before release. We understand the critical nature of supply continuity for your drug development pipelines. Our team is dedicated to supporting your long-term growth with consistent quality.
We invite you to contact our technical procurement team to discuss your specific requirements in detail. Request a Customized Cost-Saving Analysis to understand the economic benefits for your specific project. You may also ask for specific COA data and route feasibility assessments to verify compatibility. Our experts are ready to provide the information needed for your vendor qualification process. Initiating this dialogue early can accelerate your project timelines significantly. Let us help you optimize your supply chain for these high-value intermediates today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
