Advanced Transition-Metal-Free Synthesis of Indeno[2,1-b]indole-6(5H)-one Derivatives for Commercial Scale-up
Advanced Transition-Metal-Free Synthesis of Indeno[2,1-b]indole-6(5H)-one Derivatives for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic scaffolds that serve as core structures for bioactive molecules. Patent CN113620859A introduces a groundbreaking methodology for synthesizing indeno[2,1-b]indole-6(5H)-one derivatives, a privileged structural motif found in numerous natural products and therapeutic agents such as midostaurin and ellagitannins. This innovation addresses critical bottlenecks in traditional synthesis by employing a transition-metal-free, one-pot cascade reaction. By utilizing readily available 1-(2-bromophenyl)-2,2-dihydroxyethan-1-one compounds and 2-indolinones, the process achieves the construction of the tetracyclic skeleton under mild basic conditions without the need for expensive palladium or copper catalysts. This technical breakthrough offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to streamline their production pipelines while adhering to stringent regulatory standards regarding heavy metal residues.
![General reaction scheme for synthesizing indeno[2,1-b]indole-6(5H)-one derivatives using base and heat](/insights/img/indeno-indole-ketone-synthesis-pharma-supplier-20260304134229-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indole-fused indanone skeletons has been plagued by significant synthetic challenges that hinder efficient commercial scale-up. Early approaches, such as the three-step synthesis reported by Martarello et al., relied on the formation of aryl hydrazone compounds via diazonium salts, followed by Fischer indole synthesis. This pathway is not only cumbersome due to the multi-step nature but also involves hazardous diazonium intermediates that pose safety risks during large-scale operations. Furthermore, transition-metal-catalyzed methods, such as the palladium-catalyzed cyclocarbonylation described by Larock et al., introduce the persistent issue of metal contamination. In the context of cost reduction in API manufacturing, the removal of trace palladium or copper to meet ppm-level specifications requires additional purification steps, such as specialized scavenger resins or recrystallization, which drastically increase production costs and reduce overall yield. Additionally, methods involving Ullmann reactions with stoichiometric copper, as seen in later literature, generate substantial amounts of heavy metal waste, creating environmental compliance burdens and complicating waste stream management.
The Novel Approach
The methodology disclosed in CN113620859A represents a paradigm shift by enabling the direct assembly of the target scaffold through a base-mediated cascade reaction. By simply dissolving the bromoketone precursor and the 2-indolinone derivative in a polar aprotic solvent like DMSO and adding cesium carbonate (Cs2CO3), the reaction proceeds smoothly at temperatures between 100-140°C. This approach completely bypasses the need for transition metal catalysts, thereby eliminating the risk of metal leaching into the final product. The reaction is remarkably efficient, achieving the formation of two carbon-carbon bonds and one carbon-nitrogen bond in a single operational step. For procurement teams, this translates to a simplified bill of materials where expensive catalysts and ligands are removed from the equation. The use of inexpensive inorganic bases and common solvents further enhances the economic viability of the process, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
![Specific synthesis example of indeno[2,1-b]indol-6(5H)-one showing 72% yield](/insights/img/indeno-indole-ketone-synthesis-pharma-supplier-20260304134229-03.webp)
Mechanistic Insights into Base-Mediated Cascade Cyclization
Understanding the mechanistic pathway is crucial for R&D directors evaluating the robustness and reproducibility of this synthesis. The transformation is not a simple substitution but a sophisticated cascade involving multiple bond-breaking and bond-forming events. Initially, the 1-(2-bromophenyl)-2,2-dihydroxyethan-1-one undergoes a dehydration condensation with the 2-indolinone to form an exocyclic double bond intermediate. This is followed by an intramolecular Michael addition, which sets the stage for the subsequent ring closure. The key step involves a radical-nucleophilic aromatic substitution (SrNAr) mechanism, where the electron-rich indole system attacks the aryl bromide moiety. This is a rare and valuable transformation that typically requires harsh conditions or metal catalysis but is facilitated here by the specific electronic activation provided by the adjacent carbonyl and hydroxyl groups.
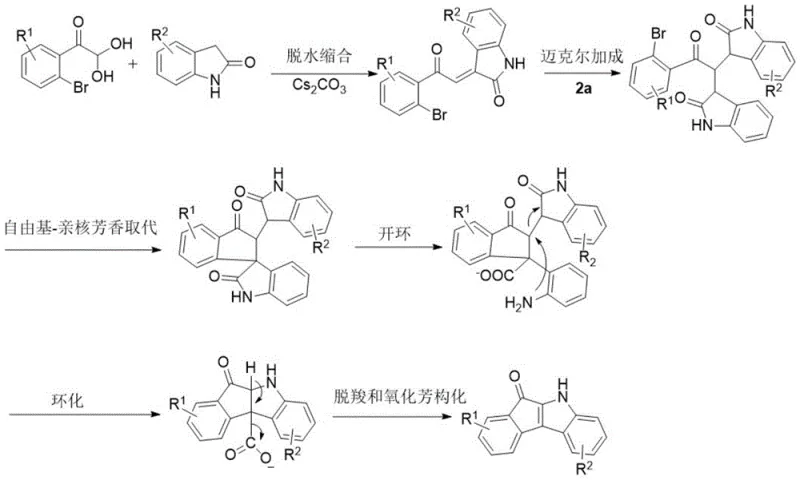
Following the initial cyclization, the mechanism proceeds through a fascinating ring-opening and reclosure sequence. The five-membered lactam ring of the indolinone opens, followed by decarboxylation and subsequent oxidative aromatization to restore aromaticity and finalize the indeno[2,1-b]indole core. This intricate dance of electrons results in the net fracture of one C-N and one C-C bond, accompanied by the formation of three new bonds. From an impurity control perspective, this mechanism is highly advantageous because it avoids the formation of stable organometallic intermediates that often lead to homocoupling byproducts. The absence of metal catalysts means that the impurity profile is dominated by organic side products which are generally easier to separate via standard chromatography or crystallization, ensuring high-purity pharmaceutical intermediates suitable for downstream drug synthesis.
How to Synthesize Indeno[2,1-b]indole-6(5H)-one Efficiently
The practical execution of this synthesis is designed for operational simplicity, minimizing the need for specialized equipment or inert gas manifolds beyond standard nitrogen protection. The protocol utilizes a pressure tube to maintain the solvent integrity at elevated temperatures, ensuring consistent reaction kinetics. The workup procedure is straightforward, involving standard aqueous extraction and drying, followed by purification via silica gel chromatography. This ease of handling makes the technology accessible for both laboratory-scale optimization and pilot plant production.
- Dissolve 1-(2-bromophenyl)-2,2-dihydroxyethan-1-one and 2-indolinone in DMSO solvent within a pressure tube.
- Add Cs2CO3 base (3 equivalents) and heat the mixture to 120°C under nitrogen atmosphere for 1 hour.
- Extract the reaction mixture with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this transition-metal-free methodology offers tangible strategic advantages beyond mere technical novelty. The primary benefit lies in the drastic simplification of the supply chain for raw materials. By removing the dependency on precious metal catalysts like palladium or copper, manufacturers are insulated from the volatile pricing and geopolitical supply risks associated with these commodities. Furthermore, the elimination of metal scavenging steps reduces the consumption of auxiliary materials and shortens the overall cycle time per batch. This efficiency gain allows for faster throughput and improved responsiveness to market demand fluctuations, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of removing transition metals cannot be overstated. Traditional cross-coupling reactions often require catalyst loadings that, while low in molar percentage, represent a significant cost center due to the high price of the metals and ligands. More importantly, the downstream cost of removing these metals to meet regulatory limits often exceeds the cost of the catalyst itself. By utilizing a base-mediated organic transformation, this process eliminates the need for expensive metal scavengers, specialized filtration equipment, and extensive ICP-MS testing for residual metals. This holistic reduction in processing requirements leads to substantial cost savings per kilogram of produced material, directly improving the margin profile for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain stability. The reagents used, such as cesium carbonate and DMSO, are commodity chemicals with stable global supply chains, unlike specialized organometallic catalysts which may have limited suppliers. Additionally, the tolerance of the reaction to various functional groups (halogens, esters, ethers) means that a wide range of substituted derivatives can be produced using the same standard operating procedure. This flexibility allows manufacturers to pivot quickly between different analogues without requalifying the entire process, ensuring a continuous and reliable supply of diverse building blocks for drug discovery and development programs.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles. The absence of heavy metals simplifies waste treatment protocols, as the aqueous waste streams do not require specialized heavy metal precipitation or containment procedures. The one-pot nature of the reaction minimizes solvent usage and energy consumption associated with intermediate isolation and drying. These factors collectively reduce the environmental footprint of the manufacturing process, facilitating easier regulatory approval and supporting corporate sustainability goals. The process is inherently scalable, as the exothermic profile is manageable and does not rely on sensitive catalytic cycles that might fail upon scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implications for industrial adoption.
Q: Why is the transition-metal-free aspect critical for pharmaceutical intermediates?
A: Traditional methods often utilize palladium or copper catalysts which leave toxic metal residues. Removing these residues requires expensive scavenging steps and rigorous testing. This novel method eliminates transition metals entirely, significantly simplifying downstream purification and ensuring compliance with strict ICH Q3D guidelines for elemental impurities.
Q: What is the substrate scope of this cyclization reaction?
A: The method demonstrates excellent functional group tolerance. It accommodates various substituents on both the phenyl ring of the bromoketone precursor (such as -OMe, -F, -Cl) and the benzene ring of the 2-indolinone (including -Me, -OMe, -F, -Cl, and ester groups), allowing for the diverse synthesis of derivatives needed for SAR studies.
Q: How does the one-pot nature of this reaction impact manufacturing efficiency?
A: By combining dehydration, Michael addition, radical-nucleophilic substitution, and oxidative aromatization into a single vessel, the process eliminates the need for isolating unstable intermediates. This reduces solvent consumption, labor hours, and overall waste generation, leading to a more sustainable and cost-effective manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indeno[2,1-b]indole-6(5H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this transition-metal-free synthesis route for the production of high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of indeno[2,1-b]indole-6(5H)-one derivatives meets the exacting standards required for clinical and commercial applications.
We invite pharmaceutical partners to leverage our technical expertise to optimize this route for their specific pipeline needs. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this metal-free process for your specific target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring a seamless integration of this advanced technology into your supply chain.
