Revolutionizing Pharmaceutical Intermediates: Scalable Rhodium-Catalyzed Synthesis of High-Purity Trifluoromethyl Enamine Compounds
Chinese patent CN115925692A introduces a groundbreaking methodology for synthesizing trifluoromethyl-substituted enamine compounds through a novel rhodium-catalyzed C-H activation process that represents a significant advancement in the field of complex molecule construction for pharmaceutical applications. This innovative approach addresses longstanding challenges in the synthesis of multifunctionalized enamine structures by eliminating the need for pre-functionalized substrates typically required in conventional transition metal-catalyzed reactions. The patent details a streamlined reaction pathway that operates under mild conditions with exceptional functional group tolerance, enabling the production of diverse enamine derivatives with indole and trifluoromethyl moieties that are highly valuable in drug discovery programs. By utilizing readily available starting materials and standard laboratory equipment, this method offers a practical solution for both research-scale synthesis and potential commercial manufacturing of bioactive molecules. The documented high yields exceeding 80% across multiple substrate variations demonstrate the robustness and versatility of this synthetic strategy, positioning it as a valuable tool for medicinal chemistry and process development teams working on next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing enamine compounds with complex substitution patterns typically rely on transition metal-catalyzed carbon-hydrogen activation reactions that require pre-installed directing groups on the indole scaffold, significantly limiting substrate scope and increasing synthetic complexity through additional functionalization steps. These conventional methodologies often suffer from narrow functional group tolerance, necessitating extensive protection/deprotection strategies that reduce overall efficiency and increase production costs for pharmaceutical intermediates. The requirement for specialized substrates with specific directing groups restricts structural diversity and hampers the ability to rapidly generate compound libraries for structure-activity relationship studies in drug discovery programs. Furthermore, many existing protocols employ harsh reaction conditions or expensive catalysts that complicate scale-up and create challenges for maintaining consistent product quality in commercial manufacturing environments. The limited substrate scope and intricate multi-step sequences inherent in traditional approaches result in lower overall yields and higher impurity profiles that require extensive purification efforts, making them less suitable for producing the high-purity intermediates demanded by modern pharmaceutical development.
The Novel Approach
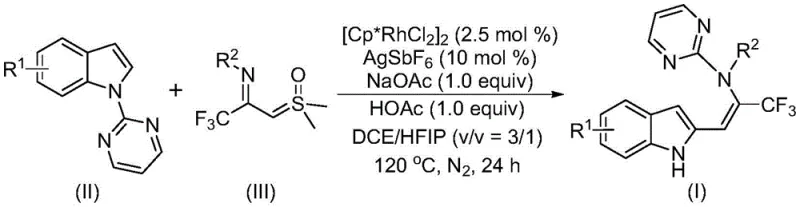
The patented methodology overcomes these limitations through an innovative rhodium-catalyzed tandem reaction that directly utilizes simple indole derivatives and trifluoroacetimide sulfur ylides without requiring pre-functionalization or specialized directing groups on the substrate. This streamlined process operates under relatively mild conditions (120°C) with a well-defined catalyst system comprising dichlorocyclopentyl rhodium(III) dimer, silver hexafluoroantimonate, and sodium acetate/acetic acid additives in a dichloroethane/hexafluoroisopropanol solvent mixture. The reaction demonstrates exceptional functional group tolerance across a wide range of substituents on both reaction partners, enabling the synthesis of diverse enamine structures with indole and trifluoromethyl moieties that were previously difficult to access through conventional methods. With documented yields consistently exceeding 80% across multiple substrate variations, this approach provides a practical solution for generating structurally diverse compound libraries essential for medicinal chemistry optimization campaigns. The straightforward reaction setup and standard purification protocol using column chromatography make this method readily adaptable to both laboratory-scale synthesis and potential commercial manufacturing operations, addressing key pain points in pharmaceutical intermediate production.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation for Enamine Synthesis
The reaction mechanism begins with rhodium(III)-catalyzed pyrimidine nitrogen-directed carbon-hydrogen activation of the indole substrate, forming a cyclometalated rhodium intermediate that subsequently reacts with the trifluoroacetimide sulfur ylide to establish a new carbon-carbon bond. This key step is facilitated by the unique electronic properties of the pyrimidine directing group, which enables selective activation of the indole C-H bond under mild conditions without requiring harsh reagents or elevated temperatures typically associated with traditional C-H activation protocols. The resulting intermediate undergoes isomerization to form an enamine structure, followed by an intramolecular pyrimidine group migration through a 1,5-migration process that ultimately delivers the final trifluoromethyl-substituted enamine product with high regioselectivity. This tandem sequence represents an elegant solution to constructing complex molecular architectures through multiple bond-forming events in a single reaction vessel, significantly improving atom economy and reducing waste generation compared to stepwise synthetic approaches.
The mechanism inherently minimizes impurity formation through its well-defined reaction pathway, where the rhodium catalyst directs selective bond formation while the carefully optimized additive system controls reaction selectivity and prevents undesired side reactions. The use of sodium acetate/acetic acid as additives creates a buffered environment that maintains optimal reaction conditions throughout the process, preventing decomposition of sensitive intermediates and ensuring consistent product quality across different substrate variations. The solvent system comprising dichloroethane and hexafluoroisopropanol provides the ideal polarity balance to solubilize all reaction components while facilitating the key migration steps through specific solvent-solute interactions that promote the desired transformation pathway. This precise control over reaction parameters results in exceptional product purity without requiring specialized purification techniques beyond standard column chromatography, making it particularly suitable for producing pharmaceutical intermediates that must meet stringent quality specifications.
How to Synthesize Trifluoromethyl Enamine Compounds Efficiently
This patented methodology represents a significant advancement in the synthesis of complex trifluoromethyl-substituted enamine compounds, offering pharmaceutical researchers and process chemists a reliable pathway to access these valuable building blocks with unprecedented efficiency and structural diversity. The reaction protocol has been meticulously optimized through extensive experimentation to balance yield, purity, and practicality for both research-scale applications and potential commercial manufacturing scenarios. By eliminating the need for pre-functionalized substrates and operating under relatively mild conditions, this approach substantially reduces synthetic complexity while maintaining excellent functional group tolerance across diverse substrate combinations. The detailed experimental procedures documented in the patent provide clear guidance for implementing this methodology in laboratory settings, with specific attention given to critical parameters that influence reaction success and product quality.
- Prepare the reaction mixture by combining indole compounds, trifluoroacetimide sulfur ylide, rhodium catalyst, promoter, and additives in dichloroethane/hexafluoroisopropanol solvent system.
- Conduct the reaction at 120°C under nitrogen atmosphere for 24 hours, monitoring reaction progress through standard analytical techniques.
- Perform post-reaction processing including filtration, silica gel mixing, and column chromatography purification to obtain high-purity trifluoromethyl enamine products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic approach addresses critical pain points in pharmaceutical intermediate supply chains by providing a robust manufacturing solution that enhances reliability while reducing complexity in the production of high-value enamine building blocks essential for drug development programs. The methodology's compatibility with standard manufacturing equipment and processes eliminates the need for specialized infrastructure investments, allowing seamless integration into existing production facilities without significant capital expenditures or operational disruptions. By utilizing readily available starting materials from established chemical suppliers, this process mitigates supply chain vulnerabilities associated with specialty chemicals while ensuring consistent raw material quality essential for maintaining product specifications across production batches.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps significantly reduces raw material costs while the high-yielding process minimizes waste generation and solvent consumption, creating substantial cost savings through improved process efficiency and reduced environmental impact management requirements. The use of commercially available catalysts and additives at optimized loadings further contributes to economic viability without compromising product quality or reaction performance.
- Enhanced Supply Chain Reliability: The broad availability of starting materials from multiple global suppliers creates redundancy in sourcing options while the straightforward reaction protocol enables rapid technology transfer between manufacturing sites, ensuring consistent product supply even during market fluctuations or regional supply constraints. This flexibility is particularly valuable for maintaining uninterrupted production of critical pharmaceutical intermediates required for ongoing drug development programs.
- Scalability and Environmental Compliance: The documented scalability from laboratory to commercial production with consistent high yields demonstrates excellent process robustness, while the simplified purification requirements reduce energy consumption and waste streams compared to traditional multi-step syntheses. The elimination of hazardous reagents and transition metal residues through optimized reaction design enhances environmental compliance and simplifies regulatory documentation for commercial manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical details documented in Chinese patent CN115925692A, addressing common concerns from procurement, R&D, and supply chain professionals regarding the implementation and benefits of this innovative synthetic methodology for pharmaceutical intermediate production. These insights reflect practical considerations based on the patent's experimental data and technical specifications, providing valuable guidance for organizations evaluating this technology for their manufacturing operations.
Q: How does this novel synthesis method improve upon conventional approaches for enamine compound production?
A: The patented rhodium-catalyzed C-H activation method eliminates the need for pre-functionalized substrates required in traditional transition metal-catalyzed approaches, significantly simplifying the synthetic route while maintaining high functional group tolerance and substrate diversity.
Q: What are the key advantages of this process for pharmaceutical manufacturing scale-up?
A: This method demonstrates excellent scalability from laboratory to commercial production with consistent high yields above 80%, utilizing readily available starting materials and standard purification techniques that integrate seamlessly with existing pharmaceutical manufacturing infrastructure.
Q: How does the process ensure high purity required for pharmaceutical applications?
A: The reaction mechanism inherently minimizes byproduct formation through selective C-H activation and controlled migration processes, while the straightforward purification protocol using column chromatography consistently delivers products meeting stringent pharmaceutical purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Enamine Compound Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global pharmaceutical clients. As a specialized CDMO provider with deep expertise in complex molecule synthesis, we have successfully implemented similar rhodium-catalyzed methodologies across multiple therapeutic areas, demonstrating our capability to translate innovative academic discoveries into robust commercial manufacturing processes. Our state-of-the-art facilities feature rigorous QC labs equipped with advanced analytical instrumentation to ensure consistent product quality meeting or exceeding industry standards for high-value pharmaceutical intermediates.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can optimize your supply chain for trifluoromethyl enamine compounds. Contact us today to discuss your specific requirements and request detailed COA data along with route feasibility assessments tailored to your manufacturing needs.
