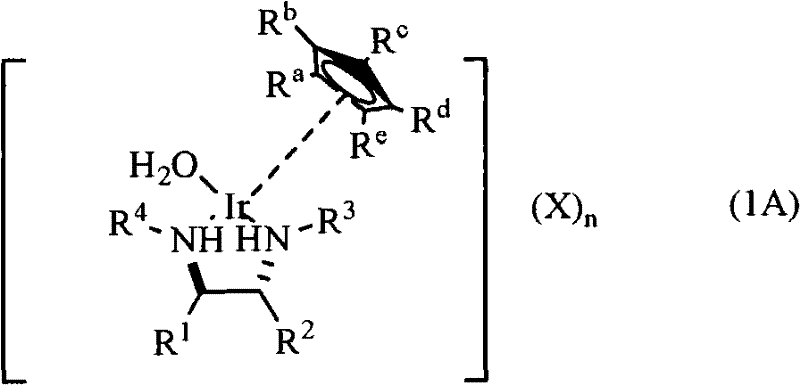
Advanced Chiral Iridium Aqua Complexes for Scalable Asymmetric Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign catalytic systems for the production of chiral intermediates. Patent CN102119165A introduces a significant breakthrough in this domain with the development of novel chiral iridium aqua complexes. These complexes, represented by general formula (1A), address critical limitations found in prior art, such as poor water solubility and safety hazards associated with perchlorate counterions. By utilizing a sulfate anion system and specific chiral diamine ligands, this technology enables asymmetric transfer hydrogenation to proceed safely and effectively in aqueous solvents. This innovation not only aligns with modern green chemistry mandates but also offers a robust pathway for generating high-purity optically active hydroxy compounds and nitroalkanes, which are essential building blocks in drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
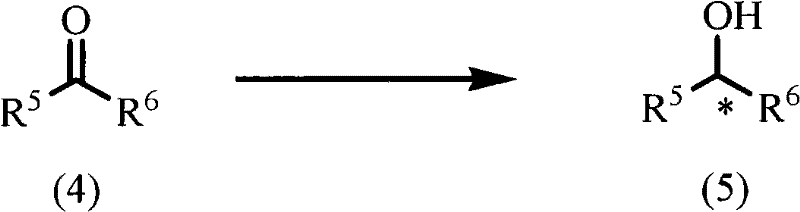
Historically, the preparation of optically active hydroxy compounds via asymmetric transfer hydrogenation has relied heavily on ruthenium-based catalysts like Ru(II)-TsDPEN or non-aqueous iridium complexes. While effective, these traditional methods suffer from significant drawbacks that hinder large-scale adoption. Ruthenium catalysts often exhibit slower reaction kinetics compared to their iridium counterparts, leading to longer processing times and reduced throughput in manufacturing settings. Furthermore, many existing iridium complexes are poorly soluble in water, necessitating the use of volatile organic compounds (VOCs) which increase environmental burden and disposal costs. Perhaps most critically, some prior art complexes utilize perchlorate anions, which present severe safety risks, including potential explosiveness when in contact with hydrogen sources like formic acid, thereby complicating process safety management.
The Novel Approach
The technology disclosed in CN102119165A overcomes these hurdles by introducing a water-soluble chiral iridium complex stabilized by a sulfate counterion. This structural modification ensures excellent solubility in aqueous media, allowing reactions to be conducted in environmentally friendly water or water-alcohol mixtures without compromising catalytic activity. The new complexes demonstrate remarkable stability in both air and water, eliminating the stringent storage conditions required for sensitive organometallic reagents. Moreover, the replacement of hazardous perchlorates with sulfates removes the risk of explosion during hydrogenation, significantly enhancing operational safety. This approach delivers higher yields and superior stereoselectivity, often exceeding 95% ee, making it a commercially viable alternative for the production of high-value chiral intermediates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Transfer Hydrogenation
The catalytic cycle of these chiral iridium aqua complexes involves a sophisticated interplay between the metal center, the pentamethylcyclopentadienyl (Cp*) ligand, and the chiral diamine ligand. In the presence of a hydrogen donor such as formic acid, the iridium center facilitates the transfer of hydride species to the substrate. The unique geometry imposed by the chiral diamine, specifically the N-substituted sulfonyl-ethylenediamine moiety, creates a highly defined chiral environment around the iridium atom. This steric and electronic environment dictates the facial selectivity of the hydride attack on the prochiral ketone or nitroalkene substrate. The aqueous nature of the solvent plays a crucial role in stabilizing the transition states and facilitating proton transfer steps, which are often rate-limiting in non-polar media. The pH of the reaction medium, typically maintained between 2 and 5, is critical for optimizing the concentration of the active hydride species while preventing catalyst decomposition.
Impurity control is inherently managed through the high specificity of the catalyst. The robust coordination of the chiral ligand prevents the formation of achiral byproducts that are common with less defined catalytic systems. Additionally, the water-soluble nature of the catalyst allows for facile separation strategies; post-reaction, the catalyst can often remain in the aqueous phase while the organic product is extracted, minimizing metal contamination in the final API intermediate. This feature is particularly valuable for pharmaceutical manufacturers who must adhere to strict limits on residual heavy metals. The stability of the complex ensures that it does not degrade into inactive iridium black or other insoluble species during the reaction course, maintaining consistent turnover numbers throughout the batch process.

How to Synthesize Chiral Iridium Aqua Complex Efficiently
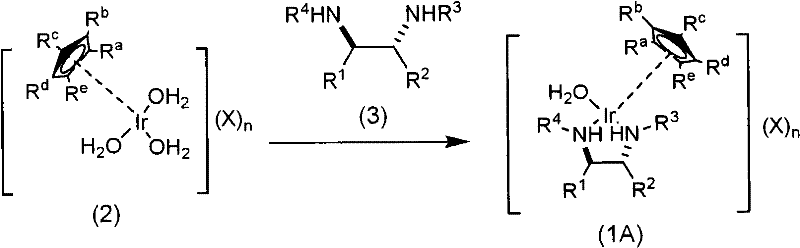
The preparation of these high-performance catalysts is designed for operational simplicity and scalability. The synthesis begins with the generation of a stable iridium precursor, which is then reacted with a commercially available or easily synthesized chiral diamine. This two-step protocol avoids the need for inert atmosphere gloveboxes for the final catalyst isolation, as the resulting complexes are air-stable. The use of water and methanol as solvents further simplifies the process infrastructure, removing the need for specialized anhydrous solvent handling systems. Detailed standardized synthesis steps for replicating this catalyst in a GMP environment are outlined in the guide below.
- Prepare the iridium precursor [Cp*Ir(H2O)3](SO4) by reacting [Cp*IrCl2]2 with Ag2SO4 in water, followed by filtration and evaporation.
- Mix the iridium precursor with the specific chiral diamine ligand (e.g., N-substituted sulfonyl-ethylenediamine) in a water-methanol solvent system.
- Stir the mixture at room temperature for 1 to 24 hours, then concentrate under reduced pressure to isolate the red powder catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous iridium catalytic technology presents a compelling value proposition centered on risk mitigation and process efficiency. The shift from organic solvents to aqueous systems drastically reduces the volume of hazardous waste generated, leading to substantial cost savings in waste disposal and regulatory compliance. The enhanced stability of the catalyst means longer shelf life and reduced spoilage losses during storage and transportation, ensuring a more reliable supply of critical reagents. Furthermore, the elimination of explosive perchlorate salts simplifies the safety protocols required for manufacturing facilities, potentially lowering insurance premiums and reducing the need for specialized explosion-proof equipment.
- Cost Reduction in Manufacturing: The ability to run reactions in water or water-alcohol mixtures eliminates the need for expensive, anhydrous organic solvents and the energy-intensive drying processes associated with them. The high catalytic activity allows for lower catalyst loadings, often in the range of 0.001 to 0.1 mol%, which significantly reduces the consumption of precious iridium metal per kilogram of product. Additionally, the simplified workup procedures, such as direct extraction from aqueous media, reduce labor hours and processing time, contributing to a leaner manufacturing cost structure without compromising product quality.
- Enhanced Supply Chain Reliability: The air and water stability of these complexes means they are less susceptible to degradation during shipping and storage compared to moisture-sensitive traditional catalysts. This robustness ensures that the quality of the catalyst remains consistent from the point of manufacture to the point of use, reducing the risk of batch failures due to reagent degradation. The use of common, non-hazardous solvents like water and methanol also mitigates supply chain risks associated with the volatility of organic solvent markets, ensuring continuous production capability even during raw material shortages.
- Scalability and Environmental Compliance: Scaling up aqueous reactions is generally safer and more straightforward than scaling exothermic reactions in organic solvents, as water has a higher heat capacity and better thermal conductivity. This facilitates the transition from laboratory scale to commercial production (100 kgs to 100 MT) with minimal process re-engineering. The alignment with green chemistry principles enhances the environmental profile of the final pharmaceutical product, which is increasingly important for meeting corporate sustainability goals and satisfying regulatory requirements regarding solvent residues and heavy metal impurities in drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral iridium aqua complex technology. These insights are derived directly from the experimental data and embodiments provided in the patent literature, offering a clear understanding of the catalyst's capabilities and limitations in real-world applications. Understanding these nuances is essential for R&D teams evaluating this technology for process integration.
Q: Why are aqueous iridium complexes preferred over traditional ruthenium catalysts?
A: Aqueous iridium complexes offer superior reaction rates compared to ruthenium analogs and eliminate the need for hazardous organic solvents, aligning with green chemistry principles while maintaining high stereoselectivity.
Q: What safety advantages do sulfate-based iridium catalysts provide?
A: Unlike perchlorate-based complexes which pose explosion risks, especially in the presence of formic acid, sulfate-based iridium aqua complexes are stable and safe to handle during asymmetric transfer hydrogenation processes.
Q: Can this catalyst system be used for nitroalkene reduction?
A: Yes, the patented chiral iridium aqua complexes are highly effective for the asymmetric transfer hydrogenation of nitroalkene compounds to produce optically active nitroalkanes with high enantiomeric excess.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the chiral iridium aqua complexes described in CN102119165A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements and residual metal analysis, guaranteeing that every batch meets the highest industry standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore how this aqueous catalytic system can optimize your specific synthesis routes. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis tailored to your production volumes and current process constraints. We encourage you to contact our technical procurement team today to request specific COA data for our catalyst offerings and comprehensive route feasibility assessments, ensuring your supply chain is built on a foundation of innovation, safety, and reliability.
