Revolutionizing Asymmetric Synthesis with Stable Chiral Iridium Aqua Complexes for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient and environmentally benign methods for constructing chiral centers, a critical requirement for the synthesis of active pharmaceutical ingredients. Patent CN102119165A presents a groundbreaking advancement in this field by disclosing a novel class of chiral iridium aqua complexes that facilitate asymmetric transfer hydrogenation with exceptional performance. Unlike conventional catalysts that often require hazardous organic solvents or unstable counter-ions, these new complexes are designed to operate effectively in aqueous environments, aligning perfectly with the principles of green chemistry. The technology addresses long-standing challenges regarding catalyst stability, solubility, and safety, offering a robust platform for the production of optically active hydroxy compounds and nitroalkane derivatives. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes while maintaining rigorous purity standards required for regulatory compliance in global markets.
The limitations of conventional methods for asymmetric transfer hydrogenation have historically constrained process efficiency and safety profiles in industrial settings. Traditional approaches often rely on ruthenium-based catalysts such as Ru(II)-TsDPEN, which, while effective, suffer from slow reaction kinetics and poor solubility in environmentally friendly aqueous solvents. Furthermore, earlier iridium-based systems frequently utilized perchlorate anions to stabilize the metal center, introducing severe safety hazards due to the potential explosiveness of perchlorates, especially when combined with hydrogen donors like formic acid. These legacy methods also often necessitate the use of volatile organic compounds (VOCs) as solvents, which complicates waste management and increases the overall environmental footprint of the manufacturing process. The inability to perform these reactions safely and efficiently in water has been a major bottleneck for scaling up the production of high-value chiral intermediates without incurring prohibitive costs related to solvent recovery and safety protocols.
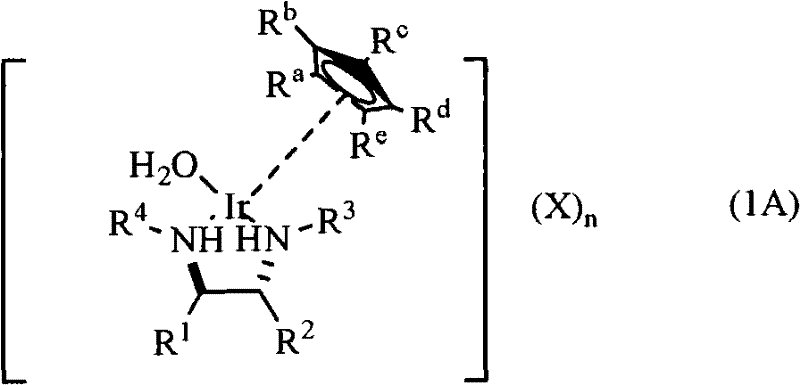
The novel approach detailed in the patent overcomes these barriers by introducing a chiral iridium aqua complex that is inherently stable in both air and water. This new catalyst system utilizes a pentamethylcyclopentadienyl (Cp*) iridium core coordinated with a chiral diamine ligand and a water molecule, stabilized by safe anions such as sulfate rather than perchlorate. This structural innovation allows the asymmetric transfer hydrogenation to proceed smoothly in aqueous solvents or water-alcohol mixtures, eliminating the need for large volumes of hazardous organic solvents. The catalyst demonstrates superior reactivity compared to traditional ruthenium complexes, achieving higher yields and significantly enhanced stereoselectivity, often exceeding 95% ee. By enabling the use of formic acid as a hydrogen source without the risk of dangerous side reactions, this technology provides a safer, more sustainable, and economically viable pathway for the synthesis of complex chiral molecules essential for modern drug development.
Mechanistic Insights into Chiral Iridium-Catalyzed Asymmetric Transfer Hydrogenation
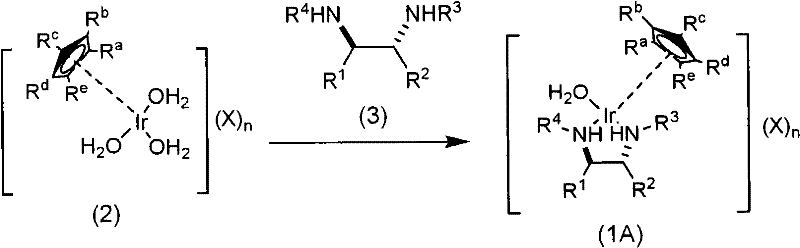
The mechanistic superiority of this chiral iridium aqua complex lies in its unique coordination geometry and the lability of the aqua ligand, which facilitates the catalytic cycle in polar media. The catalyst is typically prepared by reacting a precursor iridium complex, such as [Cp*IrCl2]2, with a specifically designed chiral diamine ligand in a water-containing solvent system. During this preparation, the chloride ligands are displaced by the chiral diamine and water molecules, forming the active cationic species stabilized by a sulfate anion. This aqueous solubility is crucial as it allows the catalyst to interact efficiently with substrates in a homogeneous phase, reducing mass transfer limitations that often plague heterogeneous or poorly soluble catalytic systems. The chiral environment created by the N-substituted sulfonyl-ethylenediamine ligand ensures that the hydride transfer from the formic acid source to the prochiral ketone or nitroalkene occurs with high facial selectivity, dictating the stereochemical outcome of the reaction.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this catalytic system offers distinct advantages in minimizing byproduct formation. The high stereoselectivity inherent to the chiral iridium aqua complex means that the formation of the unwanted enantiomer is drastically suppressed, simplifying downstream purification processes such as crystallization or chromatography. Additionally, the stability of the catalyst in water prevents decomposition pathways that might lead to metal contamination or the generation of toxic organic byproducts often associated with solvent degradation. The reaction conditions, typically maintained at a pH between 2.0 and 5.0 and temperatures ranging from 30°C to 100°C, are mild enough to preserve sensitive functional groups on the substrate while being robust enough to drive the reaction to completion. This precise control over reaction parameters ensures a clean impurity profile, which is critical for meeting the stringent quality specifications demanded by regulatory agencies for drug substances.
How to Synthesize Optically Active Hydroxy Compounds Efficiently
The synthesis of optically active hydroxy compounds using this patented technology involves a straightforward protocol that leverages the water-soluble nature of the catalyst for ease of handling and processing. The process begins with the preparation of the active chiral iridium species, followed by the addition of the carbonyl substrate and a hydrogen donor, typically formic acid or a formate salt, directly into the aqueous reaction mixture. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, pH adjustments, and temperature controls necessary to maximize yield and enantiomeric excess. This methodology is particularly advantageous for process chemists looking to transition from laboratory scale to pilot plant operations, as the use of water as a primary solvent reduces the complexity of reactor requirements and safety systems.
- Prepare the chiral iridium aqua complex by reacting the iridium precursor with a chiral diamine ligand in a water-alcohol mixed solvent.
- Dissolve the carbonyl substrate in the aqueous catalyst solution and add formic acid or its salt as the hydrogen donor source.
- Maintain the reaction pH between 2.0 and 5.0 and stir at temperatures ranging from 30°C to 100°C until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this chiral iridium aqua complex technology translates into tangible strategic benefits regarding cost structure and operational reliability. The ability to conduct reactions in aqueous media significantly reduces the dependency on expensive and regulated organic solvents, leading to substantial cost savings in raw material procurement and waste disposal. Furthermore, the enhanced stability of the catalyst in air and water extends its shelf life and simplifies storage and transportation logistics, reducing the risk of supply disruptions caused by degraded materials. The elimination of hazardous perchlorate salts also lowers insurance premiums and safety compliance costs associated with handling explosive materials, contributing to a more resilient and cost-effective supply chain. These factors collectively enhance the overall economic viability of producing high-value chiral intermediates, making the technology attractive for long-term commercial partnerships.
- Cost Reduction in Manufacturing: The shift to aqueous solvents eliminates the need for costly organic solvent recovery systems and reduces the volume of hazardous waste generated during production. By removing the requirement for expensive transition metal scavengers often needed to meet residual metal limits, the downstream processing costs are significantly optimized. The high catalytic efficiency allows for lower catalyst loading while maintaining high turnover numbers, further driving down the cost per kilogram of the final product. These qualitative improvements in process efficiency directly impact the bottom line, offering a competitive pricing advantage in the global market for fine chemical intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: The robust stability of the chiral iridium aqua complex ensures consistent performance across different batches, minimizing the risk of production failures due to catalyst degradation. Since the catalyst can be stored and handled under ambient conditions without special inert atmosphere requirements, the logistics of sourcing and inventory management are greatly simplified. This reliability extends to the raw material supply, as the ligands and iridium precursors are commercially available or easily synthesized, reducing the risk of bottlenecks associated with exotic or hard-to-source reagents. Consequently, manufacturers can maintain tighter delivery schedules and respond more agilely to fluctuating market demands for critical pharmaceutical building blocks.
- Scalability and Environmental Compliance: Scaling up reactions in water is inherently safer and more straightforward than in organic solvents, as issues related to heat dissipation and flammability are mitigated. The process aligns with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste discharge, facilitating smoother regulatory approvals for new manufacturing sites. The simplified workup procedure, often requiring only extraction or crystallization from the aqueous phase, reduces the number of unit operations needed, thereby increasing throughput capacity. This scalability ensures that the technology can support commercial production volumes ranging from hundreds of kilograms to multi-ton annual capacities, securing a stable supply for large-scale drug manufacturing campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral iridium catalyst technology in industrial processes. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this method into existing production workflows.
Q: What are the safety advantages of this iridium catalyst compared to traditional perchlorate complexes?
A: Unlike traditional complexes that utilize potentially explosive perchlorate anions, this novel chiral iridium aqua complex employs sulfate or other stable anions, allowing for safe operation even in the presence of formic acid without the risk of hazardous gas generation or explosion.
Q: Can this catalyst be used for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the catalyst exhibits excellent stability in both air and water, and the use of aqueous solvents significantly simplifies post-reaction workup and waste treatment, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What level of stereoselectivity can be expected using this technology?
A: The patent data indicates that reactions using these chiral iridium aqua complexes consistently achieve high stereoselectivity, often exceeding 80% ee and frequently reaching above 95% ee, which is critical for producing high-purity optically active compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Iridium Aqua Complex Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced catalytic technologies like the chiral iridium aqua complex to deliver high-quality pharmaceutical intermediates to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards of enantiomeric excess and chemical purity required by the industry. Our commitment to green chemistry and process safety aligns perfectly with the advantages offered by this aqueous catalytic system, making us an ideal partner for your next project.
We invite you to engage with our technical procurement team to discuss how this innovative technology can be tailored to your specific synthesis needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this aqueous process. We encourage you to contact us to obtain specific COA data and route feasibility assessments for your target molecules. Let us collaborate to optimize your supply chain and accelerate the development of your next-generation therapeutics with reliable, high-performance chiral intermediates.
