Advanced Nickel-Catalyzed Hydrocyanation for High-Purity Pharmaceutical Intermediates
The chemical industry is constantly evolving towards greener, safer, and more efficient synthetic methodologies, particularly in the realm of fine chemical and pharmaceutical intermediate production. A significant breakthrough in this domain is documented in Chinese Patent CN110845364B, which discloses a novel preparation method for nitrile compounds utilizing formamide as a cyanogen source. This technology represents a paradigm shift from traditional hydrocyanation reactions that rely on hazardous hydrogen cyanide. By employing a nickel-catalyzed system where formamide undergoes spontaneous dehydration to generate cyano anions in situ, this method offers a robust pathway for converting various olefins into valuable nitrile derivatives. The reaction operates under relatively mild thermal conditions ranging from 60-160°C and demonstrates exceptional tolerance to air and moisture, addressing critical pain points in industrial scalability and operator safety. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this patent provides a compelling framework for cost reduction in API manufacturing while maintaining high purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrile compounds via hydrocyanation has been plagued by significant safety and operational challenges. Conventional literature methods predominantly utilize highly toxic hydrogen cyanide (HCN) gas, cyanates, or their analogs as the direct source of the cyano group. Handling these virulent reagents necessitates expensive, specialized containment infrastructure and rigorous safety protocols to prevent lethal exposure. Furthermore, the catalysts traditionally employed, such as zero-valent cobalt (Co(0)) or nickel (Ni(0)) complexes, are notoriously sensitive to oxygen and moisture. This sensitivity mandates the use of strictly inert atmospheres, often requiring gloveboxes or Schlenk lines, which drastically increases the complexity and cost of the operation. The instability of these low-valent metal catalysts also leads to inconsistent reaction performance and potential deactivation, resulting in lower yields and difficult purification processes that hinder commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology outlined in patent CN110845364B fundamentally reengineers the hydrocyanation process by replacing dangerous gaseous reagents with stable, commodity-grade formamide. In this innovative system, formamide acts dually as the reactant and the solvent, undergoing Lewis acid-catalyzed dehydration to release the active cyano species directly within the reaction mixture. This eliminates the need for external dehydrating agents like phosphorus pentoxide or phosphorus oxychloride, streamlining the workflow. The use of stable Ni(II) precursors, such as nickel acetylacetonate, in conjunction with zinc powder additives, creates a catalytic cycle that is remarkably insensitive to air and light. This robustness allows the reaction to proceed in standard laboratory glassware without elaborate inert gas protection, significantly lowering the barrier to entry for implementation. The general reaction scheme illustrates this elegant transformation where diverse olefins are efficiently converted into nitriles.
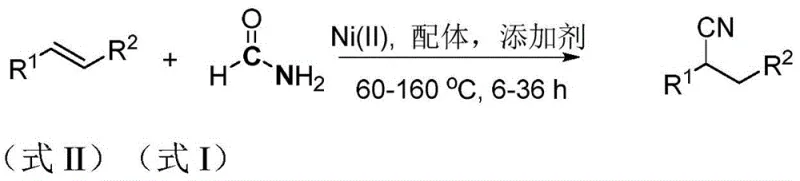
Mechanistic Insights into Nickel-Catalyzed Hydrocyanation
The mechanistic pathway of this transformation involves a sophisticated interplay between the nickel catalyst, the zinc additive, and the formamide solvent. Initially, the Ni(II) precursor is reduced in situ by the zinc powder to generate the active low-valent nickel species capable of oxidative addition. Simultaneously, the formamide molecules undergo dehydration, potentially facilitated by Lewis acidic species generated during the catalytic cycle, to produce hydrocyanic acid or cyano anions transiently. The active nickel center then undergoes oxidative addition with the generated HCN species, forming a nickel-hydride-cyano intermediate. Subsequently, the olefin substrate coordinates to the metal center and undergoes migratory insertion into the nickel-hydride bond. This step is critical for determining the regioselectivity of the final product. Finally, reductive elimination releases the desired nitrile compound and regenerates the active nickel catalyst, closing the catalytic loop. This mechanism ensures high atom economy and minimizes the formation of side products associated with free radical pathways.
Impurity control is a paramount concern for any process intended for pharmaceutical applications, and this method offers distinct advantages in this regard. The mild reaction conditions and the specific coordination environment provided by ligands like Xantphos help suppress oligomerization or polymerization of the olefin substrates, which are common side reactions in harsh acidic or basic conditions. Furthermore, because the cyanide source is generated in situ in controlled amounts rather than added in bulk, the risk of over-cyanation or formation of polynitrile byproducts is significantly mitigated. The use of formamide also simplifies the workup procedure; since it acts as the solvent, removal is straightforward, and the resulting crude products often require minimal purification to achieve high-purity nitrile compounds suitable for downstream synthesis. This level of control over the impurity profile is essential for meeting the stringent quality specifications required by global regulatory bodies.
How to Synthesize 2-Phenylpropionitrile Efficiently
The synthesis of 2-phenylpropionitrile serves as a representative example of the broader utility of this technology for producing valuable aromatic nitriles. The process begins by charging a reaction vessel with styrene, a nickel catalyst such as Ni(acac)2, a bidentate phosphine ligand like Xantphos, and zinc powder as a reductant. Formamide is then added in a significant molar excess to serve as both the cyanide donor and the reaction medium. The mixture is sealed and heated to approximately 150°C for a duration of 24 hours. Under these conditions, the styrene is quantitatively consumed, and the desired nitrile is formed with high selectivity. The detailed standardized synthesis steps for this and other derivatives are provided in the guide below.
- Prepare the reaction mixture by combining the olefin substrate, nickel catalyst (e.g., Ni(acac)2), ligand (e.g., Xantphos), and zinc powder additive in a reaction vessel.
- Add formamide which serves as both the cyanide source and the solvent, ensuring a molar excess relative to the olefin substrate.
- Heat the sealed reaction mixture to temperatures between 60-160°C for 6-36 hours, then purify the resulting nitrile product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this formamide-based hydrocyanation technology offers transformative benefits regarding cost structure and logistical reliability. The most immediate impact is the drastic simplification of safety infrastructure. By eliminating the need to store, transport, and handle bulk hydrogen cyanide gas, facilities can avoid the capital expenditure associated with specialized gas detection systems, scrubbers, and containment units. This reduction in overhead directly translates to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the catalyst system utilizes inexpensive, air-stable Ni(II) salts and commodity zinc powder, avoiding the premium pricing and short shelf-life associated with sensitive organometallic Ni(0) or Co(0) complexes. The ability to run reactions without rigorous exclusion of air and moisture further reduces operational costs by minimizing the consumption of inert gases and allowing for the use of standard stainless steel reactors rather than Hastelloy-lined vessels designed for corrosive cyanide service.
- Cost Reduction in Manufacturing: The economic advantages of this process are multifaceted, stemming primarily from the substitution of hazardous reagents with benign alternatives. Formamide is a widely available, low-cost commodity chemical, whereas anhydrous HCN requires specialized production and logistics. The elimination of expensive dehydrating agents and the use of robust, non-precious metal catalysts further drive down the raw material costs. Moreover, the simplified workup and purification procedures reduce solvent consumption and waste disposal fees. These factors collectively contribute to a leaner manufacturing process that enhances profit margins without compromising product quality, making it an attractive option for large-scale production.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials. Olefins and formamide are produced globally in massive quantities, ensuring a stable supply base that is less susceptible to geopolitical disruptions or vendor shortages compared to specialized cyanating agents. The insensitivity of the reaction to environmental factors like humidity means that production schedules are less likely to be delayed by equipment failures related to inert atmosphere breaches. This reliability ensures consistent lead times for high-purity pharmaceutical intermediates, allowing downstream customers to maintain just-in-time inventory strategies with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental hurdles, but this technology is inherently designed for scalability. The absence of toxic gas evolution simplifies reactor design and venting systems, facilitating a smoother transition from pilot plant to commercial production volumes. From an environmental perspective, the process aligns with green chemistry principles by using a non-toxic cyanide source and generating fewer hazardous byproducts. This compliance with stringent environmental regulations reduces the risk of fines and shutdowns, ensuring long-term operational continuity. The ease of waste treatment, given the biodegradable nature of formamide residues compared to heavy metal cyanide complexes, further enhances the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed hydrocyanation technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is critical for R&D teams evaluating process feasibility and for procurement officers assessing supplier capabilities. The insights provided here clarify the operational boundaries and potential advantages of adopting this method for your specific synthesis needs.
Q: What are the safety advantages of using formamide over traditional cyanide sources?
A: Traditional hydrocyanation often requires handling highly toxic hydrogen cyanide (HCN) gas or volatile cyanates. This patented method utilizes formamide, a stable, non-toxic liquid that generates the cyano group in situ through dehydration, significantly reducing operational hazards and eliminating the need for specialized gas handling infrastructure.
Q: Is this nickel-catalyzed process sensitive to air and moisture?
A: Unlike conventional methods utilizing sensitive Ni(0) or Co(0) complexes that require rigorous inert atmosphere techniques, this protocol employs stable Ni(II) precursors and zinc additives. The process demonstrates robust tolerance to air and moisture, simplifying the operational requirements for large-scale manufacturing.
Q: What types of olefin substrates are compatible with this synthesis method?
A: The method exhibits broad substrate universality, successfully converting various mono-substituted and di-substituted aliphatic and aromatic olefins. It accommodates diverse functional groups including alkyl, alkoxy, fluoro, and chloro substituents on aromatic rings, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Phenylpropionitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our technical team has extensively evaluated the potential of the formamide-based hydrocyanation route described in CN110845364B and possesses the expertise to implement this technology effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of nitrile intermediate meets the highest standards required for API synthesis. We are committed to delivering high-purity pharmaceutical intermediates that empower your drug development pipelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. By partnering with us, you gain access to not just a product, but a comprehensive solution that optimizes your supply chain and reduces overall manufacturing costs. Please contact us today to request specific COA data and route feasibility assessments for your target nitrile compounds, and let us demonstrate how our technical prowess can drive value for your organization.
