Advanced Selectfluor-Mediated Synthesis of N-Phenyl-3-Morpholine Propionamide for Commercial Scale-Up
Advanced Selectfluor-Mediated Synthesis of N-Phenyl-3-Morpholine Propionamide for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the urgent need for safer, more efficient, and environmentally sustainable synthetic routes. A pivotal advancement in this domain is documented in patent CN109232476B, which discloses a novel method for preparing N-phenyl-3-morpholine propionamide, a critical intermediate in the synthesis of bioactive molecules and pharmaceuticals. This technology represents a significant departure from legacy processes that rely on hazardous reagents, offering a streamlined pathway that aligns with modern green chemistry principles. By leveraging the unique electrophilic activation capabilities of Selectfluor (1-chloromethyl-4-fluoro-1,4-diazabicyclo[2.2.2]octane bis(tetrafluoroborate)), this method transforms a traditionally sluggish or dangerous transformation into a robust, high-yielding operation. For R&D directors and procurement strategists alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the baggage of toxic waste streams associated with older technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-phenyl-3-morpholine propionamide has been plagued by significant operational and safety challenges that hinder efficient cost reduction in fine chemical manufacturing. The first conventional approach involves the Michael addition of morpholine to N-phenylacrylamide under basic conditions. While conceptually simple, this route suffers from prohibitively long reaction times, often requiring up to four days to reach completion, which severely limits throughput and increases energy consumption. A second, more common industrial route utilizes 3-chloropropionyl chloride as a key building block. This method is fundamentally flawed due to the extreme toxicity, corrosiveness, and volatility of the acid chloride reagent. Furthermore, this two-step process is operationally tedious and environmentally unfriendly, generating substantial hazardous waste. Most critically from a commercial perspective, the yield of this traditional acid chloride route is abysmally low, reported at merely 13%, rendering it economically unviable for large-scale production where raw material efficiency is paramount.
The Novel Approach
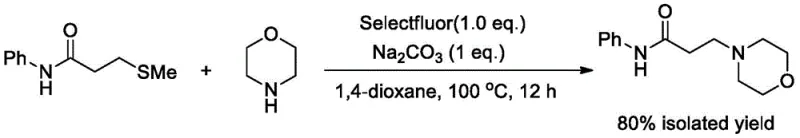
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes N-phenyl-3-methylthiopropionamide as a stable, non-volatile precursor. This strategy completely eliminates the need for handling dangerous acid chlorides, thereby simplifying the engineering controls required for production. The reaction proceeds via a single-pot protocol where the thioether moiety is activated in situ by Selectfluor, facilitating a rapid nucleophilic substitution by morpholine. This innovation drastically reduces the reaction time from days to just 12 hours, significantly enhancing facility utilization rates. Moreover, the process demonstrates exceptional efficiency, achieving an isolated yield of 80% at optimized conditions. This leap in performance not only improves the economic feasibility of producing this valuable high-purity pharmaceutical intermediate but also aligns with stringent environmental regulations by avoiding the generation of corrosive hydrochloric acid byproducts typically associated with acid chloride chemistry.
Mechanistic Insights into Selectfluor-Mediated Thioether Activation
The core of this technological breakthrough lies in the electrophilic activation of the sulfur atom within the thioether group. Selectfluor acts as a potent electrophilic fluorinating agent that interacts with the lone pair electrons on the sulfur atom of N-phenyl-3-methylthiopropionamide. This interaction generates a highly reactive sulfonium intermediate, effectively turning the sulfur into a superior leaving group compared to the native methylthio group. Once activated, the electron-rich nitrogen of the morpholine molecule performs a nucleophilic attack on the beta-carbon relative to the carbonyl group, displacing the activated sulfur species. This mechanism bypasses the high energy barriers associated with direct nucleophilic substitution on unactivated sulfides. The presence of sodium carbonate serves as a mild base to neutralize any acidic byproducts generated during the fluorination event, maintaining a pH environment that preserves the integrity of the amide bond while promoting the substitution reaction. This precise control over the reaction pathway is what allows for the high selectivity observed in the process.
From an impurity control perspective, this mechanism offers distinct advantages over acid chloride routes. In traditional methods involving 3-chloropropionyl chloride, the high reactivity of the acid chloride often leads to over-acylation or hydrolysis if moisture is present, creating difficult-to-remove impurities that compromise the purity profile of the final API intermediate. In the Selectfluor-mediated pathway, the reaction conditions are significantly milder, operating at 100 °C in 1,4-dioxane. The specificity of the electrophilic activation ensures that the morpholine attacks primarily at the intended site, minimizing side reactions such as polymerization or degradation of the amide backbone. Furthermore, the byproducts formed from the sulfur moiety are generally more manageable and less corrosive than the hydrogen chloride gas evolved in acid chloride reactions. This cleaner reaction profile translates directly to simplified downstream processing, reducing the burden on purification units and ensuring that the final product meets the stringent purity specifications required for downstream pharmaceutical applications.
How to Synthesize N-Phenyl-3-Morpholine Propionamide Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits of the Selectfluor activation. The patent outlines a straightforward procedure where N-phenyl-3-methylthiopropionamide, morpholine, Selectfluor, and sodium carbonate are combined in a specific molar ratio of 1:2:1:1. The choice of solvent, 1,4-dioxane, is critical for solubilizing both the organic substrates and the ionic Selectfluor reagent, ensuring a homogeneous reaction mixture. The process is designed to be robust, tolerating a temperature range of 80 to 120 °C, though 100 °C is identified as the optimal setpoint for balancing reaction rate and yield. Detailed standardized synthesis steps, including precise addition rates and workup procedures, are provided in the guide below to assist process chemists in replicating this high-efficiency protocol.
- Charge N-phenyl-3-methylthiopropionamide, morpholine, Selectfluor, and sodium carbonate into a reactor with 1,4-dioxane.
- Heat the reaction mixture to 100 °C and stir vigorously for 12 hours to ensure complete conversion.
- Concentrate the reaction liquid and perform column chromatography to isolate the pure N-phenyl-3-morpholine propionamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers transformative benefits that extend far beyond simple yield improvements. The shift away from 3-chloropropionyl chloride eliminates a major supply chain bottleneck, as acid chlorides often require specialized storage and transport due to their hazardous nature. By utilizing stable solid reagents like Selectfluor and liquid amines, the logistics of raw material handling become significantly simpler and safer. This change reduces the regulatory burden and insurance costs associated with storing highly corrosive chemicals on-site. Furthermore, the dramatic increase in yield from 13% to 80% implies a massive reduction in the effective cost of goods sold (COGS), as significantly less raw material is wasted per kilogram of finished product. This efficiency gain provides a substantial buffer against raw material price volatility, ensuring more stable pricing for long-term contracts.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the elimination of expensive and hazardous reagents and the drastic improvement in atom economy. By avoiding the use of 3-chloropropionyl chloride, manufacturers save on the costs associated with corrosion-resistant reactors and scrubbing systems required to handle HCl off-gassing. The higher yield means that for every ton of input material, the output of saleable product is multiplied several times over compared to the old method. Additionally, the shorter reaction time of 12 hours versus 4 days allows for faster turnover of reactor vessels, increasing the overall capacity of the manufacturing plant without the need for capital expenditure on new equipment. These factors combine to deliver significant cost savings that can be passed down the supply chain.
- Enhanced Supply Chain Reliability: Reliability in the supply of pharmaceutical intermediates is often compromised by the availability of niche, hazardous reagents. The new method relies on commoditized chemicals like morpholine and sodium carbonate, alongside Selectfluor, which is widely produced for various fluorination applications. This diversification of the raw material base reduces the risk of supply disruptions caused by regulatory shutdowns of acid chloride manufacturers. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in utility supplies or environmental conditions, ensuring consistent delivery schedules. For supply chain planners, this predictability is invaluable for maintaining lean inventory levels while preventing stockouts of critical downstream APIs.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden safety and environmental liabilities, but this route is inherently designed for scale. The absence of highly exothermic acid chloride additions reduces the risk of thermal runaways, a common concern in batch processing. From an environmental standpoint, the process generates less hazardous waste, simplifying effluent treatment and reducing disposal costs. The use of 1,4-dioxane, while requiring careful recovery, is a standard solvent with well-established recycling protocols. This alignment with green chemistry principles facilitates easier permitting for new production lines and helps multinational clients meet their own sustainability goals, making the supplier a more attractive partner for long-term strategic alliances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in patent CN109232476B. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages of the Selectfluor-mediated pathway over traditional methods, focusing on yield, safety, and operational simplicity.
Q: What is the primary advantage of this Selectfluor-mediated route over traditional acid chloride methods?
A: The primary advantage is safety and efficiency. Traditional methods use toxic, corrosive 3-chloropropionyl chloride with low yields (13%), whereas this method uses stable sulfides and achieves 80% yield under mild conditions.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes common solvents like 1,4-dioxane and operates at standard heating temperatures (100 °C), making it highly suitable for commercial scale-up without requiring specialized corrosion-resistant equipment.
Q: How does the reaction temperature affect the yield of N-phenyl-3-morpholine propionamide?
A: Temperature significantly impacts efficiency. At 100 °C, the isolated yield reaches 80%, while lowering the temperature to 80 °C reduces the yield to 65%, indicating that higher thermal energy facilitates the activation of the thioether bond.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenyl-3-Morpholine Propionamide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patent can be fully realized at an industrial level. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying the identity and purity of every batch, adhering to the most stringent purity specifications demanded by the global pharmaceutical industry. We understand that consistency is key, and our process engineering teams are dedicated to optimizing parameters such as temperature and stirring rates to maintain the high 80% yield benchmark established in the patent literature.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this greener, more efficient route for your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments tailored to your project timelines. Let us help you secure a sustainable, cost-effective, and reliable supply of this critical intermediate.
