Revolutionizing Indolizinopyrrolone Production: A Scalable Copper-Catalyzed Route for Global Pharmaceutical Supply Chains
Revolutionizing Indolizinopyrrolone Production: A Scalable Copper-Catalyzed Route for Global Pharmaceutical Supply Chains
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for more efficient and sustainable synthetic pathways in the pharmaceutical industry. Patent CN113788831B introduces a groundbreaking methodology for the synthesis of dihydroindolizinopyrrolone derivatives and their analogues, addressing critical bottlenecks in current manufacturing processes. This innovation leverages a copper-catalyzed cyclization strategy that merges an α-diethyl malonate substituted O-acyl ketoxime with various pyridine derivatives to construct complex polycyclic skeletons in a single operational step. For R&D directors and procurement managers alike, this represents a paradigm shift from laborious multi-step sequences to a streamlined, high-yielding process that promises to redefine cost structures and supply chain reliability for high-value pharmaceutical intermediates.
The significance of this technology extends beyond mere academic interest; it offers a tangible solution for the commercial scale-up of complex heterocyclic compounds that serve as key scaffolds in anticancer drug discovery. By utilizing inexpensive copper salts instead of rare earth metals and operating under relatively mild thermal conditions, the process drastically reduces the environmental footprint and operational costs associated with traditional synthesis. As we delve deeper into the technical specifics, it becomes evident that this patent provides a robust framework for producing high-purity indolizinopyrrolone derivatives with excellent functional group tolerance, thereby opening new avenues for medicinal chemists to explore structure-activity relationships without being constrained by synthetic feasibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indolizine and pyrrolopyrrolone skeletons has been fraught with significant challenges that hinder efficient large-scale production. Traditional approaches often rely on precious or hazardous metal catalysts, such as samarium trifluoromethanesulfonate, which necessitate harsh reaction conditions like heating to 120°C under solvent-free environments. These extreme parameters not only escalate energy consumption but also pose substantial safety risks in a manufacturing setting, complicating the engineering controls required for safe operation. Furthermore, alternative routes developed by research groups such as Macdonald and Borthwick involve convoluted multi-step sequences starting from amino acids like methionine or diaminobutyric acid.
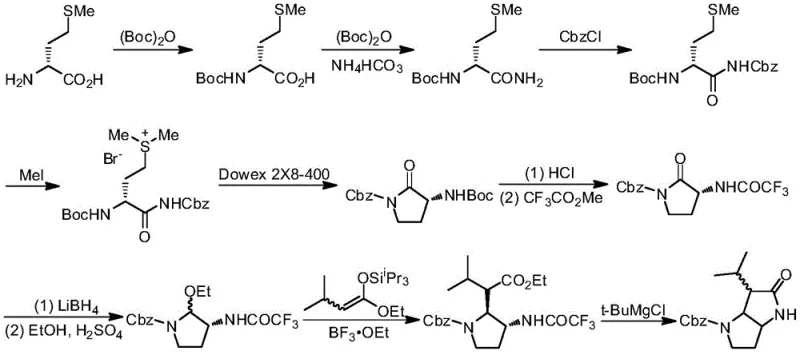
These legacy methods typically suffer from abysmal overall yields, often hovering around 8% to 10% after eight or nine distinct chemical transformations. Such inefficiencies result in massive material waste, inflated production costs, and extended lead times, making them economically unviable for the cost-sensitive pharmaceutical market. The reliance on protecting group strategies, such as Boc protection and subsequent deprotection, further adds to the step count and purification burden, creating a bottleneck that limits the availability of these valuable scaffolds for drug development programs. Consequently, there has been a persistent demand for a more direct and atom-economical approach that can bypass these inherent limitations.
The Novel Approach
In stark contrast to the cumbersome legacy techniques, the methodology disclosed in patent CN113788831B offers a remarkably concise and efficient pathway to dihydroindolizinopyrrolone derivatives. This novel approach capitalizes on the unique reactivity of α-diethyl malonate substituted O-acyl ketoximes, which, under the influence of a copper salt catalyst, generate a cyclic intermediate possessing dual electrophilic centers. This reactive species then undergoes a double nucleophilic addition with pyridine derivatives, effectively stitching together the complex fused ring system in a single pot. The reaction proceeds smoothly in common organic solvents like toluene at moderate temperatures ranging from 60°C to 100°C, eliminating the need for extreme thermal inputs or specialized solvent-free setups.
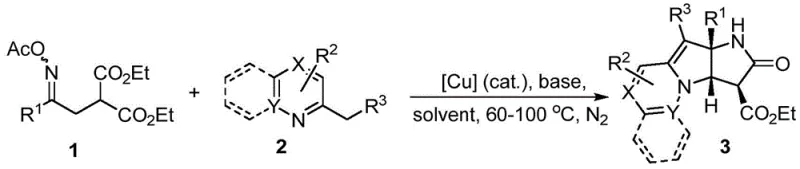
The versatility of this method is underscored by its broad substrate scope, accommodating various substituents on both the oxime and the pyridine components, including electron-withdrawing and electron-donating groups. Experimental data from the patent demonstrates that this one-step cyclization can achieve moderate to excellent yields, with specific examples reporting yields as high as 92% for certain substrates. This dramatic improvement in efficiency translates directly into reduced raw material consumption and simplified downstream processing, positioning this technology as a superior choice for reliable pharmaceutical intermediate supplier operations seeking to optimize their manufacturing portfolios.
Mechanistic Insights into Copper-Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to replicate and optimize the process for specific target molecules. The reaction initiates with the activation of the O-acyl ketoxime by the copper catalyst, which facilitates the formation of a key cyclic intermediate. This intermediate is characterized by the presence of two electrophilic sites that are strategically positioned to engage in nucleophilic attack. When introduced to a pyridine derivative, the nitrogen atom of the pyridine ring acts as a nucleophile, attacking one of the electrophilic centers to form a new carbon-nitrogen bond. Subsequently, an intramolecular cyclization occurs, closing the second ring and establishing the characteristic indolizinopyrrolone core structure.
This mechanism highlights the dual role of the copper catalyst in both activating the oxime precursor and stabilizing the transition states involved in the bond-forming events. The use of a base, such as lithium carbonate or potassium carbonate, is essential to neutralize the acidic byproducts generated during the reaction, thereby driving the equilibrium towards the desired product. Importantly, the mild conditions employed prevent the decomposition of sensitive functional groups, ensuring high purity of the final product. This level of control over the reaction pathway minimizes the formation of side products and impurities, which is a critical factor for meeting the stringent quality standards required in API manufacturing. The robustness of this catalytic cycle suggests that it can be adapted for continuous flow chemistry, further enhancing its potential for industrial application.
How to Synthesize Dihydroindolizinopyrrolone Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to ensure optimal outcomes. The general procedure involves combining the O-acyl ketoxime and the pyridine derivative in a suitable solvent, followed by the addition of the copper catalyst and base under an inert atmosphere. The mixture is then heated to the specified temperature range, and the progress of the reaction is closely monitored using thin-layer chromatography (TLC) to determine the point of complete substrate conversion. Once the reaction is deemed complete, a standard aqueous workup involving extraction and drying is performed to isolate the crude product, which is subsequently purified via column chromatography to obtain the target compound in high purity.
- Mix diethyl malonate substituted O-acyl ketoxime and pyridine derivatives with a copper salt catalyst and base in an organic solvent.
- Heat the reaction mixture to 60-100°C under nitrogen protection for 1-5 hours until substrate disappearance is monitored by TLC.
- Quench with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed synthesis offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing achieved by replacing expensive and scarce rare earth metal catalysts with abundant and inexpensive copper salts. This switch not only lowers the direct material costs but also simplifies the supply chain by reducing dependency on volatile markets for specialty chemicals. Furthermore, the one-step nature of the reaction significantly shortens the production cycle time, allowing for faster turnaround from order to delivery and enhancing the overall responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences drastically reduces the consumption of reagents, solvents, and labor hours. By consolidating what was previously an eight or nine-step process into a single transformation, manufacturers can achieve significant savings in operational expenditures. The use of common solvents like toluene and easily removable inorganic bases further streamlines the waste management process, lowering the costs associated with environmental compliance and disposal. This economic efficiency makes the production of high-purity indolizinopyrrolone derivatives financially viable even at smaller scales, facilitating rapid screening and development.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable raw materials ensures a consistent and uninterrupted supply of key inputs. Unlike processes that require custom-synthesized precursors or air-sensitive reagents, this method utilizes off-the-shelf chemicals that can be sourced from multiple vendors, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less susceptible to variations in ambient conditions, leading to more predictable production schedules and reliable delivery timelines for downstream customers. This stability is crucial for maintaining long-term partnerships with pharmaceutical clients who require guaranteed availability of critical intermediates.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of hazardous reagents make this process inherently safer and easier to scale up from gram to kilogram quantities. The reduced generation of chemical waste aligns with green chemistry principles, helping companies meet increasingly stringent environmental regulations without compromising on productivity. The simplicity of the workup procedure minimizes the volume of wastewater and organic solvents requiring treatment, thereby reducing the environmental footprint of the manufacturing facility. This sustainability profile enhances the corporate image and ensures long-term operational viability in a regulatory environment that prioritizes eco-friendly practices.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial aspects of this technology, we have compiled a set of frequently asked questions based on the detailed findings within the patent documentation. These answers provide clarity on the practical implementation of the synthesis, the scope of applicable substrates, and the potential biological applications of the resulting compounds. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this method into their existing production workflows or drug discovery pipelines.
Q: What are the primary advantages of this copper-catalyzed method over traditional samarium catalysis?
A: Unlike traditional samarium-catalyzed methods requiring high temperatures (120°C) and solvent-free conditions, this copper-catalyzed route operates under milder conditions (60-100°C) in common solvents like toluene, significantly reducing energy consumption and safety risks while maintaining high yields.
Q: Is this synthesis method scalable for commercial production of API intermediates?
A: Yes, the method utilizes cheap and readily available raw materials such as copper acetate and lithium carbonate. The one-step nature of the reaction eliminates the need for complex multi-step sequences found in older methodologies, making it highly suitable for commercial scale-up.
Q: What biological activities do these dihydroindolizinopyrrolone derivatives exhibit?
A: Preliminary pharmacological tests indicate that specific derivatives, such as compound 3ad, demonstrate potent inhibitory effects against HepG2 and A549 cancer cell lines at 50 μM concentrations, suggesting strong potential for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydroindolizinopyrrolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies described in patent CN113788831B and are fully equipped to leverage this technology for our global clientele. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of dihydroindolizinopyrrolone derivatives meets the highest quality standards required for pharmaceutical applications. Our commitment to excellence extends beyond mere compliance; we strive to deliver value through continuous process optimization and technical support.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of these novel heterocyclic scaffolds in drug development. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our optimized synthesis routes can reduce your overall development costs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments for your target molecules, allowing us to demonstrate our capability as a trusted partner in your supply chain. Together, we can accelerate the discovery and delivery of next-generation therapeutics to patients worldwide.
