Advanced Chiral NNN Tridentate Ligands: Revolutionizing Asymmetric Synthesis and Commercial Manufacturing
Introduction to Next-Generation Chiral Catalysis
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the need for more efficient and sustainable manufacturing processes for high-value pharmaceutical intermediates. Patent CN113999207B introduces a breakthrough class of pyridyl-containing chiral NNN tridentate ligands that address long-standing challenges in catalyst design and accessibility. Unlike traditional ligands that often rely on cumbersome multi-step syntheses from the chiral pool, these novel structures are accessible via a direct and highly selective asymmetric catalytic hydrogenation pathway. This technological leap not only enhances the stereochemical control in downstream reactions but also fundamentally alters the economic feasibility of producing complex chiral building blocks. For R&D directors and procurement strategists, understanding the implications of this patent is crucial for securing a competitive edge in the supply of high-purity organic compounds.
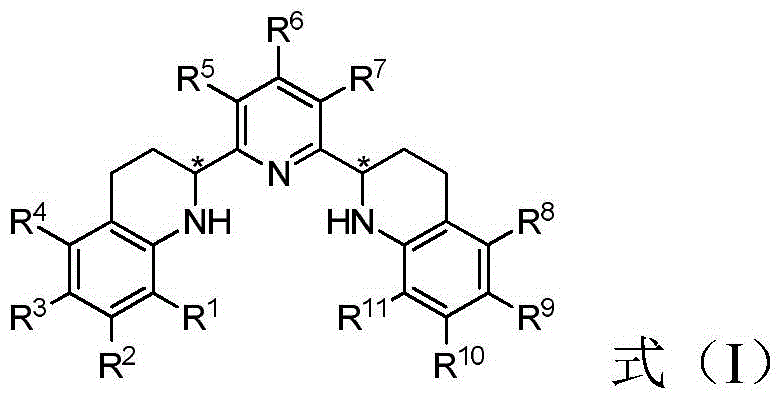
The core innovation lies in the versatile structural framework of Formula (I), which allows for extensive modulation of electronic and steric properties through various substituents (R1-R11). This tunability ensures that the ligand can be optimized for specific substrate classes, providing a level of customization that rigid, naturally derived ligands cannot match. Furthermore, the inherent stability of the pyridine backbone against air and moisture simplifies handling and storage requirements, a critical factor for industrial applications where operational simplicity translates directly to cost efficiency. By integrating these advanced ligands into existing catalytic workflows, manufacturers can achieve superior enantioselectivity while mitigating the risks associated with catalyst deactivation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral NNN tridentate ligands has been plagued by inefficiencies that hinder their widespread adoption in large-scale manufacturing. Traditional routes typically depend on the resolution of racemic mixtures or the use of expensive, naturally occurring chiral amino acids and diamines as starting materials. These methods are inherently step-intensive, often requiring protection and deprotection sequences that generate substantial chemical waste and lower overall atom economy. Moreover, the reliance on the chiral pool limits the structural diversity of the final ligand, restricting the ability to fine-tune catalytic performance for non-standard substrates. From a supply chain perspective, the volatility in the pricing and availability of these natural chiral precursors introduces significant risk and uncertainty into production planning.
The Novel Approach
The methodology disclosed in CN113999207B represents a paradigm shift by employing asymmetric catalytic hydrogenation to construct the chiral centers directly. This approach utilizes achiral or readily available precursors (Formula II) and converts them into the desired chiral ligands (Formula I) with high fidelity using a ruthenium-based catalyst system. 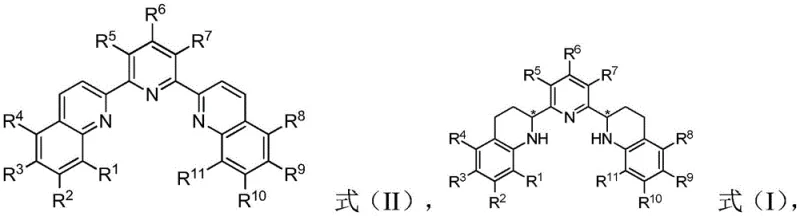 This catalytic strategy eliminates the need for stoichiometric chiral auxiliaries and reduces the synthetic sequence to a single, highly efficient transformation. The result is a drastic reduction in process mass intensity (PMI) and a significant improvement in yield. For procurement managers, this means a more robust and predictable supply chain, as the dependency on scarce natural resources is replaced by scalable chemical synthesis. The ability to produce these ligands on demand with consistent quality marks a major advancement in the commercialization of asymmetric catalysis technologies.
This catalytic strategy eliminates the need for stoichiometric chiral auxiliaries and reduces the synthetic sequence to a single, highly efficient transformation. The result is a drastic reduction in process mass intensity (PMI) and a significant improvement in yield. For procurement managers, this means a more robust and predictable supply chain, as the dependency on scarce natural resources is replaced by scalable chemical synthesis. The ability to produce these ligands on demand with consistent quality marks a major advancement in the commercialization of asymmetric catalysis technologies.
Mechanistic Insights into Ruthenium-Catalyzed Hydrogenation and Copper-Mediated Alkylation
The success of this technology hinges on the sophisticated interplay between the ligand architecture and the transition metal center. In the synthesis phase, the chiral ruthenium catalyst (Formula III) facilitates the enantioselective reduction of the imine or olefinic bonds within the precursor. The specific coordination geometry enforced by the NNN tridentate binding mode ensures that hydrogen delivery occurs from a single facial direction, locking in the desired stereochemistry with exceptional precision. 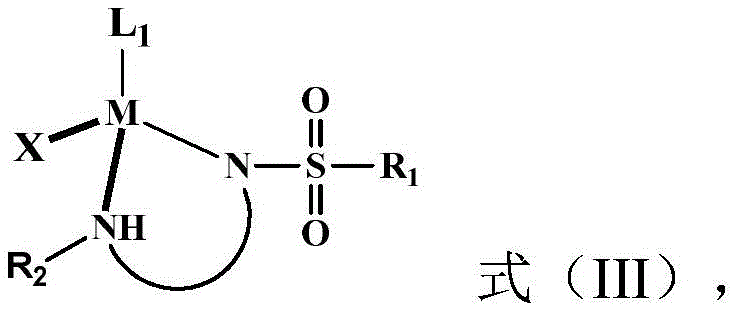 The robustness of the ruthenium-sulfonamide complex allows the reaction to proceed under relatively mild temperatures (0-50°C) and moderate hydrogen pressures, minimizing energy consumption and safety hazards. This mechanistic efficiency is further evidenced by the high turnover numbers observed, indicating that minimal catalyst loading is required to drive the reaction to completion, thereby reducing the burden of heavy metal removal in downstream processing.
The robustness of the ruthenium-sulfonamide complex allows the reaction to proceed under relatively mild temperatures (0-50°C) and moderate hydrogen pressures, minimizing energy consumption and safety hazards. This mechanistic efficiency is further evidenced by the high turnover numbers observed, indicating that minimal catalyst loading is required to drive the reaction to completion, thereby reducing the burden of heavy metal removal in downstream processing.
Beyond their own synthesis, these ligands demonstrate remarkable utility when complexed with copper for asymmetric Friedel-Crafts alkylation reactions. The resulting copper complexes activate nitroalkenes towards nucleophilic attack by indoles, creating new carbon-carbon bonds with high regio- and stereocontrol. 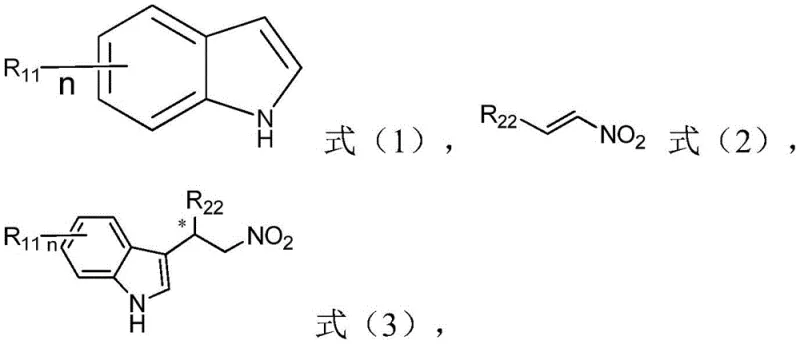 The mechanism involves the formation of a chiral Lewis acid complex that organizes the transition state, effectively shielding one face of the electrophile. This precise spatial arrangement prevents the formation of unwanted diastereomers and enantiomers, ensuring that the final product meets the stringent purity standards required for pharmaceutical applications. The ability to tune the ligand's steric bulk allows chemists to optimize this transition state for a wide variety of substituted indoles and nitroalkenes, making this a versatile platform technology for drug discovery and development.
The mechanism involves the formation of a chiral Lewis acid complex that organizes the transition state, effectively shielding one face of the electrophile. This precise spatial arrangement prevents the formation of unwanted diastereomers and enantiomers, ensuring that the final product meets the stringent purity standards required for pharmaceutical applications. The ability to tune the ligand's steric bulk allows chemists to optimize this transition state for a wide variety of substituted indoles and nitroalkenes, making this a versatile platform technology for drug discovery and development.
How to Synthesize Pyridyl-Containing Chiral NNN Ligands Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize enantiomeric excess and yield. The process begins with the preparation of the achiral precursor via condensation, followed by the critical asymmetric hydrogenation step. Optimization of solvent systems, particularly the use of fluorinated alcohols like hexafluoroisopropanol, has been shown to enhance both reaction rate and selectivity. The detailed standardized synthesis steps for replicating this high-efficiency pathway are outlined below.
- Prepare the precursor compound of Formula (II) through condensation reactions involving 2,6-diacetylpyridine derivatives and aminobenzaldehyde derivatives.
- Conduct asymmetric catalytic hydrogenation using a chiral Ruthenium catalyst (Formula III) under hydrogen pressure (10-50 atm) in solvents like hexafluoroisopropanol.
- Purify the resulting chiral NNN tridentate ligand (Formula I) via column chromatography to achieve high enantiomeric excess (>99% ee).
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the adoption of this ligand technology offers compelling strategic benefits. The shift from extraction-based or resolution-based sourcing to catalytic synthesis fundamentally de-risks the supply chain. By removing the bottleneck of limited natural chiral sources, manufacturers can secure a stable flow of critical intermediates regardless of agricultural fluctuations or geopolitical tensions affecting raw material exports. This independence fosters a more resilient supply network capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive chiral starting materials and the reduction of synthetic steps lead to substantial cost savings. Traditional methods often incur high costs due to the loss of 50% of material during resolution steps; this catalytic approach theoretically allows for 100% atom utilization regarding the carbon skeleton. Furthermore, the simplified purification protocols reduce solvent consumption and waste disposal costs, contributing to a leaner and more economically viable production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard transition metals like ruthenium and copper ensures that raw material availability is not a constraint. Unlike specialized bio-based reagents that may have long lead times, the precursors for this synthesis are widely available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source failure and ensures consistent delivery schedules, which is vital for maintaining uninterrupted production lines in the pharmaceutical sector.
- Scalability and Environmental Compliance: The process operates under mild conditions and generates significantly less hazardous waste compared to classical resolution techniques. The high selectivity minimizes the formation of byproducts, simplifying effluent treatment and helping facilities meet increasingly stringent environmental regulations. The robust nature of the catalyst system also supports seamless scale-up from laboratory benchtop to multi-ton commercial reactors without the need for extensive process re-engineering, facilitating rapid technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential adopters.
Q: What are the primary advantages of these new NNN ligands over traditional Pybox ligands?
A: The novel pyridyl-containing chiral NNN tridentate ligands described in CN113999207B offer superior air stability and easier structural tunability compared to traditional oxazoline-based ligands. Their synthesis via direct asymmetric hydrogenation bypasses the need for expensive chiral pool starting materials, significantly reducing raw material costs and simplifying the supply chain for large-scale production.
Q: Can these ligands be scaled for industrial manufacturing of API intermediates?
A: Yes, the patent demonstrates that the ligands can be synthesized using robust transition metal catalysts (Ruthenium) under relatively mild conditions (0-50°C). The process avoids sensitive reagents and complex multi-step resolutions, making it highly amenable to commercial scale-up from kilogram to metric ton quantities while maintaining strict purity specifications.
Q: What specific reactions do these ligands catalyze effectively?
A: These ligands form active copper complexes that exhibit exceptional performance in asymmetric Friedel-Crafts alkylation reactions, specifically between indole derivatives and nitroalkenes. The system achieves high conversion rates and excellent stereoselectivity (often >99% ee), which is critical for constructing chiral centers in bioactive molecules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral NNN Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in CN113999207B for the modern pharmaceutical industry. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory findings into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from concept to market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral ligand or intermediate meets the highest international standards.
We invite you to collaborate with us to leverage these advanced catalytic solutions for your specific drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current synthesis routes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your time to market.
