Advanced Synthesis of Iodo Trifluoromethoxy Compounds for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are witnessing a paradigm shift in the construction of fluorinated molecular architectures, driven by the unique physicochemical properties that fluorine atoms impart to drug candidates. Patent CN110724040A discloses a groundbreaking synthetic method for iodo trifluoromethoxy compounds, addressing a long-standing challenge in organic synthesis regarding the simultaneous installation of iodine and trifluoromethoxy groups. This dual functionalization strategy is particularly valuable because the trifluoromethoxy group (OCF3) offers superior lipophilicity and metabolic stability compared to traditional methoxy groups, while the iodine atom serves as a versatile handle for subsequent cross-coupling reactions. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is essential for R&D directors seeking to optimize their synthesis pipelines for next-generation small molecule drugs. The technology described herein not only expands the chemical space available for medicinal chemistry but also provides a robust pathway for the commercial manufacturing of high-value fluorinated building blocks.
The significance of this invention lies in its ability to construct complex functional group compounds under relatively mild conditions, which is a critical factor for the scalability of sensitive pharmaceutical intermediates. Historically, the introduction of fluorine-containing groups has often required harsh reagents or multi-step sequences that compromise overall efficiency and increase waste generation. This patent proposes a streamlined approach that leverages the reactivity of alkenes to achieve difunctionalization in a single operational sequence. By utilizing specific iodinating agents and fluorine salts in a carefully controlled solvent system, the method achieves high conversion rates and selectivity. For procurement managers and supply chain heads, this represents a tangible opportunity for cost reduction in fine chemical manufacturing, as fewer processing steps generally translate to lower operational expenditures and reduced environmental footprint. The ability to access these motifs efficiently is a key competitive advantage in the global market for specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing fluorinated compounds often suffer from significant limitations that hinder their application in large-scale commercial production. Conventional routes typically involve the separate introduction of fluorine and iodine atoms, requiring distinct reaction vessels, purification steps, and often incompatible reaction conditions. This stepwise approach not only increases the total processing time but also leads to cumulative yield losses at each stage, significantly impacting the overall economics of the synthesis. Furthermore, many traditional fluorination reagents are hazardous, difficult to handle, or generate substantial amounts of toxic waste, posing challenges for environmental compliance and worker safety. The lack of stereocontrol in many non-catalytic methods also results in racemic mixtures, necessitating expensive chiral resolution processes later in the synthesis tree. These inefficiencies create bottlenecks in the supply chain, extending lead times for high-purity organic intermediates and increasing the risk of supply disruptions for critical drug substances.
The Novel Approach
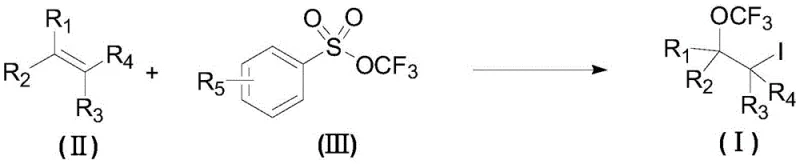
The novel approach detailed in patent CN110724040A overcomes these hurdles by employing a concerted difunctionalization strategy that installs both the iodine and trifluoromethoxy groups in a single reaction pot. A key innovation of this method is the specific order of reagent addition, where the alkene substrate is first mixed with the iodinating agent and fluorine salt before the introduction of the trifluoromethoxysulfonyl reagent. This counter-intuitive sequence was found to dramatically improve the yield of the desired product compared to the reverse addition order, highlighting the importance of kinetic control in this transformation. The use of mild reaction temperatures, ranging from 30 degrees Celsius down to -20 degrees Celsius, allows for the processing of thermally sensitive substrates that would decompose under harsher conditions. This methodological advancement enables the commercial scale-up of complex fluorinated compounds with greater efficiency and reliability, providing a robust solution for the manufacturing of advanced pharmaceutical intermediates.
Mechanistic Insights into AgF-Catalyzed Difunctionalization
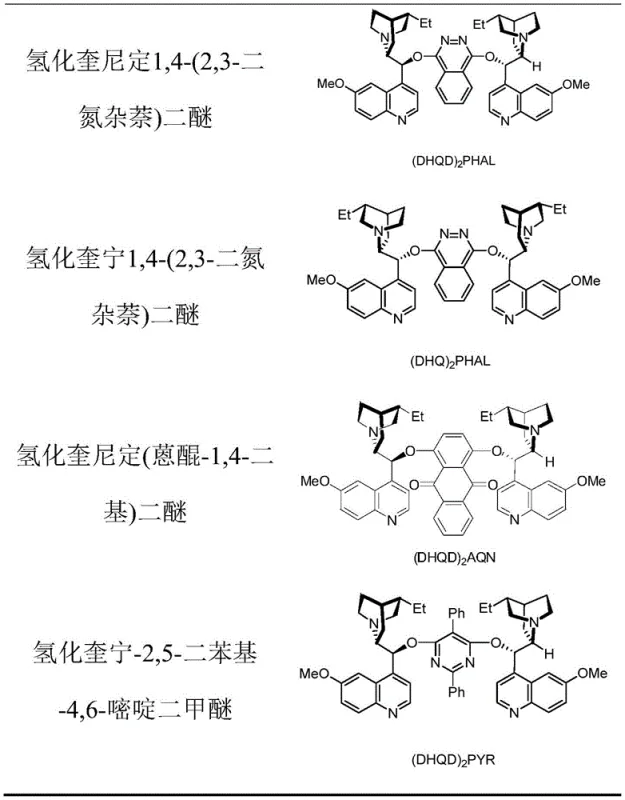
The mechanistic underpinnings of this synthesis involve a sophisticated interplay between the silver salt, the chiral catalyst, and the iodinating agent to activate the alkene double bond. The silver fluoride (AgF) acts as a crucial promoter, likely facilitating the generation of an electrophilic iodine species that initiates the reaction by attacking the electron-rich alkene. This forms a cyclic iodonium intermediate, which is then intercepted by the trifluoromethoxy nucleophile derived from the sulfonyl reagent. The presence of a chiral catalyst, such as the cinchona alkaloid derivatives shown in the patent data, creates a chiral environment around the reaction center, guiding the approach of the nucleophile to one face of the planar intermediate. This asymmetric induction is vital for producing enantiomerically enriched products, which are often required for biological activity in drug molecules. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as catalyst loading and stoichiometry, to maximize enantioselectivity and minimize the formation of undesired regioisomers or byproducts.
Impurity control is another critical aspect of this mechanism, particularly given the high reactivity of the intermediates involved. The patent specifies the use of specific solvents, such as mixtures of acetonitrile and dichloromethane, which help to stabilize the ionic intermediates and prevent side reactions like polymerization or elimination. The stoichiometry of the iodinating agent is also carefully optimized, with a molar excess relative to the substrate ensuring complete consumption of the starting material while avoiding excessive iodine waste. By maintaining strict control over the reaction temperature and addition rates, the formation of thermal degradation products is minimized, resulting in a cleaner crude reaction profile. This high level of purity at the reaction stage reduces the burden on downstream purification processes, such as chromatography or crystallization, thereby enhancing the overall throughput and cost-effectiveness of the manufacturing process for high-purity API intermediates.
How to Synthesize Iodo Trifluoromethoxy Compound Efficiently
The synthesis of these valuable intermediates requires precise adherence to the protocol outlined in the patent to ensure reproducibility and high yield. The process begins with the preparation of a pre-reaction system under an inert atmosphere, typically within a glove box or using standard Schlenk techniques to exclude moisture and oxygen which could deactivate the catalysts. The detailed standardized synthesis steps involve the careful weighing and addition of silver fluoride, the chiral ligand, and the iodinating agent into a sealed vessel containing the appropriate solvent mixture. Once the substrate is added and the mixture is cooled to the target temperature, the trifluoromethoxy reagent is introduced slowly to manage the exotherm and control the reaction kinetics. For a comprehensive guide on the specific quantities and workup procedures, please refer to the standardized protocol below.
- Prepare a pre-reaction system containing fluorine salt, iodinating agent, alkene substrate, and solvent under inert atmosphere.
- Cool the reaction mixture to a temperature range of 0 to -20 degrees Celsius to ensure optimal stereocontrol.
- Add the trifluoromethoxysulfonyl reagent slowly to the pre-reaction system to initiate the difunctionalization reaction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement and supply chain teams looking to optimize their sourcing strategies for fluorinated intermediates. The primary advantage lies in the simplification of the manufacturing process, which directly translates to significant cost savings by eliminating the need for multiple isolation and purification steps associated with traditional sequential functionalization. The reagents utilized in this process, such as N-iodosuccinimide and various fluorine salts, are commercially available commodity chemicals, ensuring a stable and reliable supply chain without dependence on exotic or single-source materials. This availability reduces the risk of supply disruptions and allows for more accurate forecasting of production timelines. Furthermore, the mild reaction conditions reduce energy consumption and equipment wear, contributing to a more sustainable and economically viable production model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction significantly reduces the operational costs associated with solvent usage, labor, and equipment time. By avoiding the isolation of unstable intermediates, the process minimizes material loss and waste disposal costs, leading to a more favorable cost of goods sold (COGS). The high yields reported in the patent examples indicate that raw material utilization is efficient, further driving down the unit cost of the final product. These economic efficiencies make the technology highly attractive for large-scale production where margin optimization is critical for competitiveness in the global fine chemical market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and standard laboratory equipment enhances the robustness of the supply chain. Unlike processes that require custom-synthesized reagents or specialized high-pressure reactors, this method can be implemented in existing multipurpose chemical manufacturing facilities with minimal capital investment. This flexibility allows for rapid scale-up from pilot plant to commercial production, ensuring that supply can meet demand fluctuations without long lead times. The stability of the reagents also simplifies logistics and storage requirements, reducing the complexity of inventory management and ensuring continuous availability for downstream synthesis campaigns.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are amenable to large-volume processing. The ability to operate at near-ambient or slightly sub-ambient temperatures reduces the energy load on cooling systems, which is a significant factor in the environmental footprint of chemical manufacturing. Additionally, the high atom economy of the difunctionalization reaction means that less waste is generated per kilogram of product, simplifying waste treatment and helping facilities meet stringent environmental regulations. This alignment with sustainability goals is increasingly important for multinational corporations seeking to reduce their carbon footprint and adhere to corporate social responsibility mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the scope and limitations of the method. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific project needs. The information provided here serves as a foundational reference for further discussions on process optimization and regulatory compliance.
Q: What is the critical process parameter for maximizing yield in this synthesis?
A: The order of addition is critical. The patent specifies that mixing the alkene substrate with reagents first, followed by the addition of the trifluoromethoxy reagent, significantly improves yield compared to the reverse order.
Q: Can this method be applied to complex natural product derivatives?
A: Yes, the method demonstrates broad substrate scope, including complex structures like Tiamulin derivatives, making it suitable for late-stage functionalization of bioactive molecules.
Q: What catalysts are required for enantioselective variations of this reaction?
A: Chiral catalysts such as (DHQD)2PHAL or (DHQ)2PHAL are employed to induce asymmetry, allowing for the production of enantiomerically enriched intermediates essential for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iodo Trifluoromethoxy Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced fluorination technologies in the development of modern therapeutics and specialty materials. Our team of expert process chemists has extensively evaluated the methodology described in patent CN110724040A and possesses the technical capability to adapt and optimize this route for your specific molecular targets. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our facility is equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of iodo trifluoromethoxy compound meets the highest standards of quality required for pharmaceutical applications.
We invite you to collaborate with us to leverage this innovative synthesis for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this more efficient manufacturing route. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your compound of interest. Let us help you accelerate your development timeline and secure a competitive advantage in the market with our reliable supply of high-quality fluorinated intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
