Revolutionizing Asymmetric Catalysis: High-Purity Spiro-Bis-Dihydrobenzothiole Chiral Phosphoric Acid for Commercial Scale-Up
The Chinese patent CN112920221B introduces a groundbreaking class of chiral phosphoric acid catalysts featuring a novel spiro-bis-dihydrobenzothiole skeleton, representing a significant advancement in asymmetric organocatalysis for pharmaceutical intermediate synthesis. This innovative molecular architecture addresses critical limitations in existing chiral catalyst frameworks by providing enhanced stereochemical control and broader reaction applicability. The patent details comprehensive synthetic methodologies that enable precise construction of these catalysts with defined stereochemistry at the silicon center, offering pharmaceutical manufacturers a powerful tool for producing high-purity chiral compounds with superior enantioselectivity. This development is particularly timely given the increasing demand for efficient asymmetric catalytic processes in the production of complex pharmaceutical intermediates where traditional methods often fall short in terms of selectivity and scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral phosphoric acid catalysts primarily rely on axial chiral binaphthyl or spiro-dihydroindene frameworks, which present significant constraints in asymmetric catalysis applications. The all-carbon spiro structure of conventional spiro-dihydroindene-based catalysts severely limits structural modification possibilities, restricting their ability to accommodate diverse substrate geometries and reaction types required in modern pharmaceutical synthesis. These conventional frameworks often demonstrate inadequate enantioselectivity for challenging transformations, particularly with sterically demanding substrates or when precise stereochemical control is critical for pharmaceutical efficacy and safety profiles. Furthermore, the limited structural diversity among commercially available chiral phosphoric acids creates bottlenecks in process development, forcing manufacturers to compromise on reaction efficiency or invest substantial resources in developing custom catalysts for specific applications. The restricted tunability of existing catalyst systems also hinders optimization for large-scale manufacturing where process robustness and reproducibility are paramount concerns for regulatory compliance and commercial viability.
The Novel Approach
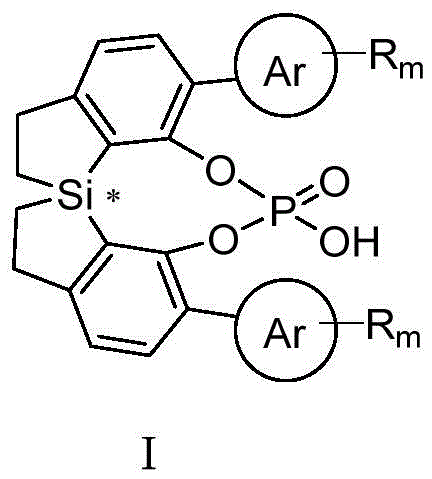
The patented spiro-bis-dihydrobenzothiole skeleton represents a paradigm shift in chiral phosphoric acid design by introducing a silicon-centered chiral element that provides unprecedented structural flexibility and electronic properties. This innovative framework enables precise tuning of steric and electronic parameters through strategic substitution patterns on the aromatic rings, allowing for optimization of catalytic performance across a wide range of asymmetric transformations. The silicon-based spiro structure creates a more rigid yet adaptable chiral environment that significantly enhances enantioselectivity compared to conventional systems, as demonstrated by the patent's experimental data showing up to 99% ee in key reactions. The synthetic methodology described in the patent provides a versatile platform for generating diverse catalyst variants with tailored properties, addressing specific challenges in pharmaceutical intermediate synthesis while maintaining excellent scalability from laboratory to commercial production scales.
Mechanistic Insights into Spiro-Bis-Dihydrobenzothiole Chiral Phosphoric Acid Catalysis
The unique catalytic performance of these novel phosphoric acids stems from their distinctive molecular architecture, which creates an optimal chiral pocket for substrate binding and activation. The silicon-centered spiro structure generates a well-defined three-dimensional environment where the phosphate group is positioned to simultaneously activate both reaction partners through dual hydrogen bonding interactions, while the extended aromatic framework provides precise steric control over the approach trajectory of substrates. This geometric arrangement enables superior facial discrimination during bond formation, resulting in exceptional enantioselectivity across various asymmetric transformations including imine additions, Mannich reactions, and Friedel-Crafts alkylations. The electronic properties of the silicon-containing framework also contribute to enhanced acidity modulation, allowing fine-tuning of catalytic activity without compromising stereoselectivity—a critical advantage for complex pharmaceutical syntheses where reaction conditions must be precisely controlled to avoid side products.
Impurity control is significantly enhanced through this novel catalytic system due to its highly selective reaction pathways that minimize undesired byproducts. The well-defined chiral environment prevents alternative reaction trajectories that typically lead to diastereomeric impurities in traditional catalytic systems, resulting in cleaner reaction profiles that simplify downstream purification processes. This selectivity advantage is particularly valuable in pharmaceutical manufacturing where strict regulatory requirements mandate rigorous control over impurity profiles, often requiring multiple costly purification steps with conventional catalysts. The patent demonstrates through multiple experimental examples that reactions catalyzed by these novel phosphoric acids consistently produce products with superior purity profiles, directly addressing one of the most significant challenges in commercial-scale asymmetric synthesis for pharmaceutical applications.
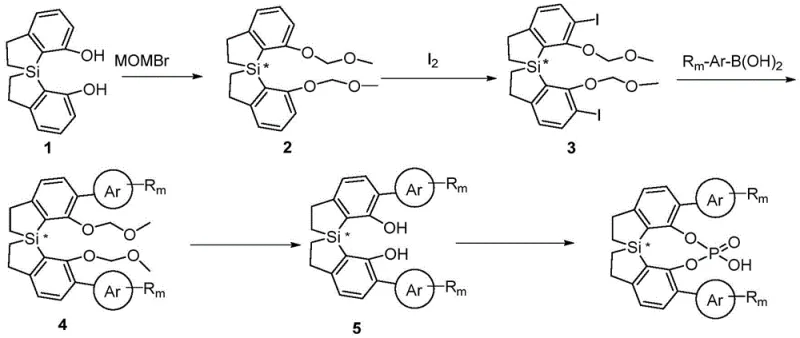
How to Synthesize Spiro-Bis-Dihydrobenzothiole Chiral Phosphoric Acid Efficiently
This innovative synthetic route represents a significant advancement over conventional methods for producing chiral phosphoric acid catalysts, offering improved yields, enhanced stereochemical control, and greater scalability potential for commercial manufacturing operations. The multi-step process begins with readily available diol precursors that undergo selective protection, followed by strategic functionalization to establish the critical silicon-centered chirality. Each transformation has been carefully optimized to ensure high reproducibility and robustness across different scales of operation, addressing key pain points encountered in traditional catalyst synthesis including inconsistent stereochemical outcomes and difficult purification requirements. Detailed standardized synthesis procedures are provided below to enable seamless implementation in pharmaceutical manufacturing environments seeking reliable access to high-performance asymmetric catalysis solutions.
- Protection of diol precursor with MOMBr under basic conditions to form bis-MOM ether intermediate
- Iodination of the protected intermediate using n-BuLi and iodine to generate diiodo compound
- Suzuki-Miyaura coupling with aryl boronic acids followed by deprotection and phosphorylation
Commercial Advantages for Procurement and Supply Chain Teams
This novel catalyst system directly addresses critical pain points in pharmaceutical intermediate manufacturing by offering a more efficient pathway to high-purity chiral compounds while simultaneously improving supply chain reliability and cost-effectiveness. The enhanced enantioselectivity reduces the need for extensive purification steps that typically consume significant resources in traditional manufacturing processes, creating substantial operational efficiencies that translate into tangible commercial benefits without requiring major capital investments or process re-engineering. By eliminating the need for transition metal catalysts in many applications, this technology also removes associated regulatory hurdles and quality control challenges that complicate supply chain management and increase time-to-market for new pharmaceutical products.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes expensive metal removal steps from the manufacturing process while reducing the need for multiple purification stages typically required to achieve pharmaceutical-grade purity standards. This streamlined approach significantly reduces raw material consumption, solvent usage, and processing time without compromising product quality, creating substantial cost savings through process intensification rather than incremental improvements.
- Enhanced Supply Chain Reliability: The synthetic route utilizes readily available starting materials with established global supply chains, minimizing vulnerability to single-source dependencies that often disrupt traditional catalyst production. The robust multi-step process demonstrates excellent reproducibility across different manufacturing scales, ensuring consistent product quality and reliable delivery timelines that support just-in-time manufacturing models preferred by leading pharmaceutical companies.
- Scalability and Environmental Compliance: The synthetic methodology has been specifically designed with green chemistry principles in mind, featuring high atom economy, reduced solvent consumption, and minimal waste generation compared to conventional approaches. The process demonstrates excellent scalability from laboratory to commercial production volumes while maintaining stringent quality specifications, enabling seamless technology transfer without requiring significant revalidation efforts that typically delay market introduction.
Frequently Asked Questions (FAQ)
The following questions and answers are derived directly from the technical details presented in patent CN112920221B, addressing common concerns raised by technical procurement teams evaluating this innovative catalyst system for pharmaceutical manufacturing applications. These responses provide evidence-based insights into the practical implementation and performance characteristics of the spiro-bis-dihydrobenzothiole chiral phosphoric acid catalysts.
Q: How does the spiro-bis-dihydrobenzothiole skeleton improve enantioselectivity compared to traditional binaphthyl-based catalysts?
A: The novel silicon-centered spiro-bis-dihydrobenzothiole framework provides enhanced steric control and electronic modulation, creating a more defined chiral environment that significantly improves enantioselectivity in asymmetric reactions as demonstrated in multiple catalytic applications including imine additions and Mannich reactions.
Q: What are the key advantages of this catalyst system for pharmaceutical intermediate manufacturing?
A: This catalyst system offers superior enantioselectivity (up to 99% ee), simplified purification processes, and eliminates the need for transition metal catalysts, which directly addresses critical pain points in pharmaceutical intermediate synthesis including impurity profile management and regulatory compliance.
Q: How does the synthetic route support scalable manufacturing of these catalysts?
A: The synthetic pathway features robust, high-yielding steps with readily available starting materials and straightforward purification procedures, enabling reliable scale-up from laboratory to commercial production volumes while maintaining stringent quality specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro-Bis-Dihydrobenzothiole Chiral Phosphoric Acid Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex chiral catalysts while maintaining stringent purity specifications required by global regulatory authorities. With state-of-the-art manufacturing facilities equipped with rigorous QC labs and dedicated process chemistry teams, we provide end-to-end solutions for implementing this patented technology into your production workflows with minimal disruption to existing operations. Our technical experts work closely with client R&D teams to optimize catalyst performance for specific applications while ensuring seamless integration with current manufacturing processes through comprehensive route feasibility assessments.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative catalyst system can enhance your specific manufacturing processes. Contact us today to obtain specific COA data and detailed route feasibility assessments tailored to your production requirements and quality standards.
