Advanced Rare Earth Catalysis for Scalable 1,5-Disubstituted-1,2,3-Triazole Manufacturing
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and regioselective synthetic routes for nitrogen-containing heterocycles, particularly 1,2,3-triazoles, due to their profound biological activities ranging from antifungal to anticancer properties. Patent CN103613551A introduces a transformative synthesis method for 1,5-disubstituted-1,2,3-triazole compounds that leverages the unique Lewis acidic properties of rare earth metal triflates. This technology represents a significant departure from conventional methodologies by utilizing nitroalkenes and organic azides as primary building blocks under mild reflux conditions. For R&D directors and process chemists, this patent offers a robust alternative to traditional click chemistry, providing a pathway to access the less common 1,5-isomer with high selectivity and yields exceeding 70%. The strategic use of catalysts such as cerium, samarium, or ytterbium trifluoromethanesulfonate not only enhances reaction efficiency but also aligns with modern green chemistry principles by reducing reliance on precious transition metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazoles has been dominated by the Huisgen 1,3-dipolar cycloaddition between alkynes and organic azides, a reaction notoriously hampered by high activation energy barriers requiring harsh thermal conditions and often resulting in inseparable mixtures of 1,4- and 1,5-regioisomers. While the advent of Copper(I)-catalyzed Azide-Alkyne Cycloaddition (CuAAC) revolutionized the field by enabling room temperature reactions, it strictly confines the product distribution to the 1,4-disubstituted isomer, rendering it useless for applications specifically requiring the 1,5-substitution pattern. Alternatively, Ruthenium(II)-catalyzed systems (RuAAC) can access the 1,5-isomer, but the prohibitive cost of Ruthenium catalysts and the complexity associated with removing trace heavy metal residues from pharmaceutical intermediates pose severe economic and regulatory bottlenecks for commercial scale-up. These traditional pathways often necessitate extensive purification protocols to meet stringent impurity specifications, thereby inflating manufacturing costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN103613551A circumvents these historical constraints by shifting the reactant paradigm from alkynes to nitroalkenes, coupled with organic azides in the presence of inexpensive rare earth metal catalysts. This innovative approach facilitates a highly regioselective formation of the 1,5-disubstituted-1,2,3-triazole core without the formation of unwanted 1,4-isomers, effectively solving the selectivity issue that plagues thermal Huisgen condensations. The reaction operates efficiently in common organic solvents like toluene or chlorobenzene at temperatures between 80°C and 110°C, conditions that are easily manageable in standard glass-lined reactors found in most fine chemical manufacturing facilities. By replacing precious metals with earth-abundant rare earth triflates, the process drastically simplifies the workup procedure, as the catalysts are generally less toxic and easier to separate than their transition metal counterparts, leading to a cleaner crude product profile.
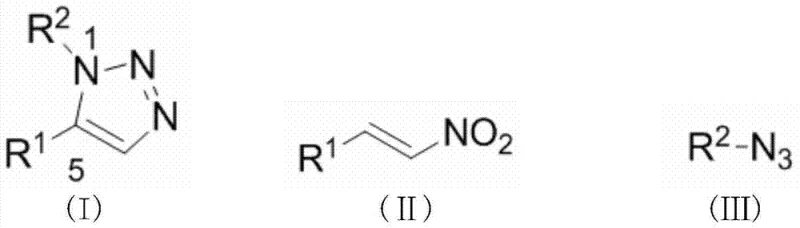
Mechanistic Insights into Rare Earth-Catalyzed Cycloaddition
The efficacy of this synthesis relies on the potent Lewis acidity of the rare earth metal cations, such as Ce(III), Sm(III), or Yb(III), which coordinate with the electron-rich nitro group of the nitroalkene substrate. This coordination significantly lowers the LUMO energy of the nitroalkene, thereby activating the beta-carbon towards nucleophilic attack by the terminal nitrogen of the organic azide dipole. Unlike the concerted mechanism often proposed for thermal cycloadditions, the rare earth catalyst likely stabilizes a stepwise zwitterionic intermediate or a tightly organized transition state that enforces the specific orientation required for 1,5-regioselectivity. This mechanistic pathway ensures that the bulky substituents on the azide and the nitroalkene are positioned to minimize steric clash while maximizing orbital overlap, resulting in the exclusive formation of the 1,5-isomer. Understanding this electronic activation is crucial for process optimization, as it suggests that electron-deficient nitroalkenes may react even more vigorously, allowing for fine-tuning of reaction kinetics through substrate modification.
From an impurity control perspective, the high regioselectivity of this catalytic system is a major advantage for maintaining a clean impurity profile in the final API intermediate. Traditional methods often generate regioisomeric impurities that possess similar physical properties to the desired product, making them extremely difficult to remove via standard crystallization or chromatography. By fundamentally preventing the formation of the 1,4-isomer through catalyst control rather than relying on post-reaction separation, this method inherently reduces the burden on downstream purification units. Furthermore, the use of stable nitroalkene precursors minimizes the risk of side reactions such as polymerization or decomposition that can occur with unstable alkyne species under thermal stress. This inherent stability contributes to a more predictable reaction outcome, ensuring consistent batch-to-batch quality which is paramount for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 1,5-Disubstituted-1,2,3-Triazoles Efficiently
The operational simplicity of this patented method makes it highly attractive for rapid technology transfer from laboratory bench to pilot plant. The standard protocol involves charging a reactor with equimolar amounts of the nitroalkene and organic azide, followed by the addition of the rare earth catalyst at a loading of 1 to 10 mol%. The mixture is then heated to reflux in a solvent system, typically toluene or a toluene-chlorobenzene blend, for a duration ranging from 8 to 48 hours depending on the specific electronic nature of the substrates. Reaction completion is conveniently monitored using thin-layer chromatography (TLC), and upon consumption of the starting materials, the crude product can be isolated via simple filtration and solvent evaporation. For detailed standard operating procedures and specific parameter optimizations for your target molecule, please refer to the standardized synthesis guide below.
- Combine nitroalkene and organic azide in a stoichiometric 1: 1 molar ratio within an organic solvent such as toluene or chlorobenzene.
- Add a rare earth metal catalyst, such as cerium trifluoromethanesulfonate, at a loading of 1-10 mol%.
- Reflux the mixture at 80-110°C for 8-48 hours, monitoring progress via TLC, followed by silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this rare earth-catalyzed synthesis route offers tangible strategic advantages in terms of cost structure and supply security. The shift away from precious metal catalysts like Ruthenium eliminates a significant variable cost component, as rare earth triflates are substantially more affordable and less subject to the volatile market fluctuations that characterize the platinum group metals market. Moreover, the simplified purification workflow reduces the consumption of silica gel and eluents during column chromatography or the need for specialized scavenger resins, directly lowering the cost of goods sold (COGS) for complex pharmaceutical intermediates. This economic efficiency is compounded by the high atom economy of the cycloaddition reaction, which generates minimal waste compared to multi-step substitution routes, aligning with corporate sustainability goals and reducing waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated heavy metal removal steps results in substantial cost savings throughout the production lifecycle. By utilizing readily available rare earth salts that can often be recovered and recycled, manufacturers can achieve a leaner cost structure without compromising on the purity or quality of the final triazole product. The ability to run the reaction in common industrial solvents like toluene further enhances cost efficiency by avoiding the need for specialized, high-boiling, or hazardous polar aprotic solvents that require complex recovery systems.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically nitroalkenes and organic azides, are commoditized chemicals with robust global supply chains, reducing the risk of production stoppages due to raw material shortages. Unlike specialized alkynes which may have limited suppliers and long lead times, nitroalkenes can be sourced from multiple vendors, ensuring continuity of supply for large-scale campaigns. This diversification of the supply base mitigates geopolitical risks and allows procurement teams to negotiate more favorable pricing terms through competitive bidding.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, utilizing standard reflux techniques that translate seamlessly from liter-scale flasks to cubic-meter reactors without the need for exotic high-pressure equipment. The reduced toxicity profile of the rare earth catalysts compared to copper or ruthenium simplifies environmental compliance and worker safety protocols, facilitating faster regulatory approvals for new manufacturing sites. Additionally, the high selectivity of the reaction minimizes the generation of isomeric byproducts, reducing the overall chemical waste load and supporting greener manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rare earth-catalyzed triazole synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific project needs. We encourage technical teams to review these points when assessing potential process improvements or cost-saving opportunities in their current triazole manufacturing workflows.
Q: What distinguishes this rare earth catalytic method from traditional CuAAC reactions?
A: Unlike Cu(I)-catalyzed azide-alkyne cycloaddition (CuAAC) which exclusively yields 1,4-isomers, this rare earth-catalyzed method utilizing nitroalkenes selectively produces 1,5-disubstituted-1,2,3-triazoles, addressing a critical regioselectivity challenge in heterocyclic synthesis.
Q: Are the raw materials for this synthesis readily available for large-scale production?
A: Yes, the process utilizes nitroalkenes and organic azides, which are commercially accessible and stable starting materials, avoiding the need for specialized alkynes often required in traditional triazole synthesis routes.
Q: How does this method impact purification costs compared to Ruthenium-catalyzed routes?
A: By employing earth-abundant rare earth triflates instead of expensive Ruthenium complexes, the method significantly reduces raw material costs and simplifies downstream processing by eliminating complex heavy metal scavenging steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Disubstituted-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality heterocyclic building blocks for the development of next-generation therapeutics and agrochemicals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN103613551A can be successfully translated into reliable industrial output. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1,5-disubstituted-1,2,3-triazole meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of rare earth catalysis, optimizing reaction parameters to maximize yield and minimize impurities for our clients.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this rare earth-catalyzed method for your supply chain. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions, ensuring a seamless partnership that drives value and efficiency in your chemical manufacturing operations.
