Advanced Nickel-Catalyzed Carbonylation for High-Purity Thioester Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of the pharmaceutical and agrochemical industries, particularly regarding the efficient construction of complex molecular scaffolds. A significant breakthrough in this domain is detailed in patent CN112239384B, which discloses a novel preparation method for thioester compounds. Thioesters are not merely academic curiosities; they are pivotal structural motifs found in a myriad of bioactive natural products and critical drug molecules, including notable examples like Timobesone, Pivalopril, and Spironolactone. 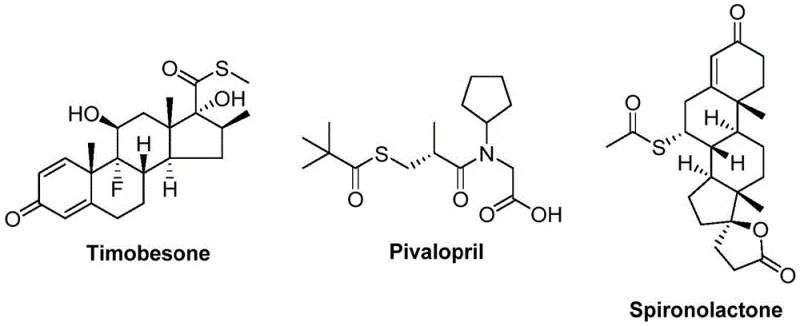 . The ability to synthesize these compounds efficiently is paramount for drug discovery and process development. This patent introduces a transition metal-catalyzed carbonylation strategy that utilizes cheap and readily available arylboronic acids and sulfonyl chlorides as starting materials. By leveraging a specific catalytic system involving nickel triflate and molybdenum carbonyl, the method achieves high reaction efficiency without the need for external oxidants or reductants, marking a substantial improvement over traditional synthetic routes.
. The ability to synthesize these compounds efficiently is paramount for drug discovery and process development. This patent introduces a transition metal-catalyzed carbonylation strategy that utilizes cheap and readily available arylboronic acids and sulfonyl chlorides as starting materials. By leveraging a specific catalytic system involving nickel triflate and molybdenum carbonyl, the method achieves high reaction efficiency without the need for external oxidants or reductants, marking a substantial improvement over traditional synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has relied heavily on the direct acylation of thiols with carboxylic acids or their derivatives. While conceptually straightforward, this classical approach suffers from severe practical drawbacks that hinder its application in large-scale industrial settings. The most prominent issue is the reliance on thiols as the sulfur source. Thiols are notoriously malodorous, creating significant safety and environmental challenges for plant operators and surrounding communities. Furthermore, thiols possess a strong affinity for transition metals, often leading to catalyst poisoning which drastically reduces turnover numbers and necessitates the use of excessive catalyst loading. In addition to these operational hazards, traditional methods often require harsh reaction conditions or stoichiometric amounts of coupling reagents, generating substantial chemical waste. The limited substrate scope of some older methodologies also restricts the ability to introduce diverse functional groups, thereby limiting the chemical space accessible to medicinal chemists during lead optimization phases.
The Novel Approach
In stark contrast to the limitations of thiol-based chemistry, the method described in CN112239384B utilizes sulfonyl chlorides as the sulfur source, effectively circumventing the issues of odor and catalyst deactivation. This innovative route employs a nickel-catalyzed carbonylation reaction where arylboronic acids react with sulfonyl chlorides in the presence of a carbon monoxide source, specifically molybdenum hexacarbonyl. The reaction design is elegant in its simplicity, operating under relatively mild thermal conditions without the requirement for additional oxidizing or reducing agents. This redox-neutral pathway not only simplifies the reaction setup but also enhances the atom economy of the process. The use of arylboronic acids, which are stable and commercially abundant, further broadens the applicability of this method. By shifting the paradigm from thiol acylation to cross-coupling carbonylation, this technology offers a cleaner, safer, and more versatile platform for constructing the thioester bond, directly addressing the pain points of modern process chemistry.
Mechanistic Insights into Nickel-Catalyzed Carbonylation
The core of this technological advancement lies in the sophisticated interplay between the nickel catalyst and the co-catalytic system. The reaction initiates with the activation of the arylboronic acid by the nickel species, likely forming an aryl-nickel intermediate through transmetallation. Simultaneously, the molybdenum hexacarbonyl serves as a solid, easy-to-handle surrogate for gaseous carbon monoxide, releasing CO in situ under the reaction temperature of 120°C. This controlled release of CO ensures a steady concentration of the carbonyl source, facilitating the insertion of the carbonyl group into the nickel-carbon bond. The sulfonyl chloride then participates in the catalytic cycle, potentially undergoing desulfitative coupling or acting as an electrophilic sulfur source that intercepts the acyl-nickel species. 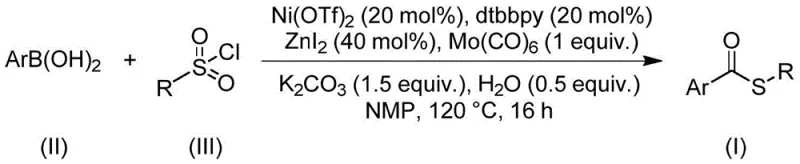 . The presence of zinc iodide and the bipyridine ligand (dtbbpy) is crucial for stabilizing the active nickel species and promoting the reductive elimination step that releases the final thioester product. This mechanistic pathway avoids the formation of unstable intermediates that often plague other carbonylation reactions, ensuring a smooth progression to the desired product.
. The presence of zinc iodide and the bipyridine ligand (dtbbpy) is crucial for stabilizing the active nickel species and promoting the reductive elimination step that releases the final thioester product. This mechanistic pathway avoids the formation of unstable intermediates that often plague other carbonylation reactions, ensuring a smooth progression to the desired product.
From an impurity control perspective, this mechanism offers distinct advantages. The absence of free thiols eliminates the risk of disulfide formation, a common side reaction that complicates purification in traditional methods. Furthermore, the high selectivity of the nickel catalyst towards the sulfonyl chloride minimizes homocoupling of the arylboronic acid, a frequent byproduct in Suzuki-type reactions. The reaction conditions, utilizing N-methylpyrrolidone (NMP) as the solvent, provide a polar environment that supports the solubility of the inorganic bases and salts involved, such as potassium carbonate and zinc iodide. This homogeneous phase promotes efficient mass transfer and consistent reaction kinetics. The robustness of the catalytic system allows it to tolerate a wide range of functional groups, including electron-donating and electron-withdrawing substituents on the aromatic rings, ensuring that the impurity profile remains manageable even with complex substrates.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific parameters to maximize yield and purity. The protocol involves charging a reaction vessel with the nickel catalyst, ligand, molybdenum source, base, and additives, followed by the introduction of the organic substrates and solvent. The mixture is then heated to promote the carbonylation event. Detailed standard operating procedures regarding the precise order of addition and safety precautions for handling pressurized vessels (due to CO generation) are critical for successful execution. For a comprehensive guide on the standardized synthesis steps, please refer to the technical protocol below.
- Combine nickel triflate, dtbbpy ligand, molybdenum carbonyl, zinc iodide, potassium carbonate, water, arylboronic acid, and sulfonyl chloride in NMP solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 16 hours to ensure complete conversion.
- Perform post-treatment involving filtration and silica gel mixing, followed by column chromatography purification to isolate the pure thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits beyond mere chemical elegance. The shift away from malodorous thiols to stable sulfonyl chlorides significantly improves the working environment and reduces the costs associated with specialized scrubbing systems and personal protective equipment. Moreover, the reliance on commodity chemicals like arylboronic acids and sulfonyl chlorides ensures a stable and competitive supply chain, mitigating the risks associated with sourcing exotic or hazardous reagents. The simplified workup procedure, which involves basic filtration and chromatography, reduces the processing time and solvent consumption, leading to a lower overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of expensive oxidants and reductants, combined with the use of earth-abundant nickel instead of precious metals like palladium, results in substantial cost savings. The high atom economy and reduced waste generation further lower the disposal costs, making the process economically attractive for large-scale production. The ability to run the reaction at moderate temperatures without extreme pressure requirements also reduces energy consumption and capital expenditure on specialized high-pressure reactors.
- Enhanced Supply Chain Reliability: Since the key starting materials, arylboronic acids and sulfonyl chlorides, are widely available from multiple global suppliers, the risk of supply disruption is minimized. This diversification of the supply base ensures continuity of production even in volatile market conditions. The robustness of the reaction means that variations in raw material quality can be better tolerated, reducing the rejection rate of incoming batches and smoothing out production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with various substrates in the patent examples. The absence of toxic thiol byproducts simplifies effluent treatment, aiding in compliance with increasingly stringent environmental regulations. The use of NMP, while requiring careful handling, is a well-understood solvent in the industry with established recovery protocols, facilitating the implementation of green chemistry principles through solvent recycling.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this thioester synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity for technical teams evaluating this methodology for their specific applications.
Q: What is the primary advantage of using sulfonyl chloride over thiols in this synthesis?
A: Sulfonyl chlorides serve as a stable, odorless sulfur source that avoids the catalyst poisoning and unpleasant odors typically associated with traditional thiol-based acylation methods.
Q: Does this nickel-catalyzed method require external oxidants or reductants?
A: No, the process is designed to operate without additional oxidants or reductants, simplifying the reaction system and reducing waste generation significantly.
Q: What is the substrate tolerance for this carbonylation reaction?
A: The method exhibits excellent functional group tolerance, successfully accommodating various substituents such as alkyl, alkoxy, and halogen groups on both the arylboronic acid and sulfonyl chloride components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering high-purity thioester intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to adapt and optimize processes like the nickel-catalyzed carbonylation described in CN112239384B allows us to offer cost-effective solutions tailored to your specific project needs.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis for your target molecule. We are ready to provide specific COA data and route feasibility assessments to help you make informed decisions and secure a reliable supply of high-quality pharmaceutical intermediates.
