Advanced One-Step Synthesis of Functionalized Tetraaryl-Thio-Chlorophyll Derivatives for Commercial Scale
Advanced One-Step Synthesis of Functionalized Tetraaryl-Thio-Chlorophyll Derivatives for Commercial Scale
The landscape of functionalized porphyrin chemistry has evolved significantly with the introduction of novel heteroatom-substituted macrocycles, as detailed in the groundbreaking patent CN109928978B. This intellectual property discloses a sophisticated yet operationally simple methodology for synthesizing 5,10,15,20-tetraaryl-3-thio-2-nitrogen-21-carbon chlorophyll compounds, a class of molecules that holds immense potential for applications ranging from molecular switches to anion sensors. The core innovation lies in the direct thionation of the porphyrin macrocycle, transforming the 5,10,15,20-tetraaryl-2-aza-21-carboporphyrin precursor into a sulfur-containing derivative through a single, mild reaction step. This breakthrough addresses long-standing challenges in modifying the electronic properties of carbaporphyrinoids without compromising the stability of the conjugated 18-pi electron system. For research directors and procurement specialists alike, this patent represents a pivotal shift towards more efficient, safer, and cost-effective manufacturing pathways for high-value specialty chemicals.
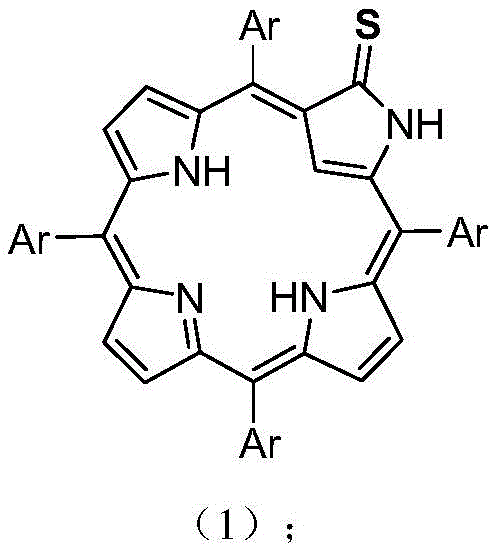
The structural integrity and versatility of these compounds are paramount for their function as reliable pharma intermediate supplier candidates or advanced material precursors. As illustrated in the patent documentation, the target molecule retains the aromatic character essential for its optical and electronic applications while introducing a sulfur atom at the 3-position, which drastically alters its coordination chemistry and redox properties. The ability to tune these properties by varying the aryl substituents (Ar) allows for precise customization of the final product's characteristics, making it an invaluable asset for developing next-generation biologically-related metal chemistry tools. This level of structural control is rarely achieved with such operational simplicity, marking a significant advancement in the field of organic synthesis for complex heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of porphyrin and carbaporphyrin macrocycles has been plagued by synthetic inefficiencies that hinder commercial scalability and increase production costs. Traditional methods often rely on harsh reaction conditions, including elevated temperatures and the use of aggressive reagents that can degrade the sensitive macrocyclic structure or lead to complex mixtures of regioisomers. Furthermore, many existing protocols necessitate the use of transition metal catalysts, which introduce significant downstream processing burdens; removing trace metals to meet the stringent purity specifications required for pharmaceutical or electronic applications is both time-consuming and expensive. These conventional routes frequently suffer from low atom economy and generate substantial hazardous waste, creating environmental compliance issues that modern supply chains strive to avoid. The complexity of multi-step syntheses also increases the risk of yield loss at each stage, ultimately driving up the cost per kilogram of the final active ingredient.
The Novel Approach
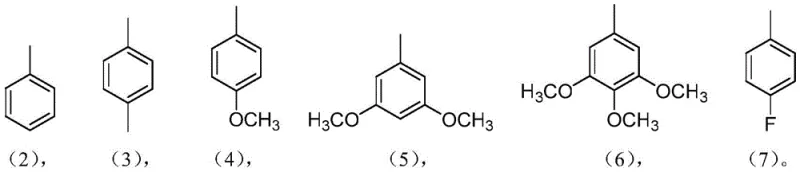
In stark contrast, the methodology described in CN109928978B offers a streamlined, one-step solution that bypasses these traditional bottlenecks entirely. By utilizing a combination of piperidine, carbon disulfide, and potassium carbonate in tetrahydrofuran at room temperature, the process achieves direct thionation with remarkable selectivity and efficiency. This approach eliminates the need for expensive transition metal catalysts and avoids the thermal stress that often compromises product quality in older methods. The reaction proceeds smoothly to convert the starting 5,10,15,20-tetraaryl-2-nitrogen-21-carbon porphyrin, shown in the structural diagrams, into the desired 3-thio derivative with high fidelity. Moreover, the versatility of this method is demonstrated by its compatibility with a wide range of aryl substituents, allowing manufacturers to produce a diverse library of derivatives from a single robust platform. This flexibility is crucial for cost reduction in fine chemical manufacturing, as it enables the rapid adaptation of production lines to meet specific client requirements without retooling entire processes.
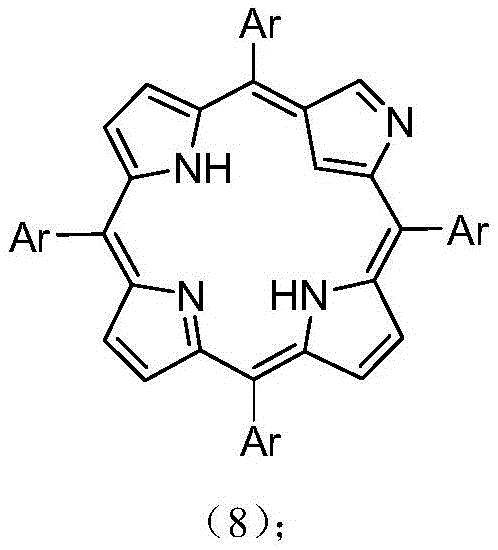
Mechanistic Insights into Piperidine-Mediated Thionation
The mechanistic pathway underpinning this transformation is a fascinating example of nucleophilic activation within a constrained macrocyclic environment. The reaction likely initiates with the deprotonation of the pyrrolic nitrogen or the activation of the carbon disulfide by the basic piperidine species, generating a reactive nucleophile that attacks the electrophilic centers of the porphyrin ring. Unlike typical electrophilic aromatic substitutions that might disrupt the conjugated system, this specific thionation preserves the aromatic 18-pi electron count, ensuring that the fundamental optical properties of the chlorophyll analogue remain intact. The presence of potassium carbonate acts as a mild base to facilitate the equilibrium without promoting side reactions such as ring opening or polymerization, which are common pitfalls in porphyrin chemistry. Understanding this mechanism is critical for R&D teams aiming to further optimize the process, as it highlights the delicate balance between reactivity and stability required to functionalize such complex architectures successfully.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in ensuring high product purity. Harsh conditions often lead to the formation of oxidized byproducts or over-substituted species that are difficult to separate chromatographically. However, the room temperature protocol described here minimizes thermal degradation and limits the formation of such impurities, resulting in a cleaner crude reaction mixture. The subsequent purification via column chromatography using a dichloromethane and ethyl acetate eluent system is highly effective at isolating the target red band, yielding a dark brown product of exceptional quality. This robustness in impurity profile management is a key selling point for procurement managers who prioritize consistent batch-to-batch quality and reduced analytical testing overheads in their supply chain operations.
How to Synthesize 5,10,15,20-Tetraaryl-3-Thio-2-Nitrogen-21-Carbon Chlorophyll Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process begins with the precise weighing of the tetraaryl-2-aza-21-carboporphyrin substrate along with piperidine, carbon disulfide, and potassium carbonate, which are then suspended in tetrahydrofuran. The mixture is stirred at ambient temperature for a prolonged period, typically around 48 hours, to ensure complete conversion as monitored by thin-layer chromatography. Once the reaction is deemed complete, the workup involves a straightforward cooling step followed by direct separation using silica gel column chromatography. The detailed standardized synthesis steps below provide the exact parameters required to replicate this high-yielding transformation reliably.
- Combine 5,10,15,20-tetraaryl-2-nitrogen-21-carboporphyrin with piperidine, carbon disulfide, and potassium carbonate in a reactor.
- Add tetrahydrofuran solvent and stir the mixture at room temperature for approximately 48 hours while monitoring progress via TLC.
- Upon completion, separate the product using column chromatography with a dichloromethane and ethyl acetate eluent, then concentrate and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere chemical curiosity. The elimination of transition metal catalysts not only simplifies the purification workflow but also removes a significant cost driver associated with metal scavenging resins and extensive washing procedures. This streamlining of the downstream processing directly translates to reduced operational expenditures and shorter cycle times, enhancing the overall agility of the manufacturing process. Furthermore, the use of common, commercially available solvents like tetrahydrofuran and standard reagents ensures that raw material sourcing remains stable and resilient against market fluctuations, a critical factor for maintaining supply continuity in volatile global markets.
- Cost Reduction in Manufacturing: The primary economic advantage stems from the drastic simplification of the reaction setup and workup. By operating at room temperature, the process eliminates the energy costs associated with heating or cooling reactors, leading to substantial utility savings over large-scale campaigns. Additionally, the avoidance of precious metal catalysts removes the need for expensive recovery systems or disposal protocols for heavy metal waste, further lowering the total cost of ownership. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, thereby improving the overall mass balance and yield efficiency of the production line.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method contributes significantly to supply chain stability. Because the reaction tolerates a variety of aryl substituents without requiring condition adjustments, manufacturers can maintain a flexible inventory of intermediates that can be rapidly converted into different final products based on demand. This adaptability reduces the risk of stockouts for specific derivatives and allows for a more responsive just-in-time manufacturing model. The reliance on stable, non-hazardous reagents also mitigates the risk of regulatory shutdowns or shipping delays often associated with more dangerous chemical precursors.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is inherently safer due to the mild exothermic profile and the absence of pyrophoric or highly toxic reagents. The simplified waste stream, devoid of heavy metals, facilitates easier treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces liability but also aligns with the sustainability goals of major multinational corporations, making the supplier a more attractive partner for long-term contracts focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized chlorophyll derivatives. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this material into their own R&D pipelines or commercial product lines.
Q: What are the key advantages of this new synthesis method over traditional porphyrin modifications?
A: This method operates at room temperature without transition metal catalysts, significantly simplifying purification and reducing heavy metal contamination risks common in older routes.
Q: Can the substituent groups on the aryl rings be varied easily?
A: Yes, the process is highly versatile and accommodates various aryl substituents including methyl, methoxy, and fluoro groups without altering the core reaction conditions.
Q: Is this process suitable for large-scale manufacturing?
A: The use of standard solvents like THF and simple column chromatography purification makes the route highly scalable for industrial production of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,10,15,20-Tetraaryl-3-Thio-2-Nitrogen-21-Carbon Chlorophyll Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the development of advanced functional materials and pharmaceutical intermediates. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market launch is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 5,10,15,20-tetraaryl-3-thio-2-nitrogen-21-carbon chlorophyll delivered meets the highest international standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative chemistry can drive value in your specific applications. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this route for your proprietary projects, our experts are ready to assist. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments tailored to your unique business needs.
