Advanced Enantioselective Reduction Technologies for High-Purity Pharmaceutical Intermediates and Commercial Scale-Up
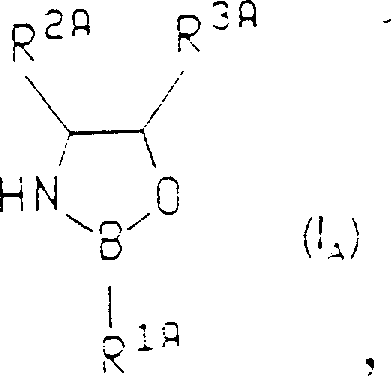
The landscape of asymmetric synthesis has been fundamentally transformed by the innovations detailed in patent CN1124962A, which introduces a novel class of enantioselective 1,3,2-oxazaborolidine catalysts designed for the efficient reduction of prochiral ketones. This technology addresses the critical industry demand for optically pure alcohols, which serve as indispensable building blocks in the manufacture of active pharmaceutical ingredients (APIs) and advanced agrochemicals. Unlike earlier methodologies that often struggled with poor stereocontrol or required expensive stoichiometric chiral auxiliaries, this invention leverages the unique Lewis acidic properties of boron within a rigid chiral framework to direct hydride delivery with exceptional precision. The patent outlines specific structural variations, including cis-disubstituted systems and fused bicyclic architectures, which create a highly defined chiral environment around the catalytic center. For R&D directors and process chemists, understanding the nuances of this catalytic system is essential for developing robust, scalable routes to high-value chiral intermediates that meet stringent regulatory purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of enantiomerically enriched alcohols relied heavily on stoichiometric chiral reducing agents or resolution techniques, both of which present significant drawbacks in a commercial manufacturing setting. Stoichiometric methods, such as those utilizing chiral hydrides, generate substantial amounts of chemical waste, as every mole of product requires a mole of expensive chiral reagent, leading to poor atom economy and inflated raw material costs. Furthermore, resolution processes inherently cap the maximum theoretical yield at 50%, necessitating complex recycling streams for the unwanted enantiomer or resulting in significant material loss. Traditional catalytic methods often suffered from inconsistent enantioselectivity, particularly with substrates where the steric differentiation between substituents was minimal, leading to products contaminated with unacceptable levels of the wrong enantiomer that are difficult and costly to remove. These inefficiencies create bottlenecks in supply chains and increase the environmental footprint of chemical production, making them less attractive for modern green chemistry initiatives.
The Novel Approach
The methodology disclosed in CN1124962A offers a transformative solution by employing chiral 1,3,2-oxazaborolidines that function effectively at catalytic loadings, typically ranging from 5 to 10 mol%. This shift from stoichiometric to catalytic usage dramatically improves the economic profile of the reaction by reducing the consumption of chiral materials while maintaining high turnover numbers. The core innovation lies in the specific substitution patterns on the oxazaborolidine ring, such as the cis-arrangement of alkyl and aryl groups, which effectively block one face of the catalyst and force the substrate to approach from a single, well-defined trajectory. This structural rigidity ensures that even challenging substrates can be reduced with high enantiomeric excess, often exceeding 90% ee, thereby simplifying purification protocols. Additionally, the ability to generate these catalysts in situ from commercially available amino alcohols further streamlines the process, removing the need for isolated catalyst handling and storage.
Mechanistic Insights into Oxazaborolidine-Catalyzed Borane Reduction
The mechanistic pathway of this transformation is driven by the coordination of the prochiral ketone to the electron-deficient boron atom of the oxazaborolidine catalyst, which activates the carbonyl group towards nucleophilic attack. Upon binding, the catalyst-substrate complex adopts a specific conformation dictated by the steric bulk of the substituents at the C4 and C5 positions of the five-membered ring, effectively shielding one face of the carbonyl oxygen. The subsequent delivery of a hydride ion from the borane reducing agent occurs exclusively from the less hindered face, resulting in the formation of the desired chiral alcohol with high stereochemical fidelity. This Lewis acid activation mechanism is distinct from simple hydride reductions, as the chiral information is transferred directly through the transition state geometry rather than relying solely on the chirality of the reducing agent itself. Understanding this interaction is crucial for optimizing reaction conditions, as factors such as solvent polarity and temperature can influence the stability of this key intermediate complex.
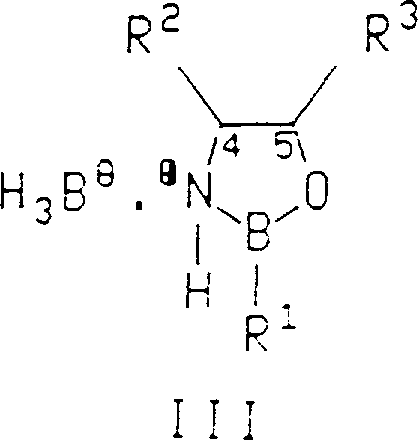
Impurity control in this system is inherently managed by the high specificity of the catalyst-substrate interaction, which minimizes the formation of the undesired enantiomer at the source. However, process chemists must also account for potential side reactions, such as the over-reduction of sensitive functional groups or the background reduction of the ketone by free borane in the absence of the catalyst. To mitigate these risks, the protocol emphasizes the controlled addition of the borane reducing agent, ensuring that the concentration of free hydride remains low relative to the catalyst-bound species. Furthermore, the choice of borane source, such as borane-dimethyl sulfide or borane-tetrahydrofuran complex, can influence the reaction kinetics and selectivity, allowing for fine-tuning of the process to match specific substrate requirements. By maintaining strict control over stoichiometry and addition rates, manufacturers can achieve consistent product quality with minimal byproduct formation, facilitating easier downstream processing and higher overall yields.
How to Synthesize Chiral Alcohols Efficiently
The synthesis of high-purity chiral alcohols using this technology involves a streamlined sequence that begins with the preparation or in situ generation of the active oxazaborolidine catalyst. Detailed procedural guidelines emphasize the importance of anhydrous conditions and inert atmospheres to preserve the integrity of the boron-containing species, which are susceptible to hydrolysis. The reaction is typically conducted in solvents like tetrahydrofuran or toluene, where the catalyst exhibits optimal solubility and stability. Following the establishment of the catalytic species, the prochiral ketone is introduced, followed by the careful, dropwise addition of the borane reducing agent to manage the exothermic nature of the hydride transfer.
- Prepare the chiral catalyst either by isolating the 1,3,2-oxazaborolidine compound or generating it in situ from chiral amino alcohols and borane sources.
- Dissolve the prochiral ketone substrate in an inert solvent like THF or toluene and cool the mixture to the specified reaction temperature range.
- Add the borane reducing agent slowly to regulate the exotherm, stir until completion, and quench with a proton source like methanol to isolate the optically pure alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this catalytic technology offers substantial strategic benefits that extend beyond mere technical performance. The shift to a catalytic regime significantly reduces the dependency on expensive chiral reagents, as the catalyst is used in substoichiometric amounts and can often be derived from inexpensive, commodity-grade chiral pool materials like ephedrine or norephedrine derivatives. This reduction in raw material intensity directly translates to lower variable costs per kilogram of product, enhancing the overall margin profile for high-volume manufacturing campaigns. Moreover, the elimination of stoichiometric chiral auxiliaries simplifies the waste stream, reducing the burden on waste treatment facilities and lowering disposal costs associated with hazardous chemical byproducts. For supply chain managers, the robustness of the reaction conditions and the availability of starting materials ensure a reliable supply of critical intermediates without the risk of bottlenecks associated with specialized reagent sourcing.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily driven by the drastic reduction in catalyst loading compared to traditional stoichiometric methods, which lowers the direct material cost significantly. By avoiding the use of precious metal catalysts or complex chiral ligands that require multi-step synthesis, the process utilizes cost-effective boron chemistry that is scalable and economically viable for large-scale production. The simplified workup procedure, which often involves standard aqueous quenching and extraction, further reduces operational expenses by minimizing solvent usage and processing time. Consequently, the total cost of goods sold (COGS) for the resulting chiral alcohols is optimized, providing a competitive edge in the marketplace for generic API intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as common amino alcohols and commercial borane complexes, ensures a stable and resilient supply chain that is less vulnerable to geopolitical disruptions or single-source supplier risks. The in situ generation capability of the catalyst means that manufacturers do not need to maintain large inventories of sensitive, pre-formed catalysts, thereby reducing storage costs and the risk of material degradation over time. This flexibility allows for just-in-time manufacturing strategies, where production can be ramped up quickly in response to market demand without lengthy lead times for specialized reagent procurement. Such agility is critical for maintaining continuity of supply in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the patent examples which cover a range of substrate loads without loss of efficiency, making it suitable for transition from pilot plant to multi-ton commercial production. The use of boron-based reagents, while requiring careful handling, avoids the generation of heavy metal waste streams associated with transition metal catalysis, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The high atom economy of the catalytic reduction minimizes waste generation, supporting green chemistry principles and reducing the environmental footprint of the manufacturing facility. This compliance with environmental standards mitigates regulatory risk and enhances the company's reputation as a responsible chemical manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enantioselective reduction technology in industrial settings. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this chemistry into existing production lines.
Q: What are the primary advantages of using oxazaborolidine catalysts over traditional stoichiometric reductions?
A: The primary advantages include the ability to use catalytic amounts (typically 5-10 mol%) rather than stoichiometric quantities, which drastically reduces reagent costs and waste generation. Furthermore, these catalysts provide superior enantioselectivity, often achieving greater than 90% ee, which simplifies downstream purification and ensures higher quality API intermediates.
Q: Can these catalysts be generated in situ, and how does this impact process efficiency?
A: Yes, the patent explicitly describes methods for in situ generation of the catalyst from readily available chiral amino alcohols and borane reagents. This approach eliminates the need for a separate catalyst isolation step, streamlining the workflow, reducing processing time, and minimizing exposure to air-sensitive intermediates, thereby enhancing overall operational efficiency.
Q: What types of ketone substrates are compatible with this enantioselective reduction method?
A: The technology demonstrates broad substrate scope, effectively reducing various prochiral ketones including aryl alkyl ketones, cyclic ketones like tetralone, and ketones containing heteroatoms. The steric difference between the substituents on the ketone influences the degree of enantioselectivity, with larger differences generally yielding higher optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazaborolidine Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that advanced catalytic technologies play in accelerating drug development and optimizing manufacturing costs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity chiral intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of complex oxazaborolidine catalysts or the toll manufacturing of chiral alcohol intermediates, our infrastructure is designed to support your most demanding projects with reliability and precision.
We invite you to engage with our technical procurement team to discuss how this patented technology can be tailored to your specific synthetic needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this catalytic route for your target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
