Advanced Silver Catalysis for High-Purity Fully Substituted Pyrazole Intermediates
Advanced Silver Catalysis for High-Purity Fully Substituted Pyrazole Intermediates
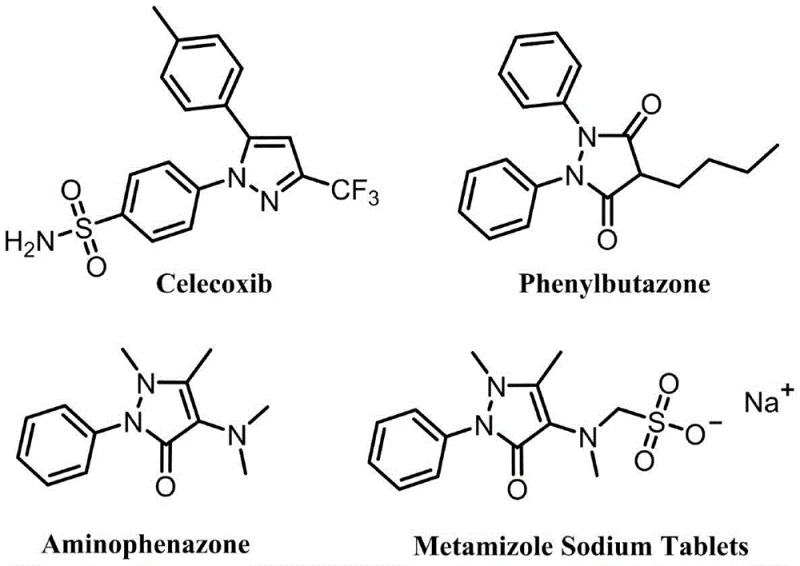
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly the privileged pyrazole scaffold. A groundbreaking development in this domain is detailed in Chinese Patent CN113773253A, which discloses a novel method for synthesizing fully substituted pyrazole compounds via silver catalysis. This technology represents a paradigm shift from traditional synthetic routes, offering a streamlined pathway to complex molecular architectures that serve as critical intermediates for blockbuster drugs such as Celecoxib and Phenylbutazone. By leveraging silver iodide (AgI) as a catalyst in a toluene solvent system, this process achieves exceptional conversion rates under remarkably mild thermal conditions. For R&D directors and procurement specialists alike, this patent offers a compelling solution to the longstanding challenges of regioselectivity and purification in pyrazole synthesis, promising a reliable pharmaceutical intermediate supplier capability for high-value active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrazole ring has relied heavily on the condensation of 1,3-dicarbonyl compounds with hydrazine derivatives. While conceptually straightforward, these classical approaches suffer from significant drawbacks that hinder their utility in modern process chemistry. The reaction often requires harsh acidic or basic conditions and elevated temperatures, which can lead to the degradation of sensitive functional groups and the formation of complex impurity profiles. Furthermore, the use of hydrazine and its analogues poses serious safety and environmental concerns due to their toxicity and potential carcinogenicity. Another critical limitation is the lack of regiocontrol; when unsymmetrical ketones are employed, mixtures of regioisomers are frequently obtained, necessitating difficult and costly separation processes that drastically reduce overall yield. These factors collectively contribute to high manufacturing costs and extended lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates.
The Novel Approach
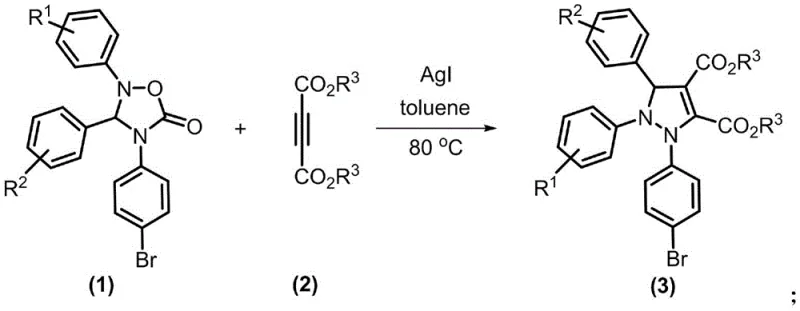
In stark contrast to these legacy methods, the technology described in patent CN113773253A introduces a sophisticated silver-catalyzed cycloaddition strategy that circumvents these issues entirely. The core innovation lies in the utilization of 1,4-oxadiazole-5-one compounds as masked 1,3-dipole equivalents, which react with alkyne compounds in the presence of a silver iodide catalyst. This approach allows for the direct assembly of the fully substituted pyrazole core in a single operational step with high atom economy. The reaction proceeds smoothly in toluene at temperatures between 70°C and 80°C, conditions that are easily maintainable in standard glass-lined reactors without the need for specialized high-pressure equipment. As illustrated in the general reaction scheme below, the transformation is clean and efficient, converting readily available starting materials into valuable heterocyclic products with minimal waste generation.
Mechanistic Insights into AgI-Catalyzed Cyclization
The efficacy of this synthetic route is rooted in the unique Lewis acidic properties of the silver cation, which plays a pivotal role in activating the alkyne component towards nucleophilic attack. Mechanistically, the silver iodide catalyst coordinates with the triple bond of the dimethyl butynedioate (or other alkyne substrates), increasing its electrophilicity and facilitating the initial interaction with the nitrogen atom of the oxadiazole ring. This coordination lowers the activation energy barrier for the ring-opening of the oxadiazole moiety, triggering a cascade of bond reorganizations that ultimately result in the formation of the stable pyrazole ring. The mild nature of the silver catalyst ensures that sensitive substituents on the aromatic rings, such as halogens or methoxy groups, remain intact throughout the process, thereby preserving the structural integrity required for downstream biological activity. This level of chemoselectivity is crucial for R&D teams aiming to synthesize libraries of analogues for structure-activity relationship (SAR) studies without encountering decomposition issues.
Furthermore, the reaction design inherently minimizes the formation of by-products, a common plague in heterocyclic synthesis. The specific stoichiometry employed, typically a 1:3 molar ratio of the oxadiazole precursor to the alkyne, drives the equilibrium towards the desired product while suppressing oligomerization of the alkyne. Post-reaction analysis via TLC and NMR confirms the high purity of the crude material, often exceeding 90% purity prior to chromatographic purification. This inherent cleanliness translates directly to simplified downstream processing; the elimination of heavy metal scavengers or complex extraction protocols reduces the burden on quality control laboratories. For process chemists, understanding this mechanism validates the robustness of the method, ensuring that scale-up efforts will not be derailed by unpredictable exotherms or runaway side reactions.
How to Synthesize Fully Substituted Pyrazole Efficiently
To implement this cutting-edge methodology in a laboratory or pilot plant setting, strict adherence to the optimized parameters outlined in the patent is essential. The process begins with the precise weighing of the 1,4-oxadiazole-5-one substrate and the alkyne coupling partner, ensuring the correct 1:3 molar ratio is maintained to maximize conversion. The reaction is conducted in anhydrous toluene, a solvent chosen for its ability to dissolve both organic substrates effectively while withstanding the reaction temperature of 80°C without degradation. The addition of silver iodide, typically at a loading of roughly 30 mol% relative to the substrate, initiates the catalytic cycle. Detailed standardized operating procedures regarding mixing speeds, heating ramp rates, and quenching protocols are critical for reproducibility. For a comprehensive breakdown of the exact experimental steps and workup procedures, please refer to the technical guide below.
- Combine 1,4-oxadiazole-5-one and dimethyl butynedioate in toluene with silver iodide catalyst.
- Heat the reaction mixture to 80°C and stir for 2.5 to 3.5 hours under inert atmosphere.
- Perform aqueous workup, extract with ethyl acetate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this silver-catalyzed route offers transformative benefits for procurement managers and supply chain heads tasked with optimizing the cost of goods sold (COGS) for API manufacturing. The primary driver of value is the drastic simplification of the synthetic sequence. By replacing multi-step, hazardous hydrazine condensations with a single-pot catalytic cycle, manufacturers can significantly reduce labor costs, energy consumption, and reactor occupancy time. The use of toluene, a commodity solvent with a well-established global supply chain, eliminates the reliance on exotic or expensive specialty solvents that often face availability fluctuations. Moreover, the high yields reported in the patent examples, ranging consistently from 70% to over 90%, ensure that raw material utilization is maximized, directly contributing to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive and hazardous reagents like hydrazine hydrate. Traditional methods often require stoichiometric amounts of toxic reagents and generate large volumes of saline waste, incurring high disposal fees. In contrast, the silver-catalyzed method utilizes a catalytic amount of AgI, which, while a precious metal, is used efficiently and can potentially be recovered from the aqueous waste streams. The mild reaction conditions (80°C) also imply lower energy costs for heating compared to refluxing conditions often required in older protocols. Additionally, the high purity of the crude product reduces the load on purification units, extending the lifecycle of chromatography columns and reducing solvent usage for recrystallization, leading to a leaner, more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. The 1,4-oxadiazole-5-one precursors and dimethyl butynedioate are commercially accessible fine chemicals with stable supply lines, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supplies, such as steam pressure or cooling water temperature, ensuring consistent batch-to-batch quality. This reliability is critical for maintaining continuous production schedules and meeting the stringent delivery deadlines demanded by multinational pharmaceutical clients. The ability to produce high-purity intermediates consistently strengthens the supplier's position as a dependable partner in the global value chain.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory synthesis to multi-ton commercial production is facilitated by the homogeneous nature of the reaction mixture and the absence of gas evolution or highly exothermic events. The use of toluene allows for straightforward solvent recovery and recycling, aligning with green chemistry principles and reducing the facility's environmental footprint. Regulatory compliance is simplified as the process avoids the generation of genotoxic impurities often associated with hydrazine chemistry. This makes the regulatory filing process for new drug applications smoother, as the impurity profile is cleaner and easier to characterize. Consequently, this method supports the commercial scale-up of complex pharmaceutical intermediates with a clear path towards sustainable and compliant manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this silver-catalyzed pyrazole synthesis. These insights are derived directly from the experimental data and beneficial effects reported in patent CN113773253A, providing clarity on the practical application of this technology in an industrial setting. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using Silver Iodide over traditional hydrazine methods?
A: The silver iodide catalyzed method operates under significantly milder conditions (70-80°C) compared to the harsh thermal requirements of traditional hydrazine cyclizations, resulting in higher product purity and reduced side reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common industrial solvents like toluene and achieves high yields (up to 92%), making it highly scalable and economically viable for commercial manufacturing.
Q: What is the typical reaction time for this synthesis?
A: The reaction typically completes within 2.5 to 3.5 hours at 80°C, representing a significant improvement in efficiency over multi-step conventional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fully Substituted Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the silver-catalyzed pyrazole synthesis in driving innovation within the pharmaceutical sector. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel pyrazole derivatives or the optimization of existing routes for cost reduction in API manufacturing, our infrastructure is designed to support your most demanding projects with speed and precision.
We invite you to leverage our technical expertise to enhance your supply chain efficiency and accelerate your drug development timelines. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize yield and reduce waste. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target compounds. Let us collaborate to bring your next-generation therapeutics to market faster and more economically.
