Advanced Metal-Free Synthesis of N-Aryl Indoline Derivatives for Commercial Pharmaceutical Intermediates
Introduction to Next-Generation N-Aryl Indoline Synthesis
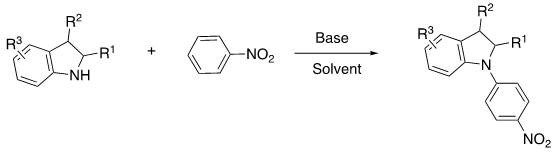
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways for constructing complex heterocyclic scaffolds, particularly N-aryl indoline derivatives which serve as critical building blocks for bioactive alkaloids and therapeutic agents. A significant breakthrough in this domain is documented in patent CN111732528A, which discloses a novel preparation method utilizing direct oxidative dehydrogenation coupling (CDC). This technology represents a paradigm shift from traditional transition-metal catalyzed cross-couplings to a more atom-economical, metal-free protocol. By leveraging nitrobenzene compounds as the aryl source and substituted indolines as the amine component, this method achieves direct C-N bond formation under mild conditions. For R&D directors and procurement specialists, this innovation offers a compelling alternative that simplifies supply chains and reduces the environmental footprint associated with heavy metal waste disposal, positioning it as a highly attractive route for the industrial production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-aryl indoline frameworks has relied heavily on classical strategies such as the partial hydrogenation of N-aryl indoles or the cyclization of 3-chlorostyrenes with primary amines. While effective, these routes often suffer from poor atom economy and require multi-step sequences involving the pre-functionalization of substrates, such as the synthesis of aryl halides. Furthermore, the ubiquitous Buchwald-Hartwig amination, although powerful, necessitates the use of expensive palladium catalysts and specialized phosphine ligands. These transition metal systems introduce significant challenges in downstream processing, including the rigorous removal of trace metal residues to meet stringent regulatory limits for active pharmaceutical ingredients (APIs). The generation of stoichiometric amounts of halide salts as by-products further complicates waste management and increases the overall cost of goods, making these conventional methods less desirable for large-scale commercial manufacturing where cost reduction in API intermediate manufacturing is a primary objective.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent employs a direct oxidative dehydrogenation coupling (CDC) strategy that bypasses the need for pre-halogenated precursors entirely. This approach utilizes readily available nitrobenzene derivatives as the arylating agent, reacting directly with the N-H bond of the indoline scaffold. The reaction is promoted by inexpensive inorganic or alkoxide bases, such as potassium tert-butoxide, in the presence of molecular oxygen as the terminal oxidant. This metal-free system not only enhances the atom economy by producing water or simple reduced nitrogen species as benign by-products but also drastically simplifies the purification process. The absence of transition metals eliminates the risk of heavy metal contamination, thereby reducing the burden on quality control laboratories and ensuring a cleaner impurity profile for the final product. This streamlined process exemplifies how modern synthetic chemistry can address both economic and environmental constraints simultaneously.
Mechanistic Insights into Base-Promoted Oxidative Coupling
The mechanistic pathway of this transformation involves a sophisticated interplay between base activation and aerobic oxidation, distinct from radical or metal-mediated cycles. Initially, the strong base deprotonates the secondary amine nitrogen of the indoline ring, generating a nucleophilic indolinyl anion. This activated species then engages with the nitrobenzene substrate, likely through a nucleophilic aromatic substitution or a single-electron transfer mechanism facilitated by the electron-deficient nature of the nitro group. Molecular oxygen plays a crucial dual role in this system, acting both as an oxidant to regenerate the active catalytic species or intermediate radicals and as a driving force to push the equilibrium towards the formation of the stable N-aryl C-N bond. The low temperature conditions, typically maintained around -30°C, are critical for controlling the reactivity of the intermediates, preventing over-oxidation or polymerization side reactions that could compromise the yield and purity of the desired N-aryl indoline derivatives.
From an impurity control perspective, this metal-free mechanism offers substantial advantages over traditional cross-coupling reactions. Without palladium or copper catalysts, there is no risk of forming metal-complexed impurities or experiencing catalyst-induced decomposition of sensitive functional groups on the substrate. The primary by-products are derived from the reduction of the nitro group and the oxidation of the hydrogen atoms, which are generally small molecules that are easily removed during the aqueous workup and subsequent column chromatography. This inherent cleanliness of the reaction profile ensures that the resulting high-purity N-aryl indoline derivatives meet the rigorous specifications required for downstream drug synthesis. For process chemists, understanding this mechanism allows for fine-tuning of base equivalents and oxygen flow rates to maximize conversion while minimizing the formation of regioisomers or over-arylated side products, ensuring a robust and reproducible manufacturing process.
How to Synthesize N-Aryl Indoline Derivatives Efficiently
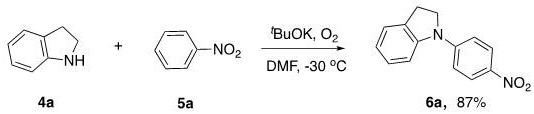
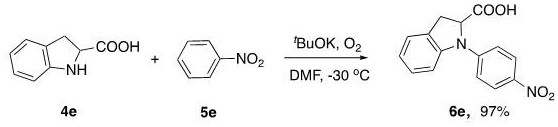
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to replicate the high efficiencies reported in the patent literature. The process begins with the dissolution of the indoline substrate and the nitrobenzene coupling partner in a polar aprotic solvent such as DMF or DMSO, which stabilizes the charged intermediates formed during the base promotion phase. The addition of the base, typically t-BuOK, must be controlled to manage the exotherm, and the reaction mixture is subsequently cooled to the optimal range of -50°C to 0°C. Oxygen is then introduced into the headspace of the reactor, and the mixture is stirred for an extended period, approximately 17 hours, to ensure complete consumption of the starting materials. Detailed standardized synthesis steps for specific substrates are provided in the guide below to assist technical teams in replicating these results.
- Dissolve the indoline derivative and nitrobenzene compound in an organic solvent such as DMF or DMSO under an inert atmosphere initially.
- Add a strong base promoter like t-BuOK or t-BuONa to the mixture while maintaining the temperature between -50°C and 0°C.
- Introduce oxygen gas into the reaction system and stir for approximately 17 hours, followed by aqueous quenching and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free CDC technology translates into tangible strategic benefits that extend beyond simple reaction yields. The elimination of precious metal catalysts removes a significant variable cost from the bill of materials, as palladium and specialized ligands are subject to volatile market pricing and supply constraints. Furthermore, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and shortens the overall production cycle time. This efficiency gain allows for faster turnover of manufacturing suites and improved responsiveness to market demand fluctuations. The use of commodity chemicals like nitrobenzene and common bases ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized reagents. Consequently, this process supports cost reduction in API intermediate manufacturing by lowering both direct material costs and indirect operational overheads.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete avoidance of expensive transition metal catalysts and ligands, which traditionally account for a significant portion of raw material costs in C-N bond formation. By substituting these with inexpensive alkali bases and using oxygen from the air as the oxidant, the direct material cost is substantially lowered. Additionally, the reduction in waste treatment costs associated with heavy metal disposal further enhances the economic viability of this route. The high atom economy means less raw material is wasted as by-products, maximizing the value derived from every kilogram of input reagent and contributing to significant overall cost savings for the manufacturing operation.
- Enhanced Supply Chain Reliability: Relying on widely available commodity chemicals such as nitrobenzene derivatives and potassium tert-butoxide ensures a robust supply chain that is less susceptible to geopolitical disruptions or supplier bottlenecks. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this process can be sourced from multiple global suppliers, enhancing procurement flexibility. This reliability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream pharmaceutical clients. The simplified logistics of handling non-hazardous bases compared to air-sensitive metal catalysts also reduce storage and transportation complexities, further securing the supply chain against potential interruptions.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic heavy metals make this process inherently safer and easier to scale from gram-level laboratory experiments to multi-ton commercial production. The patent data confirms successful gram-scale synthesis with excellent yields, demonstrating the feasibility of commercial scale-up of complex pharmaceutical intermediates. From an environmental standpoint, the generation of benign by-products aligns with increasingly strict global regulations on industrial emissions and waste discharge. This compliance reduces the regulatory burden and potential liability for the manufacturer, facilitating smoother approvals for new process validations and supporting the company's sustainability goals without compromising on production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this N-aryl indoline synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios. The responses cover aspects ranging from catalyst requirements to scalability potential, ensuring a comprehensive overview of the technology's capabilities and limitations for potential adopters.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN111732528A utilizes a metal-free system promoted by bases like t-BuOK, eliminating the need for costly palladium or copper catalysts and avoiding heavy metal contamination.
Q: What are the typical reaction conditions for this CDC coupling?
A: The reaction typically proceeds in solvents like DMF at low temperatures ranging from -50°C to 0°C, specifically around -30°C, under an oxygen atmosphere for about 17 hours to ensure high conversion.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent demonstrates successful gram-scale synthesis with yields up to 97%, indicating strong potential for commercial scale-up due to the simplicity of reagents and mild operational conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl Indoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free coupling technologies in modernizing the production of critical pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN111732528A can be successfully translated into robust industrial processes. We are committed to delivering high-purity N-aryl indoline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex synthetic challenges allows us to offer customized solutions that optimize both yield and cost-efficiency for our global partners.
We invite you to collaborate with us to explore how this advanced synthesis route can enhance your supply chain resilience and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom projects. By partnering with us, you gain access to a reliable pharmaceutical intermediates supplier dedicated to driving innovation and efficiency in the fine chemical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
