Scalable Electrochemical Synthesis of Aryl Selenocyanides from Arylboronic Acids for Industrial Applications
Scalable Electrochemical Synthesis of Aryl Selenocyanides from Arylboronic Acids for Industrial Applications
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, safer, and more cost-effective synthetic methodologies. A pivotal advancement in this domain is documented in Chinese Patent CN110724080B, which discloses a novel electrochemical approach for the synthesis of aryl selenocyanides. This technology represents a paradigm shift from traditional stoichiometric oxidants to the use of electrons as clean reagents, enabling the efficient conversion of readily available arylboronic acids into valuable aryl selenocyanide derivatives. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this innovation offers a compelling solution to long-standing challenges regarding reagent stability, toxicity, and process scalability. By leveraging electricity as the primary driving force, the method operates under mild conditions—specifically at room temperature and under an air atmosphere—thereby drastically reducing energy consumption and eliminating the need for inert gas protection systems that typically inflate operational expenditures in standard organometallic couplings.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
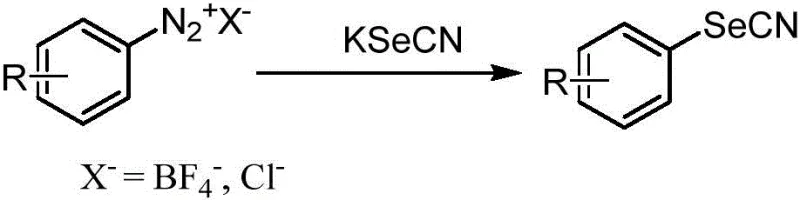
Historically, the introduction of the selenocyano group (-SeCN) onto an aromatic ring has been fraught with significant chemical and logistical hurdles that impede efficient large-scale production. Traditional synthetic routes often rely on the generation of aryl diazonium salts as key intermediates, a strategy that introduces severe safety liabilities due to the inherent thermal instability and potential explosivity of diazonium species, necessitating rigorous temperature control and specialized handling equipment. Furthermore, alternative pathways utilizing aryl hypervalent iodine salts or aryl selenyl chlorides suffer from distinct disadvantages; hypervalent iodine reagents are often expensive, difficult to synthesize in bulk, and generate substantial stoichiometric waste, while aryl selenyl chlorides are notoriously toxic and unstable, posing serious health risks to personnel and complicating waste disposal protocols. These legacy methods frequently require harsh reaction conditions, sensitive anhydrous environments, or expensive transition metal catalysts that leave behind trace metal impurities, creating a bottleneck for the production of high-purity pharmaceutical intermediates that must meet stringent regulatory standards for residual metals.
The Novel Approach
In stark contrast to these hazardous and inefficient legacy processes, the electrochemical methodology described in the patent utilizes stable, commercially abundant arylboronic acids as the starting material, coupled with potassium selenocyanate (KSeCN) as the selenium source. This approach fundamentally alters the reaction profile by replacing chemical oxidants with a controlled electric current, which acts as a green and tunable driving agent to facilitate the substitution reaction. The process is remarkably robust, tolerating a wide array of functional groups on the aromatic ring—including electron-donating groups like methoxy and amino, as well as electron-withdrawing groups like aldehydes—without the need for complex protecting group strategies. By conducting the reaction in a simple organic solvent system under ambient air and room temperature conditions, the method eliminates the capital expenditure associated with high-pressure reactors or cryogenic cooling systems, offering a streamlined pathway that is inherently safer and more environmentally benign than its predecessors.
Mechanistic Insights into Electrochemical Selenocyanation
The core of this technological breakthrough lies in the anodic oxidation process that activates the selenium species or the boron-carbon bond to enable coupling without external chemical oxidants. In this electrochemical cell, the application of a constant current generates reactive intermediates at the electrode surface, effectively driving the substitution of the boronic acid moiety with the selenocyanate group. The use of a supporting electrolyte, such as tetrabutylammonium acetate or sodium acetate, ensures sufficient conductivity within the organic solvent medium, typically acetonitrile, allowing for efficient electron transfer. This mechanism avoids the formation of toxic byproducts associated with stoichiometric oxidants like peroxides or heavy metal salts, resulting in a cleaner reaction profile that simplifies downstream processing. The ability to fine-tune the current intensity (experimentally demonstrated between 10 mA and 15 mA for laboratory scale) provides precise control over the reaction kinetics, minimizing side reactions such as homocoupling of the boronic acid or over-oxidation of the sensitive selenocyanate functionality.
From a purity and impurity profile perspective, this electrochemical route offers distinct advantages for the synthesis of high-purity aryl selenocyanides intended for biological applications. The absence of transition metal catalysts means that the final product is free from palladium, copper, or nickel residues, which are common contaminants in cross-coupling reactions and require expensive scavenging steps to remove to parts-per-million levels. Furthermore, the mild reaction conditions preserve sensitive functional groups that might otherwise degrade under the acidic or basic conditions required for diazonium salt formation or hydrolysis. The patent data indicates that the reaction proceeds efficiently within a short timeframe, typically completing within 5 hours, which reduces the residence time of reactive intermediates and limits the opportunity for decomposition pathways to occur. This results in a product stream with a simplified impurity profile, facilitating easier purification via standard column chromatography or crystallization techniques, ultimately yielding materials that meet the rigorous quality specifications demanded by the fine chemical and pharmaceutical industries.
How to Synthesize Aryl Selenocyanides Efficiently
The practical implementation of this electrochemical synthesis is designed to be straightforward and adaptable to both laboratory discovery and industrial manufacturing environments. The protocol involves charging a reaction vessel with the arylboronic acid substrate, potassium selenocyanate, and a suitable electrolyte in an organic solvent, followed by the application of a constant current under ambient conditions. This simplicity belies the sophisticated control it offers over the reaction outcome, allowing chemists to access a diverse library of selenocyanated aromatics with high efficiency. For detailed operational parameters and specific workup procedures tailored to different substrates, please refer to the standardized synthesis guide below which outlines the critical steps for reproducibility and scale-up.
- Combine arylboronic acid, potassium selenocyanate (KSeCN), and a supporting electrolyte in an organic solvent such as acetonitrile within a reaction vessel equipped with stirring.
- Apply a constant electric current (typically 10-15 mA) to the reaction mixture under air atmosphere at room temperature for 3 to 5 hours.
- Upon completion, filter the mixture, concentrate via rotary evaporation, and purify the crude product using column chromatography to isolate the pure aryl selenocyanide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology translates into tangible strategic benefits regarding cost structure, supply security, and regulatory compliance. The shift away from exotic or unstable reagents towards commodity chemicals like arylboronic acids and potassium selenocyanate significantly de-risks the supply chain, as these materials are produced globally in massive quantities and are subject to less price volatility than specialized hypervalent iodine reagents or custom-synthesized diazonium precursors. Moreover, the elimination of expensive transition metal catalysts removes a major cost center from the bill of materials, while simultaneously reducing the environmental burden associated with the disposal of heavy metal waste, which often incurs high fees and regulatory scrutiny. The operational simplicity of running reactions at room temperature and pressure further contributes to cost reduction by lowering energy consumption and allowing the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic infrastructure.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the use of electricity as the primary reagent, which is significantly cheaper on a molar basis compared to stoichiometric chemical oxidants. By removing the requirement for palladium or other precious metal catalysts, the process avoids the substantial costs associated with catalyst recovery and the rigorous analytical testing required to certify low residual metal levels in the final API intermediate. Additionally, the high atom economy of substituting a boronic acid directly with a selenocyanate group minimizes waste generation, leading to lower raw material consumption per kilogram of product and reducing the overall cost of goods sold (COGS) for the final active ingredient.
- Enhanced Supply Chain Reliability: Relying on arylboronic acids as the starting material ensures a robust and continuous supply chain, as these compounds are staple building blocks in the pharmaceutical industry with well-established global manufacturing networks. Unlike diazonium salts which must often be generated in situ due to instability, arylboronic acids are shelf-stable solids that can be stocked in bulk inventory without degradation, providing a buffer against supply disruptions. This stability also simplifies logistics and transportation, as the raw materials do not require hazardous material classification or temperature-controlled shipping, thereby streamlining the inbound logistics process and reducing lead times for production scheduling.
- Scalability and Environmental Compliance: Electrochemical synthesis is inherently scalable, moving seamlessly from milligram-scale discovery to multi-ton commercial production through the use of flow electrochemical cells or scaled-up batch reactors with optimized electrode surface areas. The green nature of this process, characterized by the absence of toxic oxidants and heavy metals, aligns perfectly with modern environmental, social, and governance (ESG) goals, facilitating easier permitting and regulatory approval in jurisdictions with strict environmental laws. The minimal waste stream and the use of benign reagents reduce the load on wastewater treatment facilities and lower the carbon footprint of the manufacturing process, positioning the supply chain as a sustainable partner for eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common inquiries regarding the operational specifics and compatibility of this electrochemical method. These insights are derived directly from the experimental data and technical disclosures within the patent literature, ensuring that the information provided is grounded in verified scientific results rather than theoretical speculation. Understanding these nuances is critical for process chemists aiming to adapt this methodology for the synthesis of complex drug candidates or advanced material precursors.
Q: What are the primary safety advantages of this electrochemical method over traditional diazonium routes?
A: Traditional methods often utilize unstable aryl diazonium salts which pose explosion risks and require strict temperature control. This electrochemical protocol operates at room temperature using stable arylboronic acids, significantly enhancing operational safety.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method utilizes electric current as the driving force for the reaction, eliminating the need for costly palladium or other transition metal catalysts, which simplifies downstream purification and reduces heavy metal contamination risks.
Q: What is the functional group tolerance of this electrochemical selenocyanation?
A: The process exhibits excellent tolerance for various functional groups including methoxy, hydroxyl, amino, and aldehyde groups, allowing for the direct synthesis of complex intermediates without extensive protecting group strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Selenocyanide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value intermediates with superior purity and sustainability profiles. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and effectively, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting demands of the global pharmaceutical market. We are committed to leveraging innovations like the one described in CN110724080B to provide our clients with a competitive edge through cost-effective and environmentally responsible manufacturing solutions.
We invite you to engage with our technical procurement team to discuss how this advanced electrochemical route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener methodology for your aryl selenocyanide needs. We encourage you to contact us today to obtain specific COA data for our reference standards and to receive comprehensive route feasibility assessments that will accelerate your development timelines and secure your supply chain for the future.
