Scalable Synthesis of 2-(2-Aminobenzoyl)Benzoic Acid Derivatives for Commercial API Production
Scalable Synthesis of 2-(2-Aminobenzoyl)Benzoic Acid Derivatives for Commercial API Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign pathways for constructing complex nitrogen-containing scaffolds. A significant breakthrough in this domain is detailed in patent CN109651179B, which discloses a novel preparation method for 2-(2-aminobenzoyl)benzoic acid derivatives. These compounds are not merely academic curiosities; they serve as critical construction units in organic synthesis, potential tridentate ligands for transition metal catalysis, and bioactive cores exhibiting anti-hyperlipidemia and antioxidant properties. The disclosed technology represents a paradigm shift from hazardous azide chemistry to a green, base-mediated aerobic oxidation process. By leveraging ambient air as the oxygen source and employing readily available alkaline reagents, this methodology addresses long-standing challenges in regioselectivity and atom economy. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with a reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(2-aminobenzoyl)benzoic acid compounds has been fraught with significant chemical and operational hurdles that impede efficient commercial scale-up. The traditional route predominantly relies on the hydrolysis of dibenzo[b,e]azacyclo-6,11-dione intermediates. Obtaining these precursors typically necessitates a Schmidt rearrangement reaction involving anthraquinone compounds and sodium azide. This classical approach is inherently problematic due to the use of sodium azide, a highly toxic and potentially explosive reagent that imposes severe safety constraints on manufacturing facilities. Furthermore, the Schmidt rearrangement is notorious for its poor regioselectivity, often generating mixtures of isomers that are difficult and costly to separate. This lack of specificity leads to substantial yield losses during purification and complicates the impurity profile of the final active pharmaceutical ingredient (API). Consequently, the reliance on such hazardous and inefficient chemistry has created a bottleneck for the cost reduction in API manufacturing, driving the urgent need for alternative synthetic strategies that prioritize both safety and precision.
The Novel Approach
In stark contrast to the hazardous legacy methods, the technology described in CN109651179B introduces a streamlined and highly specific pathway utilizing N-substituted halo-benzamides as starting materials. The core innovation lies in the reaction of these precursors in a solvent system containing alkali and water, critically under an atmosphere of air. This simple yet profound modification allows molecular oxygen to act as the terminal oxidant, driving the transformation to the desired 2-(2-aminobenzoyl)benzoic acid derivative with remarkable efficiency. 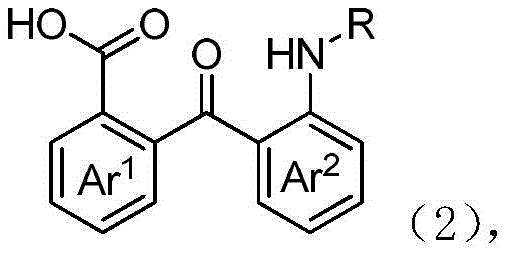 The process operates under relatively mild conditions, typically between 80°C and 140°C, and demonstrates exceptional tolerance for a wide range of functional groups including halogens and alkyl substituents. By eliminating the need for heavy metal catalysts or dangerous azide reagents, this novel approach not only simplifies the post-treatment workflow but also drastically reduces the generation of hazardous waste. The strong reaction specificity ensures that the desired regioisomer is produced predominantly, thereby enhancing the overall purity of the crude product and minimizing the burden on downstream purification processes. This represents a substantial advancement for any organization aiming for the commercial scale-up of complex pharmaceutical intermediates.
The process operates under relatively mild conditions, typically between 80°C and 140°C, and demonstrates exceptional tolerance for a wide range of functional groups including halogens and alkyl substituents. By eliminating the need for heavy metal catalysts or dangerous azide reagents, this novel approach not only simplifies the post-treatment workflow but also drastically reduces the generation of hazardous waste. The strong reaction specificity ensures that the desired regioisomer is produced predominantly, thereby enhancing the overall purity of the crude product and minimizing the burden on downstream purification processes. This represents a substantial advancement for any organization aiming for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Base-Mediated Aerobic Oxidation
The mechanistic underpinning of this transformation involves a sophisticated interplay between nucleophilic attack and oxidative cleavage facilitated by the basic environment. Initially, the strong base, such as potassium hydroxide or sodium tert-butoxide, activates the water molecules present in the system or coordinates with the carbonyl oxygen of the amide substrate. This activation enhances the electrophilicity of the carbonyl carbon, rendering it susceptible to nucleophilic attack by hydroxide ions or water. Simultaneously, the presence of the ortho-halo substituent on the aromatic ring plays a pivotal role, potentially participating in an intramolecular cyclization or acting as a leaving group during the oxidative degradation of the benzyl-amino moiety. The molecular oxygen from the air serves as the electron acceptor, regenerating the active catalytic species or directly participating in the oxidation of the intermediate imine or hemiaminal species to the final ketone and amine functionalities. This aerobic cycle is self-sustaining and requires no external stoichiometric oxidants, which is a key factor in the reaction's high atom economy.
From an impurity control perspective, the mechanism offers distinct advantages over acid-catalyzed or metal-mediated alternatives. The high regioselectivity observed is attributed to the specific orientation of the nucleophilic attack directed by the electronic effects of the substituents on the aromatic rings Ar1 and Ar2. Unlike the Schmidt rearrangement, which can proceed through multiple migration pathways leading to isomeric byproducts, this base-mediated pathway is constrained by the fixed position of the leaving group and the carbonyl functionality. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups such as esters or nitriles that might be present on the substrate, thereby preserving the integrity of the molecular scaffold. The absence of transition metals also means there is no risk of metal leaching into the final product, a critical quality attribute for pharmaceutical intermediates intended for human consumption. This inherent purity profile simplifies the analytical validation process and ensures compliance with stringent regulatory standards for residual impurities.
How to Synthesize 2-(2-Aminobenzoyl)Benzoic Acid Derivatives Efficiently
Implementing this synthesis requires careful attention to the preparation of the starting material and the optimization of the oxidation conditions. The patent outlines a two-stage strategy where the N-substituted halo-benzamide precursor is first constructed, often via amidation of a corresponding benzoic acid with a substituted benzylamine. 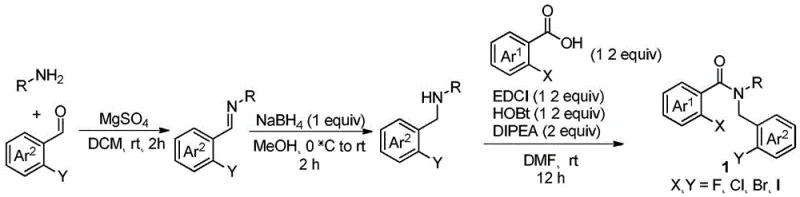 Once the precursor is secured, the key oxidative step is performed in polar aprotic solvents like dimethylsulfoxide (DMSO) or N,N-dimethylformamide (DMF), which stabilize the ionic intermediates. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below, ensuring reproducibility and safety at scale.
Once the precursor is secured, the key oxidative step is performed in polar aprotic solvents like dimethylsulfoxide (DMSO) or N,N-dimethylformamide (DMF), which stabilize the ionic intermediates. The detailed standardized synthesis steps for this high-efficiency route are provided in the guide below, ensuring reproducibility and safety at scale.
- Prepare the N-substituted halo-benzamide precursor (Formula 1) via condensation and reduction or amidation reactions.
- React the precursor with a strong base (e.g., KOH, NaOtBu) in a polar aprotic solvent like DMSO or DMF.
- Maintain the reaction at 80-140°C under air atmosphere for 1-12 hours to facilitate oxidative hydrolysis.
- Quench with water, adjust pH to acidity, and purify the resulting 2-(2-aminobenzoyl)benzoic acid derivative via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible operational benefits that extend beyond mere chemical yield. The shift away from sodium azide and heavy metal catalysts fundamentally alters the risk profile of the manufacturing process, reducing the need for specialized containment equipment and hazardous waste disposal protocols. This simplification of the safety infrastructure allows for more flexible production scheduling and lowers the barrier to entry for contract manufacturing organizations. Moreover, the use of air as a reagent eliminates the supply chain volatility associated with purchasing expensive or regulated oxidizing agents. The robustness of the reaction across a wide substrate scope means that a single production line can be adapted to manufacture a variety of derivatives simply by changing the starting amine or acid components, thereby maximizing asset utilization and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants and expensive transition metal catalysts results in a direct reduction in raw material costs. Additionally, the simplified workup procedure, which involves basic aqueous quenching and acidification, avoids the complex extraction and chelation steps required to remove metal residues. This streamlining of the downstream processing significantly lowers the operational expenditure per kilogram of product. The high regioselectivity further contributes to cost efficiency by minimizing the loss of material during chromatographic purification, ensuring that a greater proportion of the input reactants are converted into saleable product. These factors combined create a compelling economic case for switching to this greener methodology.
- Enhanced Supply Chain Reliability: The reagents required for this process, including common bases like KOH and solvents like DMSO, are commodity chemicals with stable global supply chains. This contrasts sharply with the supply risks associated with specialized azide reagents or proprietary ligand-catalyst systems. By relying on widely available inputs, manufacturers can mitigate the risk of production stoppages due to raw material shortages. Furthermore, the reaction's tolerance to moisture and air implies that it does not require rigorous anhydrous or inert atmosphere conditions, reducing the complexity of the reactor setup and allowing for faster batch turnover times. This reliability is crucial for maintaining continuous supply to downstream API producers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from milligram to multi-gram scales in the patent examples with consistent yields. The use of air oxidation aligns perfectly with modern green chemistry principles, significantly reducing the E-factor (mass of waste per mass of product) of the synthesis. This environmental advantage facilitates easier regulatory approval and supports corporate sustainability goals. The absence of heavy metals simplifies the wastewater treatment process, as there is no need for specialized heavy metal precipitation or recovery units. Consequently, this method offers a sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates that meets increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is vital for assessing the feasibility of integrating this route into existing manufacturing portfolios. The responses cover aspects of reaction specificity, reagent sourcing, and product quality assurance.
Q: How does this new method improve upon traditional Schmidt rearrangement?
A: Traditional methods using anthraquinone and sodium azide suffer from poor regioselectivity. This novel base-mediated aerobic oxidation offers strong reaction specificity, ensuring the correct isomer is formed without complex separation steps.
Q: What is the oxygen source for this oxidation reaction?
A: The process utilizes molecular oxygen from ambient air as the sole oxidant. This eliminates the need for expensive or hazardous stoichiometric oxidants, significantly enhancing the green chemistry profile and safety of the manufacturing process.
Q: What is the substrate scope for this synthesis?
A: The method demonstrates broad compatibility with various substituents including fluorine, chlorine, bromine, and iodine on the aromatic rings. It also tolerates alkyl groups and heteroaryl systems like pyridyl, making it versatile for diverse API intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Aminobenzoyl)Benzoic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN109651179B for the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying the stringent purity specifications required for advanced drug synthesis. We are committed to leveraging this green chemistry innovation to deliver products that not only meet but exceed the quality expectations of our global clientele, providing a secure and sustainable source for your critical supply chain needs.
We invite you to engage with our technical team to explore how this methodology can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this air-oxidation route for your specific targets. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of chemical excellence and supply chain resilience.
