Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Pharmaceutical Intermediates
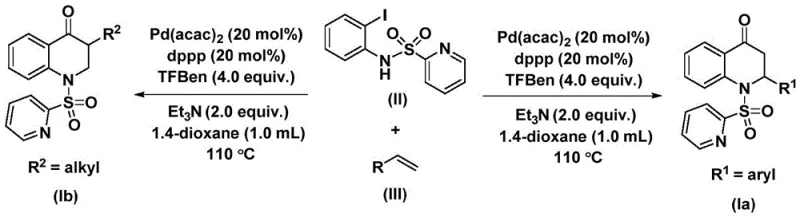
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly the 2,3-dihydroquinolone scaffold, which serves as a privileged structure in medicinal chemistry. As disclosed in patent CN112239456B, a novel preparation method has been developed that leverages transition metal palladium catalysis to efficiently synthesize these valuable compounds. This technology addresses critical bottlenecks in traditional synthesis by utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as starting materials in a carbonylation reaction. The significance of this scaffold is underscored by its presence in bioactive molecules; for instance, specific derivatives have demonstrated potent antitumor activity against human cells and significant analgesic properties, highlighting the urgent demand for reliable production methods.
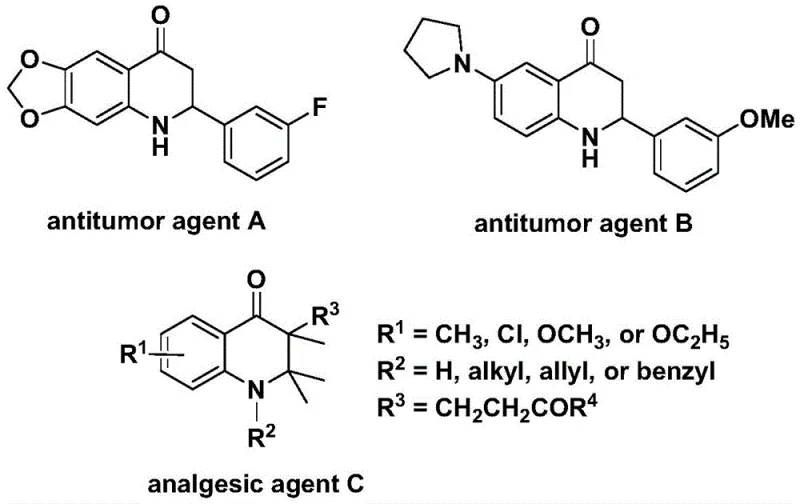
For procurement managers and supply chain directors, the shift towards this methodology represents a strategic opportunity for cost reduction in pharmaceutical intermediate manufacturing. The process utilizes readily available starting materials and operates under relatively mild conditions compared to harsh traditional protocols. By replacing gaseous carbon monoxide with a solid surrogate, the method drastically simplifies the engineering controls required for production, reducing both capital expenditure on specialized equipment and operational risks. This transition from gas-phase to solution-phase carbonylation is a pivotal advancement for any organization aiming to secure a stable supply of high-purity intermediates while maintaining rigorous safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone core has relied on multi-step sequences involving cyclization reactions that often suffer from poor atom economy and limited functional group tolerance. Traditional carbonylation approaches frequently necessitate the use of high-pressure carbon monoxide gas, which poses severe safety hazards and requires specialized high-pressure reactors that are expensive to maintain and operate. Furthermore, conventional methods often struggle with regioselectivity issues, leading to complex mixtures of isomers that are difficult and costly to separate. The reliance on stoichiometric amounts of toxic reagents and the generation of substantial waste streams further exacerbate the environmental footprint, making these legacy processes increasingly untenable in the context of modern green chemistry mandates and regulatory compliance.
The Novel Approach
The innovative strategy outlined in the patent data introduces a streamlined catalytic cycle that overcomes these historical barriers through the use of a palladium catalyst system paired with a solid carbon monoxide source. This approach allows for the direct insertion of a carbonyl group into the carbon-nitrogen bond framework under atmospheric pressure conditions, effectively bypassing the need for dangerous CO gas infrastructure. The reaction demonstrates exceptional versatility, accommodating a wide array of olefin substrates ranging from simple alkenes to complex aryl-substituted variants. By employing 1,3,5-trimesic acid phenol ester as the CO surrogate, the process ensures a controlled release of carbon monoxide in situ, which enhances reaction efficiency and selectivity. This methodological shift not only improves the overall yield but also significantly simplifies the post-reaction workup, making it highly attractive for commercial scale-up.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this synthetic breakthrough lies in the intricate catalytic cycle mediated by the palladium complex. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide, released thermally from the 1,3,5-trimesic acid phenol ester additive, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate is the key precursor to the final carbonyl functionality found in the quinolone ring. The precision of this insertion step is vital for minimizing side products and ensuring the structural integrity of the final pharmacophore.
Subsequent coordination and insertion of the olefin substrate into the acyl-palladium bond generates a new alkyl-palladium intermediate, setting the stage for ring closure. The final step involves a reductive elimination that releases the substituted 2,3-dihydroquinolone product and regenerates the active palladium catalyst, allowing the cycle to continue. This mechanism is highly efficient because the ligand system, specifically 1,3-bis(diphenylphosphino)propane (dppp), stabilizes the palladium center throughout these transitions, preventing catalyst deactivation. From an impurity control perspective, this well-defined pathway minimizes the formation of homocoupling byproducts often seen in less optimized cross-coupling reactions. The result is a cleaner reaction profile that facilitates easier purification, directly impacting the cost of goods sold by reducing solvent usage and chromatography time.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
The practical implementation of this synthesis involves a straightforward procedure suitable for both laboratory optimization and pilot plant operations. The process begins by charging a reaction vessel with the necessary catalytic components, including bis(acetylacetone)palladium and the dppp ligand, along with the base triethylamine and the solid CO source. The specific molar ratios are critical, with the patent suggesting a ratio of 0.2:0.2:4 for the catalyst, ligand, and CO surrogate respectively relative to the substrate. Once the reagents are dissolved in an aprotic solvent such as 1,4-dioxane, the olefin substrate is introduced. The detailed standardized synthesis steps for optimizing this reaction are provided in the guide below.
- Combine N-pyridine sulfonyl-o-iodoaniline, olefin, Pd(acac)2 catalyst, dppp ligand, triethylamine, and 1,3,5-trimesic acid phenol ester in an organic solvent like dioxane.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target substituted 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders, the adoption of this technology offers tangible benefits regarding reliability and operational simplicity. The elimination of high-pressure gas handling removes a significant bottleneck in production scheduling and facility qualification. Moreover, the use of commercially available olefins and iodoanilines ensures a robust supply chain with multiple sourcing options, mitigating the risk of raw material shortages. The reaction conditions, operating between 100-120°C, are compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, requiring no exotic metallurgy or specialized high-pressure autoclaves. This compatibility translates directly into faster technology transfer times and lower capital investment thresholds for new production lines.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the reaction setup and the high efficiency of the catalyst system. By avoiding the need for specialized high-pressure CO equipment, facilities can utilize existing infrastructure, resulting in substantial capital cost avoidance. Additionally, the high conversion rates and clean reaction profiles reduce the volume of solvents and silica gel required for purification, leading to significant savings in consumables and waste disposal costs. The ability to run the reaction at atmospheric pressure also lowers energy consumption associated with compression and pressurization systems, contributing to a leaner manufacturing cost structure overall.
- Enhanced Supply Chain Reliability: The reliance on stable, solid reagents rather than compressed gases enhances the logistical stability of the supply chain. Solid CO surrogates are easier to transport, store, and handle than hazardous gas cylinders, reducing regulatory burdens and shipping delays. Furthermore, the broad substrate scope means that if a specific olefin becomes unavailable, alternative analogs can often be substituted without re-optimizing the entire catalytic system. This flexibility ensures continuous production capability even in the face of raw market volatility, providing a secure supply of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram levels is facilitated by the homogeneous nature of the catalytic system and the absence of gas-liquid mass transfer limitations typical of traditional carbonylations. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations. The use of dioxane, while requiring careful management, is a well-understood solvent in the industry with established recovery protocols. The overall atom economy of the transformation is superior to stepwise alternatives, reducing the total mass intensity of the process and supporting corporate sustainability goals through greener chemical manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, offering clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities. Understanding these nuances is essential for accurate process modeling and risk assessment.
Q: What is the advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: This compound acts as a solid carbon monoxide surrogate, eliminating the need for handling hazardous CO gas cylinders, thereby significantly enhancing operational safety and simplifying reactor setup for industrial scale-up.
Q: What types of substrates are compatible with this palladium-catalyzed method?
A: The method exhibits broad substrate compatibility, successfully tolerating various functional groups including methyl, tert-butyl, methoxy, fluorine, and chlorine on the aryl ring, as well as linear, branched alkyl, and silyl groups on the olefin.
Q: How does this method improve impurity control compared to traditional routes?
A: By utilizing a direct carbonylation insertion mechanism with a specific ligand system (dppp), the reaction minimizes side reactions such as homocoupling, resulting in a cleaner crude profile that simplifies downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this palladium-catalyzed carbonylation can be seamlessly transitioned to industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling sensitive catalytic systems allows us to optimize yield and minimize impurities, providing our partners with a competitive edge in the global marketplace.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Whether you require custom synthesis of novel analogs or large-scale production of established intermediates, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality.
