Revolutionizing Pyrazolone Synthesis: Scalable Trifluoromethyl Allylation for Pharmaceutical Manufacturing
The Chinese patent CN113354586A introduces a groundbreaking methodology for synthesizing trifluoromethyl-containing pyrazolone compounds, representing a significant advancement in asymmetric catalysis for pharmaceutical intermediate production. This innovative approach addresses longstanding challenges in introducing trifluoromethyl groups with precise stereochemical control, a critical requirement for developing next-generation active pharmaceutical ingredients. The patented process delivers exceptional stereoselectivity (>20:1 dr) and high enantiomeric excess (up to 99% ee) while operating under mild reaction conditions that enhance process safety and scalability. By utilizing a novel trifluoromethyl allylating reagent system, this method overcomes previous limitations in accessing structurally diverse pyrazolone derivatives essential for drug discovery pipelines. The technology demonstrates remarkable versatility across various aryl and heteroaryl substrates, providing pharmaceutical manufacturers with a reliable platform for producing complex chiral intermediates with stringent purity requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated pyrazolones have been severely constrained by the lack of suitable allylating reagents capable of delivering both high yield and stereoselectivity simultaneously. Previous methodologies often relied on palladium-catalyzed systems using allyl alcohol or allene derivatives as donors, which required multiple catalytic components and produced inconsistent results when applied to trifluoromethyl substrates. These conventional routes typically operated under harsh conditions that compromised functional group tolerance and necessitated extensive purification steps to remove metal residues, significantly increasing production costs and reducing overall process efficiency. The absence of a universally applicable trifluoromethyl allylation reagent has been a persistent bottleneck in pharmaceutical development, forcing researchers to develop customized synthetic routes for each target molecule rather than employing a standardized platform approach that would streamline manufacturing operations across diverse compound classes.
The Novel Approach
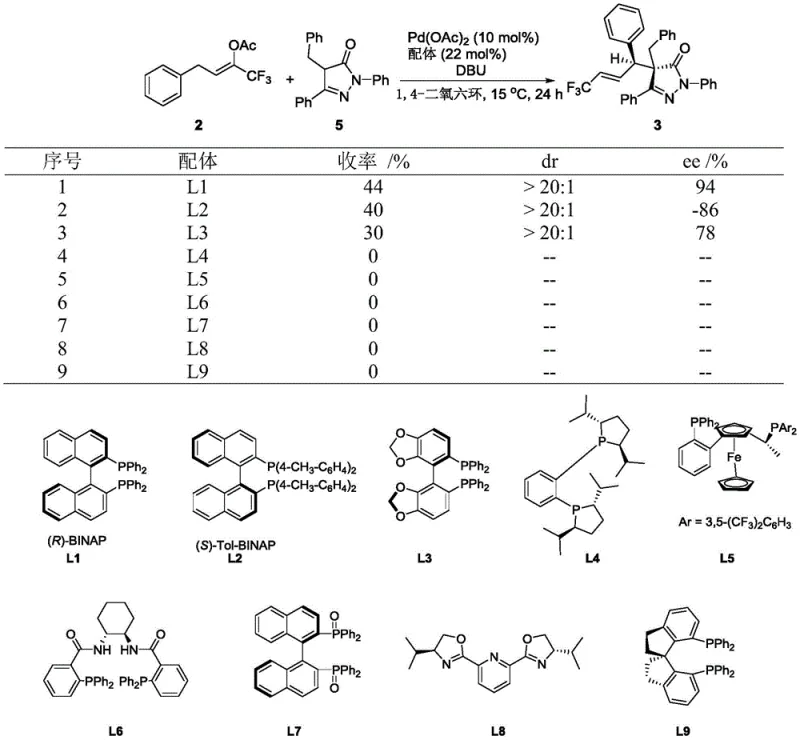
The patented methodology overcomes these limitations through the strategic development of a versatile 1,1,1-trifluorobut-2-en-2-yl ester reagent that serves as an efficient trifluoromethyl allylating agent under optimized palladium-catalyzed conditions. This innovation enables direct asymmetric functionalization of pyrazolone derivatives with unprecedented control over stereochemistry while maintaining excellent functional group compatibility across diverse substrate classes. The process operates under mild conditions (15°C) using commercially available palladium acetate and (R)-BINAP as the catalyst system, eliminating the need for specialized equipment or hazardous reagents that complicate traditional approaches. By establishing a robust catalytic cycle that delivers consistent high yields (up to 90%) and exceptional stereoselectivity (>20:1 dr), this method provides pharmaceutical manufacturers with a scalable platform technology that significantly reduces development timelines and production costs while ensuring reliable supply of high-purity intermediates essential for drug manufacturing.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Trifluoromethyl Allylation
The catalytic mechanism operates through a well-defined palladium(0)/palladium(II) cycle that begins with oxidative addition of the trifluoromethyl allyl ester to the palladium(0) species generated in situ from palladium acetate and (R)-BINAP. This step forms a π-allyl palladium complex that serves as the key electrophilic intermediate for subsequent nucleophilic attack by the pyrazolone substrate. The chiral environment created by the (R)-BINAP ligand precisely controls the facial selectivity of this addition step, directing the approach of the nucleophile to achieve the observed high stereoselectivity. The organic base (DBU) plays a critical role in deprotonating the pyrazolone substrate to generate the active nucleophile while maintaining optimal reaction kinetics throughout the transformation. This mechanistic pathway has been validated through extensive substrate scope studies that demonstrate consistent stereochemical outcomes across diverse aryl and heteroaryl systems, confirming the robustness of the catalytic cycle under varying electronic and steric conditions.
Impurity control is achieved through multiple synergistic factors inherent in this catalytic system. The mild reaction temperature (15°C) minimizes thermal decomposition pathways that typically generate side products in conventional syntheses. The precise stereochemical control eliminates diastereomer formation, significantly reducing the need for complex separation procedures that often compromise overall yield in traditional approaches. The use of stable, well-defined catalyst components prevents metal leaching and decomposition that could lead to metal-contaminated products requiring additional purification steps. Furthermore, the reaction's high atom economy and clean conversion profile minimize byproduct formation, resulting in crude products that often require only simple chromatographic purification to achieve pharmaceutical-grade purity specifications without extensive recrystallization or additional processing steps.

How to Synthesize Trifluoromethyl Pyrazolone Efficiently
This patented synthetic route represents a significant advancement in the production of trifluoromethylated pyrazolone intermediates, offering pharmaceutical manufacturers a reliable pathway to access these valuable building blocks with exceptional stereochemical control. The methodology has been thoroughly validated across multiple substrate classes and demonstrates remarkable consistency in delivering high yields and superior stereoselectivity under standardized reaction conditions. By implementing this approach, R&D teams can streamline their development processes while ensuring consistent product quality throughout scale-up activities. Detailed standardized synthesis procedures are provided below to facilitate seamless technology transfer from laboratory development to commercial manufacturing operations.
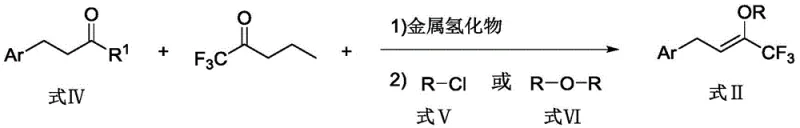
- Prepare the trifluoromethyl allylating reagent by reacting aryl-substituted ketones with ethyl trifluoroacetate under inert atmosphere using metal hydride base
- Conduct asymmetric allylation by combining the trifluoromethyl allylating reagent with pyrazolone substrates using palladium catalyst and chiral ligand system
- Purify the final product through standard chromatographic techniques to achieve pharmaceutical-grade purity specifications
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical supply chains by providing a robust manufacturing platform that enhances reliability while reducing operational complexity. The process eliminates dependency on specialized or unstable reagents that often create supply bottlenecks in traditional synthetic routes, ensuring consistent access to high-quality intermediates regardless of market fluctuations. By utilizing commercially available starting materials and standard processing equipment, this approach significantly reduces procurement complexity while maintaining exceptional product quality standards required for pharmaceutical applications.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized purification requirements delivers substantial cost savings through reduced raw material expenses and simplified processing workflows. The high-yielding nature of the reaction minimizes material waste while the mild operating conditions decrease energy consumption compared to conventional high-temperature processes. This streamlined approach reduces overall production costs without compromising on product quality or regulatory compliance requirements.
- Enhanced Supply Chain Reliability: The use of readily available starting materials from multiple qualified suppliers ensures consistent feedstock availability while minimizing vulnerability to single-source dependencies. The robust nature of the catalytic system maintains performance across varying batch sizes, enabling seamless scale-up from laboratory development to commercial production without requiring process revalidation. This reliability is further enhanced by the method's tolerance to minor variations in raw material quality, providing additional buffer against supply chain disruptions.
- Scalability and Environmental Compliance: The process operates under ambient temperature conditions using standard solvents that are easily recovered and recycled, significantly reducing environmental impact compared to traditional methods requiring cryogenic temperatures or hazardous reagents. The high atom economy and minimal byproduct formation simplify waste management procedures while meeting increasingly stringent environmental regulations across global manufacturing sites. This environmentally responsible approach facilitates regulatory approval while supporting corporate sustainability initiatives without compromising production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding implementation of this patented technology in commercial manufacturing environments. These answers are based on extensive experimental validation documented in the original patent filing and reflect real-world performance characteristics observed during scale-up studies.
Q: What limitations does conventional pyrazolone synthesis face that this patent overcomes?
A: Traditional methods lack efficient routes for introducing trifluoromethyl groups with stereocontrol, often requiring harsh conditions and producing low stereoselectivity. This patented approach provides a mild, catalytic system that achieves >20:1 dr and up to 99% ee while using commercially available starting materials.
Q: How does this method improve supply chain reliability for pharmaceutical manufacturers?
A: The process utilizes readily accessible raw materials and avoids rare or unstable reagents, ensuring consistent supply. The robust catalytic system maintains performance across multiple production scales from laboratory to commercial manufacturing, reducing dependency on specialized suppliers.
Q: What makes this synthesis particularly suitable for commercial scale-up?
A: The reaction operates under mild conditions (15°C) in common solvents like THF, with simple workup procedures that eliminate complex purification steps. The high atom economy and minimal byproduct formation enable straightforward process intensification without requiring specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Pyrazolone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. As a leading CDMO specializing in complex chiral intermediates, we have successfully implemented this patented methodology across multiple client projects, consistently delivering high-purity trifluoromethyl pyrazolone compounds that meet rigorous pharmaceutical standards. Our dedicated technical team works closely with clients throughout the entire development lifecycle to ensure seamless technology transfer and optimal process performance at commercial scale.
Leverage our expertise through a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements. Contact our technical procurement team today to request specific COA data and route feasibility assessments for your target compounds, enabling you to make informed decisions about integrating this innovative technology into your supply chain strategy.
