Advanced Synthesis of 3,5-Di-tert-butyl-4-hydroxy-benzoic Acid for Industrial Scale-Up
Advanced Synthesis of 3,5-Di-tert-butyl-4-hydroxy-benzoic Acid for Industrial Scale-Up
The chemical industry is constantly seeking more efficient and environmentally benign pathways for producing critical intermediates, and patent CN102050729A presents a significant breakthrough in the synthesis of 3,5-di-tert-butyl-4-hydroxy-benzoic acid. This compound serves as a pivotal building block for a wide array of high-value applications, ranging from broad-spectrum oxidation inhibitors and UV light absorbers in plastics and rubber to specialized intermediates in pharmaceutical synthesis. The disclosed methodology leverages a direct carboxylation strategy that fundamentally alters the economic and operational landscape of producing this sterically hindered phenolic acid. By utilizing carbon dioxide as a C1 building block under controlled high-pressure conditions, the process circumvents the need for hazardous halogenating agents or expensive aldehyde precursors that have historically plagued this sector. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable antioxidant intermediate supplier capable of delivering consistent quality at competitive price points.
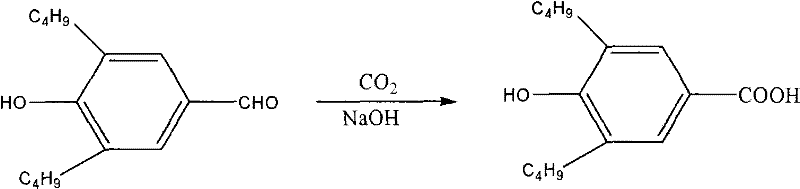
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the manufacturing of 3,5-di-tert-butyl-4-hydroxy-benzoic acid has been constrained by two primary synthetic routes, both of which suffer from significant drawbacks that impact cost reduction in polymer additive manufacturing. The first traditional method involves the oxidation of p-hydroxybenzaldehyde derivatives, a pathway often criticized for its reliance on rare and costly raw materials that create supply chain bottlenecks. Furthermore, this oxidative approach frequently results in suboptimal oxidation-hydrogenation ratios, leading to lower overall yields and a complex impurity profile that necessitates rigorous and expensive purification steps. The second conventional route employs a bromination-oxidation sequence starting from 2,6-di-tert-butyl-4-methylphenol, which introduces severe safety and environmental liabilities due to the handling of elemental bromine. The toxicity associated with bromine not only increases operational risks for plant personnel but also generates substantial hazardous waste streams that require sophisticated and costly treatment infrastructure, thereby eroding profit margins for producers.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a direct Kolbe-Schmitt type carboxylation of 2,6-di-tert-butylphenol, offering a streamlined and atom-economical solution. This innovative process initiates by forming the phenoxide salt in situ using sodium hydroxide and methanol, followed by a high-pressure reaction with carbon dioxide gas to install the carboxyl group directly onto the aromatic ring. The strategic use of elevated temperatures between 80-100°C and pressures ranging from 0.5-1.5 MPa drives the equilibrium towards the desired product, effectively overcoming the steric hindrance posed by the bulky tert-butyl groups. This methodology not only simplifies the operational workflow by eliminating multiple synthetic steps but also drastically improves the purity of the final isolate, reported to reach approximately 99%. For supply chain heads, this translates to a more robust production capability with reduced lead time for high-purity phenolic acids, ensuring continuity of supply for downstream customers in the coatings and agrochemical sectors.
Mechanistic Insights into CO2-Mediated Carboxylation
The core of this synthesis lies in the electrophilic aromatic substitution mechanism facilitated by the activation of the phenolic ring through deprotonation. When 2,6-di-tert-butylphenol is treated with sodium hydroxide in a methanol solvent system, the acidic proton of the hydroxyl group is abstracted to form the corresponding sodium phenoxide species, which is significantly more nucleophilic than the neutral phenol. This activation is crucial because the ortho positions of the ring are blocked by the bulky tert-butyl substituents, forcing the incoming electrophile to attack the para position exclusively. The introduction of carbon dioxide under pressure generates a reactive carbonate or carboxylate species that acts as the electrophile, attacking the electron-rich para-position of the phenoxide ring. The high-pressure environment is mechanistically vital as it increases the concentration of dissolved CO2 in the reaction medium, shifting the thermodynamic equilibrium towards the formation of the carboxylated intermediate despite the steric crowding around the reaction site.
Following the carboxylation event, the reaction mixture undergoes a carefully controlled workup procedure to ensure maximum recovery and purity of the target benzoic acid derivative. The transition from the high-pressure autoclave to the acidification stage involves quenching the reaction mass with dilute sulfuric acid, which protonates the carboxylate salt to liberate the free organic acid. The subsequent extraction using xylene (dimethylbenzene) exploits the differential solubility of the product versus inorganic salts, allowing for a clean separation of the organic phase. A critical dehydration step at 100-130°C under normal pressure removes residual water and solvent traces, preventing hydrolysis or degradation of the sensitive phenolic moiety during the final drying process. This meticulous control over the post-reaction processing is what enables the achievement of the reported 80% yield and 99% purity, demonstrating a deep understanding of impurity control mechanisms essential for commercial scale-up of complex fine chemicals.
How to Synthesize 3,5-Di-tert-butyl-4-hydroxy-benzoic Acid Efficiently
Implementing this synthesis route requires precise adherence to the molar ratios and thermal profiles outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the phenoxide solution, where maintaining a nitrogen atmosphere is critical to prevent premature oxidation of the phenolic starting material before the carboxylation step occurs. Operators must carefully monitor the vacuum distillation phase to ensure complete removal of methanol prior to pressurization, as residual solvent can affect the pressure dynamics and reaction kinetics inside the autoclave.
- Mix 2,6-di-tert-butylphenol and sodium hydroxide (molar ratio 1: 1 to 1:1.4) in methanol, reflux at 60-65°C while distilling off solvent under vacuum.
- Transfer the residue to a high-pressure autoclave, replace air with nitrogen, and introduce CO2 at 80-100°C under 0.5-1.5 MPa pressure for 20 hours.
- Acidify the reaction mass with 5% sulfuric acid, extract with xylene, dehydrate at 100-130°C, and dry to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis protocol offers transformative benefits for organizations aiming to optimize their sourcing strategies for antioxidant intermediates. By shifting away from bromine-based chemistry, manufacturers can eliminate the substantial costs associated with hazardous material handling, storage, and waste disposal, leading to significant cost savings in the overall production budget. The reliance on commodity chemicals such as phenol, sodium hydroxide, and carbon dioxide ensures that the raw material supply chain is resilient against market volatility, unlike routes dependent on specialized aldehydes or halogenating agents. This stability is paramount for procurement managers who need to secure long-term contracts without the fear of sudden price spikes caused by feedstock scarcity. Furthermore, the simplified operational flow reduces the requirement for specialized corrosion-resistant equipment often needed for bromine chemistry, lowering capital expenditure barriers for new production lines.
- Cost Reduction in Manufacturing: The elimination of toxic bromine and expensive oxidizing agents directly correlates to a reduction in variable production costs, as the process utilizes inexpensive and widely available carbon dioxide as the carbon source. The high yield reported in the patent minimizes raw material waste, ensuring that a greater proportion of input materials are converted into saleable product, which enhances overall process efficiency. Additionally, the simplified workup procedure reduces the consumption of solvents and energy required for extensive purification, contributing to a leaner manufacturing cost structure. These factors combined allow suppliers to offer more competitive pricing models while maintaining healthy margins, providing a distinct economic advantage in the global marketplace.
- Enhanced Supply Chain Reliability: Utilizing 2,6-di-tert-butylphenol as the primary feedstock leverages a well-established supply chain, as this material is produced in large volumes for various industrial applications. The robustness of the reaction conditions, which tolerate standard industrial autoclaves, means that production can be easily scaled across multiple facilities without requiring bespoke or hard-to-source reactor designs. This flexibility ensures that supply chain disruptions at a single site can be mitigated by shifting production to alternative locations, guaranteeing continuity of supply for critical downstream customers. The reduced dependency on niche reagents further insulates the supply chain from geopolitical or logistical shocks that often affect specialized chemical markets.
- Scalability and Environmental Compliance: The process is inherently greener, aligning with increasingly stringent global environmental regulations regarding volatile organic compounds and hazardous waste generation. By avoiding the creation of brominated byproducts, the facility significantly lowers its environmental footprint, simplifying the permitting process and reducing the risk of regulatory fines or shutdowns. The scalability of the high-pressure carboxylation step is well-understood in the chemical industry, allowing for seamless transition from pilot plant batches to multi-ton commercial production runs. This alignment with sustainability goals not only future-proofs the manufacturing asset but also appeals to end-users who are prioritizing eco-friendly sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthesis technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own formulations or production lines.
Q: What is the primary advantage of this synthesis method over traditional bromination?
A: This method eliminates the use of toxic elemental bromine and complex oxidation steps, significantly reducing environmental hazards and simplifying waste treatment protocols compared to conventional bromination-oxidation routes.
Q: What purity levels can be achieved with this process?
A: According to patent data, this optimized carboxylation process consistently achieves product purity levels of approximately 99%, meeting stringent specifications for high-performance polymer additives and pharmaceutical intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes standard industrial equipment such as high-pressure autoclaves and common commodity raw materials like phenol and CO2, making it highly scalable and economically viable for tonnage-level manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Di-tert-butyl-4-hydroxy-benzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of final products, which is why we have invested heavily in mastering advanced synthesis pathways like the one described in CN102050729A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the R&D phase or full-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 3,5-di-tert-butyl-4-hydroxy-benzoic acid meets the exacting standards required for pharmaceutical and high-performance polymer applications. Our commitment to technical excellence ensures that you receive a product with a consistent impurity profile, minimizing variability in your downstream processes.
We invite you to contact our technical procurement team to discuss how we can tailor our production capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our optimized supply chain can reduce your total landed costs. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to validate our quality and capacity before making any commitments. Let us be your partner in driving innovation and efficiency in your chemical supply chain.
