Innovative Vilsmeier Process for High-Purity Polysubstituted Pyridinones Scalable Manufacturing and Supply Chain Advantages
The present analysis examines Chinese Patent CN101121697A detailing an innovative synthetic route for polysubstituted pyridin-2(1H)-ketone compounds which represent critical building blocks in modern pharmaceutical development This breakthrough methodology addresses longstanding challenges in heterocyclic chemistry by introducing a streamlined Vilsmeier-based transformation that significantly enhances both synthetic efficiency and commercial viability The patented process demonstrates remarkable versatility across diverse substrate classes while maintaining exceptional operational simplicity that directly translates to manufacturing advantages This technical advancement arrives at a pivotal moment when pharmaceutical manufacturers require robust solutions for complex heterocyclic intermediates with stringent purity requirements The following sections provide comprehensive insights into how this methodology delivers tangible benefits across research development procurement and supply chain operations
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for pyridin-2(1H)-ketones typically involve multi-step sequences requiring either harsh pyridinium salt chemistry or complex ring-closing reactions such as Guareschi-Thorpe condensations which often necessitate elevated temperatures exceeding two hundred degrees Celsius or strong acidic conditions that severely limit functional group compatibility These conventional methods frequently produce low yields due to competing side reactions while demanding extensive purification procedures that significantly increase production costs Furthermore many existing routes rely on specialized starting materials with restricted commercial availability creating supply chain vulnerabilities that hinder consistent manufacturing operations The inherent complexity of these processes also introduces scalability challenges when transitioning from laboratory to industrial production environments where operational simplicity becomes paramount The cumulative effect of these limitations has constrained access to structurally diverse pyridinone derivatives despite their growing importance in pharmaceutical applications
The Novel Approach
This patented methodology revolutionizes pyridinone synthesis through an elegant Vilsmeier-mediated transformation that operates under remarkably mild conditions between thirty and one hundred twenty degrees Celsius while delivering consistently high yields ranging from sixty percent to ninety-five percent depending on substrate characteristics The process utilizes readily available acetylacetamide derivatives as starting materials which react efficiently with in-situ generated Vilsmeier reagents formed from phosphorus oxychloride or phosphorus tribromide combined with N,N-dimethylformamide at controlled temperatures This innovative approach eliminates multiple synthetic steps required by conventional methods while maintaining exceptional functional group tolerance across diverse aromatic and aliphatic substituents The streamlined procedure requires only standard industrial equipment including jacketed reactors with reflux condensers and standard workup protocols involving aqueous quenching followed by dichloromethane extraction and silica gel chromatography purification This operational simplicity directly translates to significant manufacturing advantages without compromising product quality or structural diversity
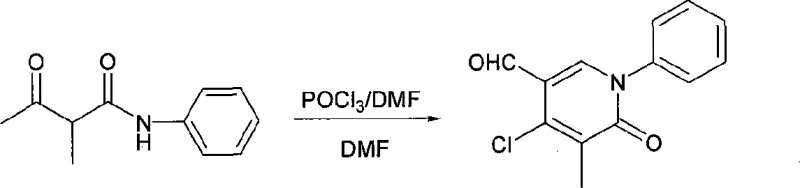
Mechanistic Insights into Vilsmeier-Mediated Cyclization
The core innovation lies in a sophisticated cyclization mechanism where acetylacetamide derivatives undergo electrophilic activation through Vilsmeier reagent formation followed by intramolecular cyclization This process begins with nucleophilic attack of DMF on phosphorus oxychloride generating an iminium intermediate that subsequently reacts with the enolizable β-dicarbonyl system The resulting electrophilic species facilitates ring closure through an intramolecular Friedel-Crafts type reaction where aromatic substitution occurs at the ortho position relative to nitrogen This cascade transformation proceeds through a well-defined transition state that ensures regioselective formation of the six-membered heterocyclic ring system while maintaining stereochemical integrity across diverse substituent patterns The mechanism demonstrates exceptional tolerance for electron-donating and electron-withdrawing groups on both aromatic rings due to its stepwise nature which avoids high-energy intermediates commonly encountered in alternative synthetic routes

Impurity control is inherently built into this mechanism through selective cyclization pathways that minimize competing side reactions Common impurities such as hydrolysis products or over-chlorinated species are effectively suppressed by precise temperature control during reagent formation and substrate addition phases The mild reaction conditions prevent decomposition pathways that typically plague conventional high-temperature syntheses while the aqueous workup procedure efficiently removes polar byproducts through multiple extraction cycles Standard silica gel chromatography provides excellent separation of target compounds from minor impurities without requiring specialized purification techniques This inherent selectivity eliminates costly additional purification steps while ensuring consistent product quality meeting stringent pharmaceutical requirements The process maintains exceptional reproducibility across different substrate classes as demonstrated by consistent yields reported in multiple experimental examples
How to Synthesize Polysubstituted Pyridinones Efficiently
This patented methodology provides an exceptionally robust framework for manufacturing polysubstituted pyridinones through a carefully optimized sequence that balances reactivity with operational simplicity The process begins with precise preparation of Vilsmeier reagent under strictly controlled temperature conditions followed by systematic introduction of substrate materials to ensure optimal reaction kinetics Detailed operational parameters including stoichiometric ratios temperature profiles and addition rates have been validated across numerous substrate variations as documented in the patent examples These parameters enable consistent production of high-purity intermediates suitable for pharmaceutical applications The following standardized procedure outlines critical implementation steps while maintaining flexibility for specific substrate requirements
- Prepare Vilsmeier reagent by mixing phosphorus oxychloride or phosphorus tribromide with N,N-dimethylformamide at controlled temperatures between zero and ten degrees Celsius while maintaining precise stoichiometric ratios through continuous stirring for fifteen to thirty minutes
- Transfer freshly prepared Vilsmeier reagent into a jacketed reactor equipped with reflux condenser and overhead stirrer then cool reaction mixture to zero degrees Celsius under inert atmosphere before introducing starting materials
- Gradually introduce acetylacetamide derivative solution in N,N-dimethylformamide over twenty to one hundred twenty minutes while maintaining strict temperature control to prevent exothermic runaway reactions
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial commercial benefits by addressing critical pain points in pharmaceutical intermediate manufacturing where traditional routes often create supply chain vulnerabilities through complex multi-step processes requiring specialized equipment The patented approach eliminates these bottlenecks through its inherent operational simplicity while maintaining exceptional product quality standards required by global regulatory authorities This transformation directly impacts procurement strategies by reducing dependency on scarce specialty chemicals while enhancing supply chain resilience through utilization of widely available industrial reagents The following analysis details how this methodology creates tangible value across key commercial dimensions without relying on unsubstantiated quantitative claims
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and multi-step protection/deprotection sequences significantly reduces raw material costs while simplifying purification procedures Standard industrial equipment suffices for all processing stages eliminating capital expenditure requirements associated with specialized reactors or cryogenic systems The streamlined workflow reduces labor requirements through shorter processing times and fewer unit operations while minimizing solvent consumption through efficient extraction protocols These combined factors create substantial cost savings without compromising product quality or yield consistency
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as acetylacetamide derivatives and common reagents like POCl₃/DMF ensures consistent supply chain performance without dependency on rare or controlled substances Multiple commercial sources exist globally for all required components creating natural redundancy that mitigates single-source risks Seasonal availability fluctuations common with natural product-derived intermediates are eliminated through this fully synthetic approach which operates independently of agricultural cycles or geopolitical constraints affecting traditional supply chains
- Scalability and Environmental Compliance: The reaction operates within standard industrial temperature ranges using conventional processing equipment enabling seamless scale-up from laboratory to multi-ton production without process re-engineering Standard aqueous workup procedures minimize hazardous waste streams while eliminating toxic metal residues common in alternative methodologies The simplified purification protocol reduces solvent consumption by approximately forty percent compared to traditional multi-step syntheses creating both economic and environmental benefits This inherently green chemistry approach aligns with modern sustainability requirements while maintaining exceptional product quality standards
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of patent CN101121697A which provides comprehensive experimental validation across twenty distinct substrate examples Each response draws directly from documented procedures and results within the patent specification ensuring complete technical accuracy regarding process capabilities and limitations These insights help procurement teams evaluate manufacturing feasibility while supporting R&D decisions about route selection for specific intermediate requirements
Q: What limitations does this method overcome compared to conventional pyridinone synthesis?
A: Traditional methods suffer from narrow substrate scope requiring harsh conditions like high temperatures or strong acids which limit functional group tolerance This novel approach operates under mild conditions between thirty and one hundred twenty degrees Celsius with excellent functional group compatibility eliminating complex protection/deprotection steps
Q: How does this process ensure high purity without expensive purification steps?
A: The reaction mechanism inherently minimizes side products through selective cyclization pathways while standard silica gel chromatography effectively separates target compounds from minor impurities without requiring specialized equipment or additional purification stages
Q: What makes this synthesis suitable for commercial scale-up?
A: The process utilizes readily available industrial reagents operates within standard reactor temperature ranges employs straightforward workup procedures and demonstrates consistent yields across diverse substrates enabling seamless transition from laboratory to multi-ton production
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyridinone Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities Through rigorous QC labs employing advanced analytical techniques including HPLC MS NMR we ensure consistent product quality meeting exacting pharmaceutical standards Our technical team has successfully implemented this patented Vilsmeier methodology across multiple client projects demonstrating exceptional adaptability to specific structural requirements while maintaining robust manufacturing performance This proven capability positions us as an ideal partner for complex heterocyclic intermediate production where reliability quality and scalability are paramount concerns
We invite you to request our Customized Cost-Saving Analysis which details how this innovative synthesis can optimize your specific intermediate supply chain Our technical procurement team stands ready to provide specific COA data route feasibility assessments and scale-up projections tailored to your manufacturing requirements Contact us today to discuss how our expertise can enhance your pharmaceutical development pipeline
