Advanced Desloratadine Manufacturing: Overcoming Hydrolysis Limitations for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes for high-volume antihistamines, and the preparation of desloratadine remains a critical focus for generic drug manufacturers. Patent CN113004245A introduces a transformative approach to synthesizing this active pharmaceutical ingredient (API) by utilizing a specific intermediate strategy that bypasses the limitations of direct hydrolysis. This technical insight report analyzes the novel methodology which employs methyl-loratadine as a starting material, reacting it with chloroethyl chloroformate to form a transient carbamate species before final deprotection. By shifting away from the traditional reliance on strong inorganic bases and prolonged heating, this process offers a pathway to significantly higher purity profiles and operational efficiency. For R&D directors and supply chain managers, understanding this shift from harsh hydrolysis to mild methanolysis is essential for optimizing production lines and ensuring consistent quality in the final API.
The Limitations of Conventional Methods vs. The Novel Approach
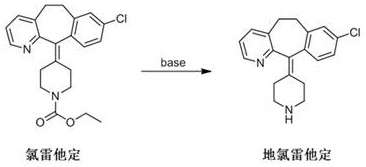
The Limitations of Conventional Methods
Historically, the industrial production of desloratadine has relied heavily on the direct hydrolysis of loratadine using strong inorganic bases such as sodium hydroxide, potassium hydroxide, or calcium hydroxide. As illustrated in the reaction scheme below, this conventional pathway typically involves heating the starting material in alcoholic solvents for extended periods, often exceeding seven hours under rigorous alkaline conditions. These harsh parameters inevitably lead to the degradation of sensitive functional groups within the complex molecular scaffold, resulting in a cocktail of difficult-to-remove impurities. Furthermore, the subsequent purification steps become increasingly burdensome as the impurity load rises, often requiring multiple recrystallizations or chromatographic separations that erode overall yield. The reliance on strong bases also introduces significant safety hazards and corrosion risks to reactor vessels, complicating the engineering controls required for safe commercial scale-up.
The Novel Approach
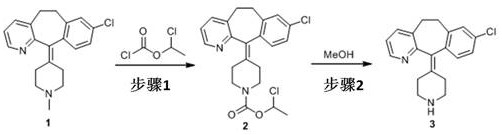
In stark contrast to the brute-force hydrolysis of the past, the methodology disclosed in CN113004245A adopts a sophisticated two-step protection-deprotection strategy that fundamentally alters the reaction landscape. Instead of attacking the ethoxycarbonyl group directly on the loratadine skeleton, the process first utilizes a precursor, methyl-loratadine, which undergoes acylation with chloroethyl chloroformate. This generates a chloroethoxy carbonyl intermediate that is chemically distinct and far more labile than its ethyl counterpart. The subsequent removal of this protecting group is achieved through a gentle reflux in methanol, eliminating the need for any acidic or basic catalysts. This mild deprotection step not only preserves the integrity of the piperidine ring and the chlorophenyl moiety but also ensures that the reaction proceeds with exceptional cleanliness, minimizing the formation of side products that plague traditional methods.
Mechanistic Insights into Chloroethoxycarbonyl Deprotection
The core innovation of this synthesis lies in the electronic and steric properties of the chloroethoxycarbonyl group compared to the standard ethoxycarbonyl group found in loratadine. The presence of the electronegative chlorine atom on the ethyl chain significantly increases the electrophilicity of the carbonyl carbon, making it much more susceptible to nucleophilic attack by methanol. In the mechanistic cycle, methanol acts as both the solvent and the nucleophile, attacking the carbonyl center to form a tetrahedral intermediate which then collapses to release the free amine (desloratadine) and the corresponding chloroethyl carbonate byproduct. This intramolecular assistance or enhanced leaving group ability allows the reaction to proceed rapidly at temperatures between 65°C and 75°C, whereas the unactivated ethyl ester would remain inert under identical neutral conditions. This mechanistic advantage translates directly to process robustness, as the reaction kinetics are favorable without requiring the extreme thermodynamic input of strong bases.
From an impurity control perspective, this mechanism offers a decisive advantage by avoiding the generation of alcoholates and other reactive basic species that can induce unwanted side reactions such as ring opening or elimination. In traditional base-mediated hydrolysis, the high concentration of hydroxide ions can attack various sites on the molecule, leading to a complex impurity spectrum that is difficult to characterize and control. By operating under neutral conditions in methanol, the novel route ensures that the only major transformation occurring is the specific cleavage of the carbamate bond. This selectivity means that the crude product obtained after solvent evaporation is of remarkably high purity, often requiring only a single recrystallization step from methyl isobutyl ketone to meet stringent pharmacopeial standards. The reduction in impurity burden simplifies the analytical workload and reduces the risk of genotoxic impurities carrying over into the final drug substance.
How to Synthesize Desloratadine Efficiently
The practical execution of this synthesis involves precise control over stoichiometry and temperature to maximize the formation of the intermediate and its subsequent conversion. The process begins with the acylation of methyl-loratadine in a halogenated solvent, followed by a straightforward workup and a final methanolysis step. The detailed standardized synthesis steps, including specific molar ratios and isolation procedures, are outlined in the guide below to assist process chemists in replicating this high-efficiency route.
- React methyl-loratadine with chloroethyl chloroformate in a solvent like 1,2-dichloroethane at 60-100°C to form the chloroethoxy carbonyl intermediate.
- Dissolve the intermediate concentrate in methanol and perform a reflux reaction at 65-75°C to remove the protecting group.
- Evaporate methanol under reduced pressure and recrystallize the residue using methyl isobutyl ketone to obtain the final desloratadine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route presents a compelling value proposition centered on cost efficiency and operational reliability. By eliminating the need for large quantities of strong inorganic bases and the associated neutralization waste streams, the process significantly reduces the consumption of auxiliary chemicals and the cost of waste disposal. The milder reaction conditions also extend the lifespan of production equipment by reducing corrosion, thereby lowering capital expenditure on maintenance and reactor replacement. Furthermore, the simplified purification protocol, which avoids complex chromatographic separations, leads to a drastic reduction in processing time and solvent usage, directly impacting the cost of goods sold (COGS) for the final API.
- Cost Reduction in Manufacturing: The elimination of harsh reagents and the reduction in purification complexity translate into substantial cost savings across the production lifecycle. Without the need for expensive ionic liquids or large volumes of neutralizing acids, the raw material costs are optimized, and the overall process mass intensity is improved. The high yield reported in the patent data suggests that less starting material is wasted, further enhancing the economic viability of the process for large-scale manufacturing operations.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents like 1,2-dichloroethane and methanol ensures that the supply chain is not vulnerable to the shortages of exotic reagents. The robustness of the reaction conditions means that batch-to-batch variability is minimized, ensuring a consistent supply of high-quality intermediates to downstream formulation teams. This reliability is crucial for maintaining uninterrupted production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: The mild nature of the deprotection step makes this process inherently safer and easier to scale from pilot plant to commercial tonnage. The absence of strong exotherms associated with base hydrolysis reduces the risk of thermal runaway, allowing for larger batch sizes with confidence. Additionally, the reduced generation of saline waste and organic byproducts aligns with increasingly strict environmental regulations, facilitating smoother regulatory approvals and sustaining long-term operational licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this desloratadine synthesis method, focusing on yield optimization, solvent selection, and impurity management. These insights are derived directly from the experimental data and comparative examples provided in the patent literature to guide decision-making.
Q: Why is the chloroethoxycarbonyl removal method superior to traditional base hydrolysis?
A: Traditional base hydrolysis requires harsh conditions (strong alkali, high temperature) leading to significant impurity formation and difficult purification. The novel method uses mild methanol reflux, drastically reducing byproduct generation and simplifying downstream processing.
Q: What solvents are suitable for the initial acylation step?
A: The patent specifies halogenated hydrocarbons such as 1,2-dichloroethane, trichloromethane, 1,2-dibromoethane, or n-bromobutane as effective reaction media for the formation of the chloroethoxy carbonyl intermediate.
Q: What is the expected yield improvement with this new route?
A: Experimental data in the patent indicates a total yield of approximately 88% for the two-step process, which is a substantial improvement over the roughly 71% yield observed in comparative examples using traditional sodium hydroxide hydrolysis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desloratadine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the chloroethoxycarbonyl route can be seamlessly transferred to industrial manufacturing. We are committed to delivering high-purity desloratadine and its intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance ensures that every batch conforms to the highest international standards, providing our partners with the confidence they need to advance their drug development pipelines.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this milder, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a strategic partner in your supply chain for high-value pharmaceutical intermediates.
