Advanced Palladium-Catalyzed Synthesis of C-1 Deuterated Aromatic Aldehydes for Commercial Scale
The landscape of modern pharmaceutical development is increasingly reliant on deuterated compounds to enhance metabolic stability and optimize pharmacokinetic profiles, a trend exemplified by the approval of drugs like Austedo. In this context, patent CN114656347A introduces a transformative methodology for the synthesis of C-1 deuterated aromatic aldehydes, which serve as critical building blocks for a wide array of bioactive molecules. This innovative approach leverages a palladium-catalyzed system that utilizes aryl sulfur salt compounds, sodium deuterated formate, and carbon monoxide to achieve exceptional deuteration efficiency. Unlike traditional methods that often struggle with selectivity and harsh conditions, this protocol operates under relatively mild thermal parameters while maintaining robust functional group compatibility. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this technology represents a significant leap forward in accessing high-value isotopic labels. The ability to introduce a deuterium atom specifically at the C-1 position of the aldehyde moiety with high fidelity opens new avenues for drug design and mechanistic studies, ensuring that supply chains can support the growing demand for deuterated active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of C-1 deuterated aromatic aldehydes has been fraught with significant technical challenges that hinder efficient commercial production. Classical strategies often depend on the reduction of carboxylic acid esters or amides using stoichiometric amounts of expensive and hazardous deuterated reducing agents such as lithium aluminum deuteride, which poses severe safety risks and generates substantial chemical waste. Alternative approaches involving photocatalytic decarboxylative deuteration or radical-mediated hydrogen-deuterium exchange frequently suffer from limited substrate scope and poor regioselectivity, leading to complex mixtures that are difficult to purify. Furthermore, many existing protocols require cryogenic temperatures or specialized light sources, making them impractical for cost reduction in pharma manufacturing on a multi-kilogram scale. These limitations not only inflate the cost of goods but also introduce variability in the isotopic purity of the final product, which is unacceptable for regulatory compliance in drug substance manufacturing. Consequently, there has been a persistent industry need for a more robust, scalable, and atom-economical solution.
The Novel Approach
The methodology disclosed in the patent data offers a compelling alternative by employing a palladium-catalyzed carbonylation strategy that circumvents the drawbacks of previous techniques. By utilizing aryl sulfur salts as the electrophilic coupling partners, the reaction benefits from enhanced oxidative addition rates compared to traditional aryl halides, facilitating smoother catalytic turnover. The use of sodium deuterated formate as the deuterium source is particularly advantageous, as it is inexpensive, stable, and easy to handle, thereby significantly simplifying the operational complexity of the synthesis. This novel route demonstrates excellent adaptability to various functional groups, including halogens, ethers, and heterocycles, ensuring that complex molecular architectures can be constructed without extensive protecting group manipulation. 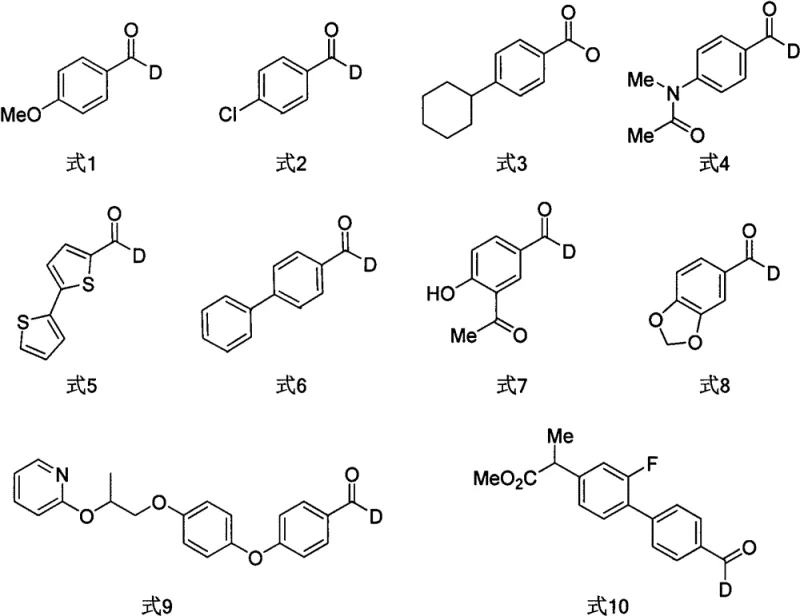 The broad substrate scope illustrated in the patent examples confirms that this method is not limited to simple benzene derivatives but extends to biologically relevant heterocyclic systems, making it an invaluable tool for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
The broad substrate scope illustrated in the patent examples confirms that this method is not limited to simple benzene derivatives but extends to biologically relevant heterocyclic systems, making it an invaluable tool for the commercial scale-up of complex polymer additives and pharmaceutical intermediates alike.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Deuteration
At the heart of this synthetic breakthrough lies a sophisticated catalytic cycle driven by palladium species coordinated with bulky phosphine ligands. The reaction initiates with the oxidative addition of the aryl sulfur salt to the active palladium(0) species, generating an aryl-palladium(II) intermediate that is primed for carbonyl insertion. Under a carbon monoxide atmosphere, the CO molecule inserts into the palladium-carbon bond to form an acyl-palladium complex, which is the key precursor to the aldehyde functionality. Subsequently, the sodium deuterated formate acts as both a base and a deuterium donor, facilitating the reductive elimination step that releases the C-1 deuterated aromatic aldehyde product while regenerating the palladium catalyst. 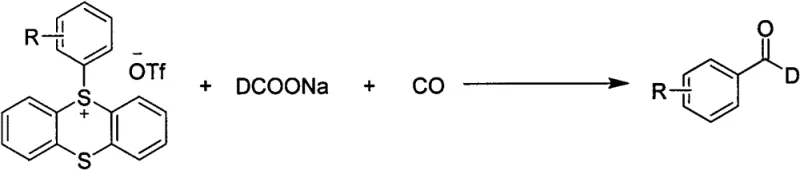 This mechanism ensures that the deuterium atom is incorporated specifically at the formyl position, avoiding the scrambling often seen in H/D exchange protocols. The choice of tris(1-naphthyl)phosphine as the ligand is critical, as its steric bulk stabilizes the active catalytic species and prevents the formation of inactive palladium black, thereby sustaining high turnover numbers throughout the reaction duration.
This mechanism ensures that the deuterium atom is incorporated specifically at the formyl position, avoiding the scrambling often seen in H/D exchange protocols. The choice of tris(1-naphthyl)phosphine as the ligand is critical, as its steric bulk stabilizes the active catalytic species and prevents the formation of inactive palladium black, thereby sustaining high turnover numbers throughout the reaction duration.
From an impurity control perspective, this mechanism offers distinct advantages over reduction-based methods. Since the deuterium source is introduced during the bond-forming event rather than through a post-synthetic exchange, the risk of incomplete labeling is minimized. The patent data indicates deuteration levels reaching up to 99%, which is essential for meeting the stringent purity specifications required for clinical trial materials. Furthermore, the use of aryl sulfur salts minimizes the formation of homocoupling byproducts that are common in aryl halide chemistry, resulting in a cleaner crude reaction profile. This high level of chemical fidelity reduces the burden on downstream purification processes, such as chromatography or crystallization, ultimately leading to higher overall yields and reduced solvent consumption. For quality assurance teams, this translates to a more predictable and controllable manufacturing process with fewer out-of-specification batches.
How to Synthesize C-1 Deuterated Aromatic Aldehydes Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and environmental controls to maximize yield and isotopic enrichment. The process begins with the careful preparation of the reaction vessel under an inert atmosphere, followed by the sequential addition of the palladium catalyst, ligand, aryl sulfur salt, sodium deuterated formate, and base in N,N-dimethylformamide solvent. Maintaining a consistent carbon monoxide pressure and a temperature of 120°C for approximately 12 hours is crucial to drive the reaction to completion. Detailed standardized synthesis steps are provided below to ensure reproducibility across different laboratory and plant settings.
- Prepare the reaction mixture by dispersing aryl sulfur salt, sodium deuterated formate, palladium catalyst, phosphine ligand, and base in N,N-dimethylformamide solvent.
- Conduct the reaction under a carbon monoxide atmosphere at 120°C for 12 hours to ensure complete conversion and high deuteration rates.
- Filter the reaction mixture, remove solvent via rotary evaporation, and purify the crude product using silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical performance. The reliance on readily available starting materials such as aryl sulfur salts and sodium deuterated formate mitigates the risk of raw material shortages that often plague specialized deuterated reagent markets. This accessibility ensures a more resilient supply chain, reducing lead time for high-purity pharmaceutical intermediates and allowing for better inventory planning. Moreover, the operational simplicity of the reaction, which avoids cryogenic conditions or exotic reagents, lowers the barrier for technology transfer from R&D to manufacturing sites. This ease of implementation accelerates the timeline from process development to commercial production, enabling faster market entry for new deuterated drug candidates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive stoichiometric reducing agents and the use of catalytic amounts of palladium. By replacing hazardous reagents like lithium aluminum deuteride with safe, solid sodium deuterated formate, facilities can reduce costs associated with special handling, waste disposal, and safety infrastructure. The high atom economy of the carbonylation reaction further contributes to substantial cost savings by minimizing raw material waste. Additionally, the high selectivity of the reaction reduces the need for extensive purification, lowering solvent usage and energy consumption during isolation. These factors collectively drive down the cost of goods sold, making deuterated intermediates more accessible for broader therapeutic applications.
- Enhanced Supply Chain Reliability: The robustness of the aryl sulfur salt substrate ensures consistent quality and availability, which is critical for maintaining continuous manufacturing operations. Unlike sensitive organometallic reagents that require cold chain logistics, the solid reagents used in this protocol are stable at ambient conditions, simplifying storage and transportation. This stability reduces the risk of degradation during transit and ensures that production schedules are not disrupted by material spoilage. Furthermore, the scalability of the reaction allows suppliers to respond flexibly to fluctuating demand, providing a secure source of critical intermediates for long-term drug development projects.
- Scalability and Environmental Compliance: From an environmental standpoint, this method aligns well with green chemistry principles by reducing the generation of hazardous waste streams. The use of DMF as a solvent is well-established in the industry, with mature recovery and recycling protocols already in place at most manufacturing sites. The absence of heavy metal reducing agents simplifies the effluent treatment process, ensuring compliance with increasingly strict environmental regulations. The reaction conditions are also amenable to large-scale reactor setups, allowing for the seamless transition from gram-scale optimization to ton-scale production without significant re-engineering. This scalability ensures that the supply of deuterated materials can grow in tandem with the clinical and commercial success of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deuterium labeling technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing supply chains.
Q: What are the advantages of using aryl sulfur salts over aryl halides for deuteration?
A: Aryl sulfur salts offer superior stability and reactivity in palladium-catalyzed carbonylation, allowing for milder conditions and higher functional group tolerance compared to traditional aryl halide substrates which often require harsher reducing agents.
Q: How does this method improve the isotopic purity of the final aldehyde?
A: By utilizing sodium deuterated formate as the specific deuterium source in conjunction with carbon monoxide insertion, the method achieves deuteration rates exceeding 99%, significantly minimizing protio-impurities common in exchange reactions.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available reagents, standard solvents like DMF, and operates at manageable temperatures, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable C-1 Deuterated Aromatic Aldehyde Supplier
As the demand for deuterated therapeutics continues to surge, partnering with an experienced CDMO is vital for navigating the complexities of isotopic synthesis. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from benchtop to market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of C-1 deuterated aromatic aldehyde meets the highest standards of isotopic enrichment and chemical purity. We understand the critical nature of these intermediates in the drug development timeline and are committed to delivering consistent quality.
We invite you to engage with our technical procurement team to discuss how this advanced palladium-catalyzed technology can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this superior synthetic route. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted ally in your supply chain for high-value fine chemicals.
