Advanced Palladium-Catalyzed Synthesis of Polysubstituted (E)-Trifluoromethyl Olefins for Commercial Scale-Up
The rapid evolution of modern medicinal chemistry and materials science has placed a premium on fluorinated building blocks, particularly those containing the trifluoromethyl group due to its profound impact on metabolic stability and lipophilicity. Patent CN113754544A introduces a groundbreaking preparation method for polysubstituted (E)-trifluoromethyl olefins, addressing critical bottlenecks in the synthesis of these high-value intermediates. Unlike traditional approaches that struggle with stereochemical control and waste generation, this invention leverages a sophisticated palladium-catalyzed allylic substitution strategy. By utilizing readily available allyl carbonate compounds and a diverse range of nucleophiles, the process delivers target molecules with unprecedented efficiency. This technological leap is particularly relevant for the development of next-generation bioactive molecules and photoluminescent materials, where precise structural definition is paramount for performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted olefins has relied heavily on the Horner-Wittig reaction, a method fraught with significant operational and environmental drawbacks for large-scale manufacturing. The primary disadvantage lies in the generation of substantial quantities of phosphorus-containing byproducts, which are notoriously difficult to separate from the desired product, thereby complicating downstream purification and increasing waste disposal costs. Furthermore, the Horner-Wittig reaction typically yields a mixture of E and Z isomers, necessitating energy-intensive separation processes to isolate the biologically active E-isomer, often resulting in significant material loss. Additionally, this classical method is structurally limited, generally restricted to the synthesis of tri-substituted olefins and failing to accommodate the synthesis of more complex tetra-substituted variants required for advanced drug candidates. The multi-step nature of subsequent conversions further depresses overall yields and escalates production costs, rendering it suboptimal for the cost reduction in fine chemical manufacturing demanded by today's competitive market.
The Novel Approach
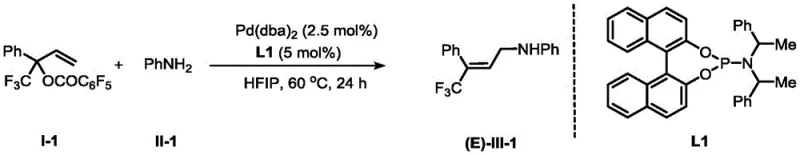
In stark contrast, the methodology disclosed in CN113754544A utilizes a palladium-catalyzed allylic substitution that fundamentally redefines the synthetic landscape for these compounds. This innovative route employs allyl carbonates as electrophiles, which react with nucleophiles such as aromatic amines, sodium aryl sulfinates, phenols, or water under mild conditions to directly forge the carbon-carbon or carbon-heteroatom bond. The process is characterized by its exceptional atom economy and the complete avoidance of stoichiometric phosphorus waste, aligning perfectly with green chemistry principles. Most critically, the reaction exhibits superb stereoselectivity, consistently producing the (E)-isomer with ratios exceeding 99:1, effectively bypassing the need for difficult isomer separations. This direct and efficient pathway not only simplifies the operational workflow but also significantly enhances the overall yield, with some examples demonstrating yields up to 89%, making it a superior choice for reliable pharmaceutical intermediate supplier operations.

Mechanistic Insights into Palladium-Catalyzed Allylic Substitution
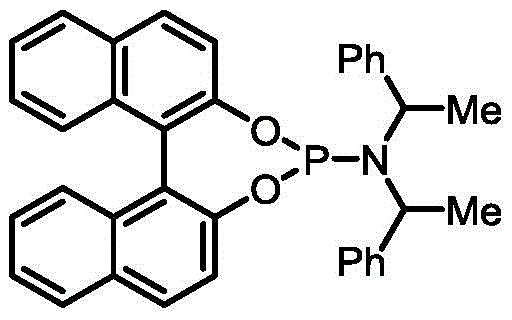
The success of this transformation hinges on the precise interplay between the palladium catalyst and the specialized organophosphorus ligand L1, which orchestrates the stereochemical outcome of the reaction. The catalytic cycle initiates with the oxidative addition of the allyl carbonate to the zero-valent palladium species, generating a cationic pi-allyl palladium intermediate that is stabilized by the electron-rich environment provided by the ligand. The unique steric bulk and electronic properties of ligand L1, a binaphthyl-derived phosphoramidite, play a pivotal role in directing the nucleophilic attack to occur exclusively at the terminal position of the allyl system with high facial selectivity. This steric guidance ensures that the incoming nucleophile approaches the intermediate in a trajectory that favors the formation of the thermodynamically stable (E)-alkene geometry, suppressing the formation of the Z-isomer. The robustness of this catalytic system allows it to tolerate a wide array of functional groups on both the allyl carbonate and the nucleophile, ensuring high substrate universality.
Furthermore, the mechanism facilitates excellent impurity control by minimizing side reactions such as beta-hydride elimination or homocoupling, which are common pitfalls in palladium chemistry. The use of hexafluoroisopropanol (HFIP) or acetonitrile as solvents further enhances the ionization of the allyl carbonate, accelerating the formation of the reactive pi-allyl species while maintaining a homogeneous reaction environment. For nucleophiles with lower acidity, such as phenols or water, the addition of a mild inorganic base like cesium carbonate activates the nucleophile without compromising the integrity of the sensitive trifluoromethyl group. This mechanistic elegance translates directly to a cleaner reaction profile, reducing the burden on purification teams and ensuring that the final product meets stringent purity specifications required for clinical applications. The ability to synthesize tetra-substituted olefins, which are inaccessible via traditional Wittig chemistry, opens new avenues for exploring chemical space in drug discovery.
How to Synthesize Polysubstituted (E)-Trifluoromethyl Olefins Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process begins with the activation of the catalyst system, followed by the controlled addition of substrates to manage exotherms and maintain reaction homogeneity. Detailed standard operating procedures regarding stoichiometry, temperature gradients, and workup protocols are essential for translating this academic breakthrough into a robust manufacturing process. For a comprehensive guide on the specific molar ratios, solvent choices, and purification techniques validated across multiple examples, please refer to the standardized synthesis steps outlined below.
- Prepare the catalytic system by mixing palladium salt (e.g., Pd(dba)2) and the specialized organophosphorus ligand L1 in an organic solvent such as HFIP or acetonitrile.
- Introduce the allyl carbonate substrate and the chosen nucleophilic reagent (amine, sulfinate, phenol, or water) to the reaction mixture and stir at 60°C for 2 to 24 hours.
- Upon completion, remove the solvent under reduced pressure and purify the crude product via silica gel column chromatography to isolate the target (E)-olefin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this palladium-catalyzed technology offers transformative advantages that extend far beyond simple yield improvements. The elimination of phosphorus-containing byproducts fundamentally alters the cost structure of production by removing the need for expensive and time-consuming waste treatment processes associated with phosphine oxides. This streamlined workflow significantly reduces the consumption of solvents and silica gel during purification, leading to substantial cost savings in raw materials and waste disposal. Moreover, the use of economically accessible starting materials like allyl carbonates and common anilines ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or volatile reagents. The mild reaction conditions, operating effectively at temperatures as low as room temperature to 60°C, drastically reduce energy consumption compared to high-temperature traditional methods, contributing to a lower carbon footprint and reduced utility costs.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic simplification of the purification train. By avoiding the generation of difficult-to-remove phosphorus waste, manufacturers can reduce the number of chromatography columns or crystallization steps required, directly lowering labor and material costs. The high stereoselectivity means that no material is wasted on separating unwanted Z-isomers, effectively doubling the usable output per batch compared to non-selective methods. Additionally, the low catalyst loading of 2.5 mol% minimizes the expense of precious metal usage, while the potential for catalyst recovery further enhances the economic viability of the process for large-scale campaigns.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as allyl carbonates and simple aromatic amines ensures that raw material availability is not a bottleneck for production scaling. Unlike specialized phosphonates which may have limited suppliers and long lead times, the substrates for this reaction are widely produced for various industrial applications, guaranteeing consistent quality and pricing. The robustness of the reaction against moisture and air, as evidenced by the 'without nitrogen protection' examples in the patent, reduces the need for specialized inert atmosphere equipment, simplifying facility requirements and increasing operational flexibility. This resilience translates to fewer production delays and a more predictable delivery schedule for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is inherently safer and more sustainable, facilitating easier regulatory approval for commercial manufacturing. The absence of toxic phosphorus byproducts simplifies wastewater treatment and aligns with increasingly stringent global environmental regulations regarding chemical discharge. The process is highly scalable, having been demonstrated to work efficiently with varying substrate loads, which supports the commercial scale-up of complex pharmaceutical intermediates from kilogram to multi-ton quantities. This scalability ensures that supply can easily ramp up to meet market demand without the need for extensive process re-engineering or additional capital investment in specialized reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical application of the method. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing pipelines and for procurement teams assessing the long-term value proposition.
Q: What is the stereoselectivity of this palladium-catalyzed method?
A: The method demonstrates exceptional stereocontrol, consistently achieving E:Z ratios greater than 99:1 across various substrates, effectively eliminating the difficult separation of geometric isomers common in traditional Wittig reactions.
Q: Which nucleophiles are compatible with this allylic substitution protocol?
A: The process exhibits broad substrate universality, successfully accommodating aromatic amines, sodium aryl sulfinates, phenols, and even water as nucleophiles, allowing for the diverse functionalization of the trifluoromethyl olefin scaffold.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions (room temperature to 60°C) with low catalyst loading (2.5 mol%), avoids hazardous phosphorus byproducts, and utilizes commercially available starting materials, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted (E)-Trifluoromethyl Olefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in accelerating drug discovery and material innovation. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise E/Z ratios essential for the biological activity of your target molecules. We are committed to delivering not just chemicals, but reliable solutions that enhance your supply chain security and product performance.
We invite you to collaborate with our technical team to explore how this advanced palladium-catalyzed technology can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this superior synthetic route. Contact our technical procurement team today to obtain specific COA data for our catalog compounds or to discuss custom route feasibility assessments tailored to your unique molecular targets.
