Scalable Cobalt-Catalyzed Synthesis of Alpha-Oxo Acetophenones for Advanced Pharmaceutical Manufacturing
Scalable Cobalt-Catalyzed Synthesis of Alpha-Oxo Acetophenones for Advanced Pharmaceutical Manufacturing
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways, particularly for high-value intermediates used in the pharmaceutical and agrochemical sectors. Patent CN113698337B introduces a groundbreaking preparation method for alpha-oxo acetophenone compounds that addresses critical bottlenecks in traditional organic synthesis. This technology leverages a transition metal cobalt catalyst in conjunction with N-hydroxyphthalimide (NHPI) and molecular oxygen to achieve highly selective oxidation of styrene derivatives. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a pivotal shift away from toxic stoichiometric oxidants toward catalytic aerobic processes. The ability to synthesize these versatile building blocks under mild conditions not only enhances safety profiles but also aligns with stringent global environmental regulations, making it an ideal candidate for integration into modern supply chains focused on sustainability and cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic ketones and their alpha-oxo derivatives has relied heavily on classical methodologies such as Friedel-Crafts acylation or the oxidative dehydrogenation of aromatic alcohols, both of which present substantial drawbacks for modern industrial application. These traditional routes frequently necessitate the use of harsh reagents, including chromium salts and permanganates, which act as stoichiometric oxidants rather than catalytic species, leading to the generation of massive quantities of hazardous heavy metal waste that requires expensive disposal protocols. Furthermore, these reactions often suffer from poor regioselectivity and limited substrate scope, forcing manufacturers to employ complex protection-deprotection strategies that inflate production costs and extend lead times. The reliance on dangerous preparation conditions and the inability to efficiently manage the resulting environmental burden have severely restricted the utility of these legacy methods in the synthesis of complex natural products and advanced drug candidates, creating a pressing demand for alternative technologies.
The Novel Approach
In stark contrast to these outdated techniques, the methodology disclosed in the patent utilizes a sophisticated catalytic system where styrene compounds and N-hydroxyphthalimide serve as the primary raw materials, driven by the thermodynamic potential of green molecular oxygen. This novel approach eliminates the need for toxic heavy metal oxidants by employing a cobalt catalyst to facilitate a radical-mediated oxidation pathway that is both atom-economical and environmentally benign. The reaction proceeds under remarkably mild conditions, typically at temperatures around 40°C, which minimizes energy consumption and reduces the risk of thermal runaway incidents common in exothermic oxidations. By achieving high selectivity and yield without the generation of noxious byproducts, this process offers a streamlined route to alpha-oxo acetophenones that is perfectly suited for the rigorous demands of contemporary chemical manufacturing.
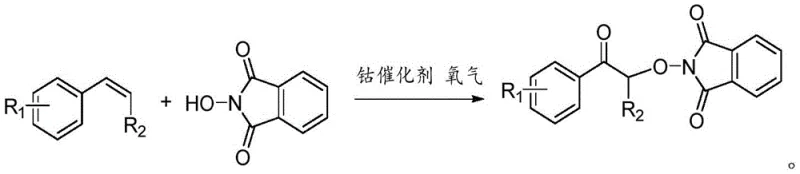
The core of this technological advancement lies in the general reaction scheme where diverse styrene substrates are converted into valuable alpha-oxo acetophenone derivatives through a cooperative catalytic cycle. As illustrated in the reaction diagram, the versatility of this method is evident in its tolerance for various electronic and steric environments on the starting material, allowing for the synthesis of a wide array of functionalized products essential for medicinal chemistry. The use of oxygen as the terminal oxidant ensures that the only byproduct is water or incorporated oxygen atoms, drastically simplifying the downstream purification process and enhancing the overall green chemistry metrics of the synthesis. This level of efficiency and cleanliness is precisely what forward-thinking procurement managers look for when evaluating partners for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cobalt-Catalyzed Aerobic Oxidation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific target molecules or scale-up scenarios. The reaction operates through a radical chain mechanism initiated by the interaction between the cobalt catalyst and N-hydroxyphthalimide (NHPI) under an oxygen atmosphere. The cobalt species facilitates the generation of phthalimide-N-oxyl (PINO) radicals from NHPI, which are highly effective hydrogen abstractors capable of activating the benzylic C-H bonds of the styrene substrate. This activation step is the rate-determining factor that dictates the overall efficiency of the transformation, and the specific choice of cobalt salt, such as cobalt acetate, plays a pivotal role in modulating the redox potential to ensure rapid turnover without catalyst deactivation. The resulting carbon-centered radicals react swiftly with molecular oxygen to form peroxy intermediates, which subsequently undergo rearrangement and further oxidation to yield the desired alpha-oxo ketone functionality with high fidelity.
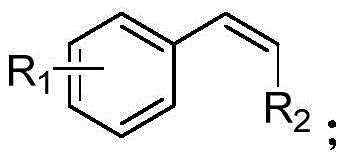
The structural diversity accommodated by this catalytic system is extensive, as evidenced by the general formula of the styrene compounds utilized in the study. The R1 group can vary from hydrogen to alkyl chains or halogens, while the R2 position tolerates hydrogen, alkyl, or phenyl substituents, demonstrating the robustness of the catalytic cycle against steric hindrance and electronic variation. This broad substrate scope is a critical advantage for pharmaceutical applications where late-stage functionalization often requires the modification of complex molecular scaffolds without compromising sensitive functional groups. The mechanism ensures that the oxidation occurs selectively at the desired position, minimizing the formation of over-oxidized byproducts like carboxylic acids or cleavage products that often plague non-selective oxidation methods. Such precise control over the reaction trajectory is indicative of a mature chemical process ready for commercial adoption.
How to Synthesize Alpha-Oxo Acetophenone Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and purity while maintaining safety standards. The process is designed to be operationally simple, involving the mixing of readily available reagents in a standard reactor setup followed by controlled heating and gas introduction. Detailed procedural steps regarding reagent ratios, solvent choices, and workup procedures are critical for reproducibility, and the patent provides comprehensive data supporting the optimization of these variables. For technical teams looking to adopt this methodology, following the standardized protocol ensures consistent results across different batches, which is fundamental for quality assurance in regulated industries.
- Mix cobalt catalyst (e.g., cobalt acetate), styrene compound, N-hydroxyphthalimide, and organic solvent (preferably acetonitrile) in a reactor.
- Introduce oxygen gas (1 atm) into the reaction mixture and stir while heating to 40°C for approximately 2 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, distill under reduced pressure, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this cobalt-catalyzed oxidation technology offers profound benefits that extend beyond mere chemical efficiency, directly impacting the bottom line and supply chain resilience for chemical manufacturers. The shift from stoichiometric heavy metal oxidants to a catalytic system using air or oxygen fundamentally alters the cost structure of production by eliminating the procurement of expensive and hazardous reagents. Furthermore, the reduction in hazardous waste generation translates to significantly lower disposal costs and reduced regulatory compliance burdens, which are increasingly significant factors in the total cost of ownership for chemical processes. For supply chain heads, the reliance on abundant and inexpensive raw materials like styrenes and oxygen ensures a stable supply base that is less susceptible to market volatility compared to specialized oxidizing agents.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of costly stoichiometric oxidants with molecular oxygen, which is effectively free when sourced from air, and the use of earth-abundant cobalt catalysts instead of precious metals. This substitution leads to a drastic reduction in raw material expenses while simultaneously removing the need for complex wastewater treatment facilities required to handle heavy metal effluents. The simplified purification workflow, often requiring only extraction and distillation, further reduces energy consumption and labor costs associated with multi-step chromatographic separations. Consequently, manufacturers can achieve substantial cost savings that can be passed down to clients or reinvested into further process optimization.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the availability of starting materials contribute to a highly reliable supply chain capable of meeting demanding delivery schedules. Since the reagents involved are commodity chemicals with established global supply networks, the risk of production delays due to raw material shortages is minimized. Additionally, the mild reaction conditions reduce the wear and tear on reactor equipment, leading to lower maintenance downtime and higher overall equipment effectiveness. This reliability is crucial for pharmaceutical companies that require just-in-time delivery of high-purity intermediates to maintain their own production timelines without interruption.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the homogeneous nature of the catalytic system and the use of standard organic solvents like acetonitrile, which are well-understood in industrial settings. The environmental profile of the process aligns perfectly with green chemistry principles, making it easier to obtain necessary environmental permits and maintain compliance with increasingly strict international regulations. The absence of toxic heavy metal waste simplifies the environmental impact assessment and reduces the long-term liability associated with hazardous waste storage and disposal. This sustainability advantage serves as a strong value proposition for companies aiming to reduce their carbon footprint and enhance their corporate social responsibility profiles.
Frequently Asked Questions (FAQ)
To assist stakeholders in evaluating the feasibility of integrating this technology into their existing workflows, we have compiled answers to common technical and commercial inquiries based on the patent data. These questions address key concerns regarding substrate compatibility, catalyst recovery, and product purity, providing a clear picture of the method's practical utility. Understanding these details is essential for making informed decisions about process adoption and partnership opportunities.
Q: What are the primary advantages of this cobalt-catalyzed method over traditional Friedel-Crafts acylation?
A: Unlike traditional methods that often require hazardous chromium salts or expensive auxiliary metals, this protocol utilizes inexpensive cobalt catalysts and molecular oxygen, significantly reducing heavy metal waste and operational costs.
Q: Can this synthesis method accommodate diverse styrene substrates?
A: Yes, the patent demonstrates high tolerance for various substituents including alkyl and halogen groups on the aromatic ring, as well as different R2 groups, ensuring broad applicability for diverse intermediate synthesis.
Q: Is the purification process complex for industrial scale-up?
A: The workup involves standard liquid-liquid extraction followed by distillation and chromatography, which are well-established unit operations easily adaptable for large-scale commercial production without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Oxo Acetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN113698337B and possess the technical expertise to bring such innovations to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of alpha-oxo acetophenone meets the highest standards required by the global pharmaceutical industry. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply of critical intermediates.
We invite you to collaborate with us to explore how this green synthesis method can be tailored to your specific project needs, offering a pathway to more sustainable and cost-effective manufacturing. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your target molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your R&D and production goals, ensuring a competitive edge in the marketplace through superior chemical solutions.
