Advanced Chiral Spiro Aminophosphine Ligands for High-Efficiency Asymmetric Catalysis
Advanced Chiral Spiro Aminophosphine Ligands for High-Efficiency Asymmetric Catalysis
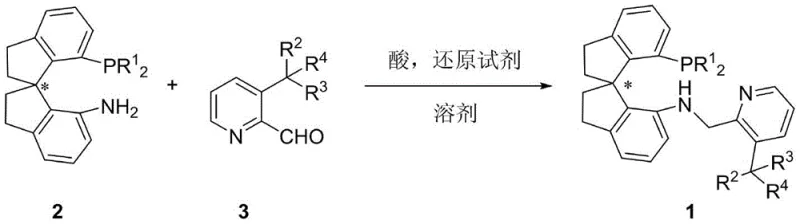
The landscape of asymmetric synthesis is continually evolving, driven by the urgent need for more efficient and sustainable methods to construct chiral centers, particularly quaternary carbons, which are ubiquitous in bioactive molecules. As detailed in patent CN110724164B, a groundbreaking advancement has been made in the development of 3-substituted chiral spiro aminophosphine ligands on a pyridine ring. These novel compounds represent a significant leap forward in ligand design, featuring a unique chiral spiro indane skeleton coupled with a sterically demanding pyridine group. This structural innovation addresses long-standing challenges in the asymmetric catalytic hydrogenation of beta-arylalkylene malonates, a reaction class critical for producing key pharmaceutical intermediates. The technology offers a robust pathway to optically active 3-aryl fatty acids and esters, which serve as vital precursors for drugs like Tipranavir and natural products such as (-)-juvabione. By integrating these advanced ligands into existing catalytic frameworks, manufacturers can achieve unprecedented levels of stereocontrol and catalytic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral compounds containing arylalkyl-substituted tertiary carbon centers at the beta-position has relied heavily on the asymmetric catalytic hydrogenation of alpha,beta-unsaturated carboxylic acids and their ester derivatives. While these methods possess high atom economy in theory, they suffer from severe practical limitations that hinder their widespread industrial adoption. A primary drawback is the narrow substrate scope; conventional catalysts often fail to accommodate diverse steric and electronic environments, limiting their utility in complex molecule synthesis. Furthermore, these traditional processes typically demand high catalyst loadings, frequently exceeding 1 mol%, which drastically increases production costs due to the expense of precious metals like iridium or rhodium. Perhaps most critically, the reaction conditions are often harsh, requiring hydrogen pressures between 50 and 100 atm to drive the reaction to completion. Such extreme conditions necessitate specialized, high-cost equipment and pose significant safety risks, making scale-up difficult and expensive for many fine chemical manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN110724164B utilizes beta-arylalkylene malonates as substrates, which do not possess the restrictive Z or E configuration issues of their unsaturated counterparts. This novel approach leverages the newly designed 3-substituted chiral spiro aminophosphine ligands to form highly active iridium complexes in situ. The introduction of sterically significant substituents at the 3-position of the pyridine ring enhances chirality transfer during the catalytic cycle, resulting in superior enantioselectivity. The synthesis of these ligands themselves is remarkably straightforward, involving a reductive amination between a chiral spiroindane amine and a substituted pyridine aldehyde, as illustrated in the reaction scheme above. This streamlined preparation avoids complex multi-step sequences, allowing for rapid access to a library of ligand variants. The resulting catalytic system operates under significantly milder conditions, often at room temperature and moderate hydrogen pressures, while maintaining exceptional activity and selectivity, thereby overcoming the major bottlenecks of previous generations of catalysts.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The exceptional performance of this catalytic system stems from the precise three-dimensional architecture of the ligand-metal complex. The chiral spiro indane backbone provides a rigid scaffold that locks the phosphine and amine donors into a specific geometry, while the bulky substituents on the pyridine ring create a well-defined chiral pocket around the iridium center. During the catalytic cycle, this steric environment effectively discriminates between the prochiral faces of the beta-arylalkylene malonate substrate. The mechanism likely involves the coordination of the substrate to the iridium center followed by hydride transfer, where the steric bulk at the pyridine 3-position plays a pivotal role in directing the approach of the hydrogen molecule and the substrate. This precise control ensures that the hydrogenation proceeds with high fidelity to a single enantiomer, minimizing the formation of unwanted isomers that would otherwise require costly separation steps. The stability of the iridium complex is also enhanced by the tridentate nature of the ligand, preventing catalyst decomposition and allowing for extended operational lifetimes.
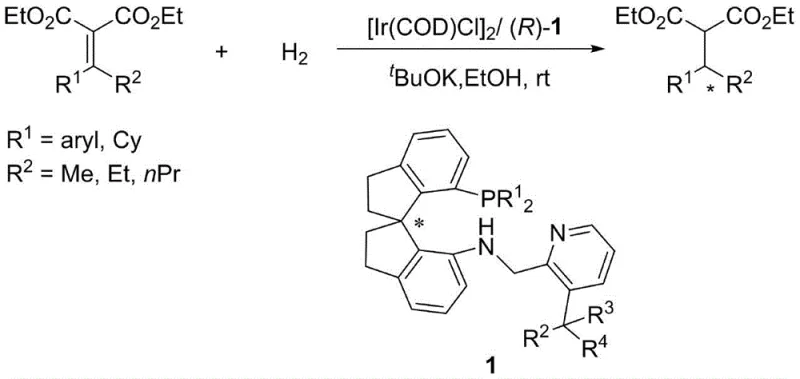
Furthermore, the impurity profile of the final product is significantly improved due to the high specificity of the catalyst. In traditional hydrogenation processes, side reactions such as over-reduction or isomerization can lead to complex mixtures that are difficult to purify. However, the tailored electronic and steric properties of the 3-substituted spiro ligands suppress these competing pathways. The high turnover numbers (TON) observed, reaching up to 19,000, indicate that each catalyst molecule can process a vast number of substrate molecules before deactivation. This efficiency not only reduces the amount of catalyst required but also minimizes the residual metal content in the final API intermediate, a critical quality attribute for pharmaceutical regulatory compliance. The ability to achieve up to 99% ee means that the downstream purification burden is drastically reduced, leading to higher overall process yields and lower manufacturing costs.
How to Synthesize 3-Substituted Chiral Spiro Aminophosphine Ligands Efficiently
The preparation of these high-value ligands follows a robust and scalable reductive amination protocol that is amenable to kilogram-scale production. The process begins with the condensation of a chiral 7-diaryl/alkyl phosphino-7'-amino-1,1'-spirodihydroindene precursor with a variety of 3-substituted-2-pyridinecarbaldehydes. This reaction is typically conducted in solvents such as 1,2-dichloroethane (DCE) at moderate temperatures around 45°C. Once the imine intermediate is formed, a mild reducing agent like sodium triacetoxyborohydride is introduced to furnish the final secondary amine linkage. The detailed standardized synthesis steps for specific ligand variants are provided in the guide below.
- React chiral 7-diaryl/alkyl phosphino-7'-amino-1,1'-spirodihydroindene with a substituted pyridine aldehyde in an organic solvent like DCE at 45°C.
- Monitor imine intermediate formation via TLC, then add a reducing agent such as sodium triacetoxyborohydride to complete the reductive amination.
- Purify the resulting crude mixture using silica gel column chromatography to isolate the high-purity chiral ligand with yields up to 97%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers compelling economic and operational benefits that extend far beyond simple yield improvements. The shift towards this novel catalytic system addresses several critical pain points in the manufacturing of chiral pharmaceutical intermediates, specifically regarding cost structure, supply reliability, and environmental compliance. By enabling reactions to proceed with ultra-low catalyst loadings and under milder conditions, the total cost of ownership for the synthesis process is significantly optimized. This makes the production of complex chiral building blocks more economically viable, especially for high-volume API manufacturing where marginal cost savings translate into substantial financial gains.
- Cost Reduction in Manufacturing: The most immediate impact is seen in the drastic reduction of catalyst consumption. With turnover numbers reaching 19,000, the molar percentage of the expensive iridium catalyst required can be lowered to as little as 0.005 mol%. This represents a massive decrease in the direct material cost associated with precious metals. Additionally, the elimination of high-pressure requirements (reducing from 100 atm to potentially ambient or low pressure) lowers the capital expenditure needed for reactor infrastructure and reduces energy consumption for compression. The simplified workup procedures, facilitated by high selectivity, further reduce solvent usage and waste disposal costs, contributing to a leaner and more cost-effective manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of the ligand synthesis and the catalytic reaction ensures a more reliable supply of critical intermediates. The starting materials, such as the spiroindane amines and substituted pyridine aldehydes, are accessible via established synthetic routes, mitigating the risk of raw material shortages. Furthermore, the tolerance of the catalyst to various functional groups means that a single ligand platform can be applied to a wide range of substrates, simplifying inventory management by reducing the need for multiple specialized catalysts. This versatility enhances supply chain resilience, allowing manufacturers to pivot quickly between different product lines without significant retooling or requalification delays.
- Scalability and Environmental Compliance: From a sustainability perspective, this technology aligns perfectly with green chemistry principles. The high atom economy of the hydrogenation reaction, combined with the reduced need for hazardous high-pressure operations, minimizes the environmental footprint of the manufacturing process. The ability to run reactions at lower temperatures and pressures also improves process safety, reducing the risk of thermal runaways or containment failures. As regulatory bodies increasingly scrutinize the environmental impact of pharmaceutical production, adopting such efficient and clean technologies positions companies favorably for future compliance audits. The scalability is proven by the high TON values, indicating that the catalyst remains active even at very low concentrations, which is a key indicator of successful translation from lab bench to commercial plant.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of what process teams can expect during technology transfer and scale-up activities.
Q: What is the maximum enantioselectivity achieved with this ligand system?
A: The novel 3-substituted chiral spiro aminophosphine ligands described in CN110724164B demonstrate exceptional stereocontrol, achieving enantiomeric excess (ee) values of up to 99% in the asymmetric hydrogenation of beta-arylalkylene malonates.
Q: How does the catalyst loading compare to conventional methods?
A: This technology allows for significantly lower catalyst loadings, ranging from 0.2 mol% down to 0.005 mol%, with turnover numbers (TON) reaching as high as 19,000, vastly outperforming traditional systems that often require over 1 mol% loading.
Q: Are the reaction conditions suitable for industrial scale-up?
A: Yes, the process operates under mild conditions, typically between 0°C and 80°C and hydrogen pressures of 0.1 to 100 atm, avoiding the extreme pressures of 50-100 atm often required by older alpha,beta-unsaturated acid hydrogenation methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Aminophosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory discovery to commercial production requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves smoothly from pilot plant to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of our chiral ligands and intermediates meets the highest industry standards. Our commitment to quality ensures that the high enantioselectivity and activity demonstrated in the patent are faithfully reproduced in our commercial supplies.
We invite you to engage with our technical procurement team to discuss how this advanced catalytic technology can be integrated into your specific process. We offer a Customized Cost-Saving Analysis to help you quantify the potential economic benefits of switching to this low-loading, high-efficiency system. Contact us today to request specific COA data for our available ligand variants and to receive comprehensive route feasibility assessments tailored to your target molecules. Let us help you optimize your synthesis and secure your supply chain with our reliable chiral solutions.
