Safe and Scalable Production of 2,5-Dibromopyridine via Novel Lewis Acid Catalysis
Safe and Scalable Production of 2,5-Dibromopyridine via Novel Lewis Acid Catalysis
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic building blocks that can be manufactured safely and cost-effectively. A significant breakthrough in this domain is detailed in Chinese Patent CN112679420B, which discloses a robust preparation method for 2,5-dibromopyridine, a critical intermediate used in the synthesis of antibiotics, antitumor drugs, and various bioactive molecules. Unlike historical methods that relied on hazardous diazotization reactions, this innovative approach utilizes a two-step bromination strategy starting from readily available 2-hydroxypyridine. The process leverages specific Lewis acid catalysts to achieve exceptional regioselectivity and final product purity exceeding 99.5% through simple recrystallization. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards greener, safer, and more economically viable manufacturing protocols that eliminate the risks associated with unstable diazonium salts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,5-dibromopyridine has been plagued by significant safety and environmental challenges inherent to diazotization chemistry. As illustrated in the traditional reaction pathways, the process typically involves converting an amino-substituted pyridine into a diazonium salt intermediate, which is then subjected to Sandmeyer-type bromination. 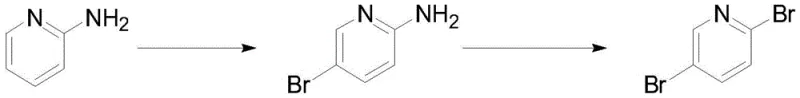 The fundamental flaw in this legacy approach lies in the intrinsic instability of the diazonium species; these intermediates are highly energetic and prone to violent decomposition or explosion when exposed to heat, light, or mechanical shock. Furthermore, the post-treatment of diazotization reactions generates substantial volumes of acidic wastewater containing nitrogenous byproducts, creating a heavy burden on waste treatment facilities and increasing the overall environmental footprint. From a supply chain perspective, the necessity for strict temperature control and specialized safety equipment to handle explosive intermediates often limits batch sizes and complicates the commercial scale-up of complex pharmaceutical intermediates, leading to higher production costs and potential supply disruptions.
The fundamental flaw in this legacy approach lies in the intrinsic instability of the diazonium species; these intermediates are highly energetic and prone to violent decomposition or explosion when exposed to heat, light, or mechanical shock. Furthermore, the post-treatment of diazotization reactions generates substantial volumes of acidic wastewater containing nitrogenous byproducts, creating a heavy burden on waste treatment facilities and increasing the overall environmental footprint. From a supply chain perspective, the necessity for strict temperature control and specialized safety equipment to handle explosive intermediates often limits batch sizes and complicates the commercial scale-up of complex pharmaceutical intermediates, leading to higher production costs and potential supply disruptions.
The Novel Approach
In stark contrast, the methodology described in patent CN112679420B offers a streamlined and inherently safer alternative that bypasses diazonium chemistry entirely. The new route initiates with the direct electrophilic bromination of 2-hydroxypyridine using reagents such as N-bromosuccinimide (NBS) or dibromohydantoin, selectively installing the first bromine atom at the 5-position. 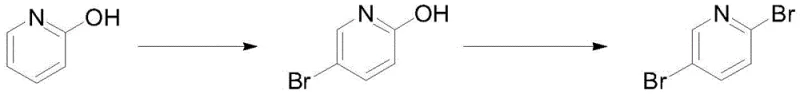 This first step exhibits remarkable regioselectivity, favoring the desired 5-bromo isomer over the 3-bromo isomer with a ratio of approximately 94:6, thereby minimizing the formation of difficult-to-separate impurities. The subsequent step employs a Lewis acid-catalyzed substitution where the hydroxyl group is replaced by a second bromine atom using phosphorus tribromide. This elegant two-step sequence not only simplifies the operational workflow by eliminating the need for intermediate purification but also ensures that the final product can be obtained with high purity (>99.5%) through a straightforward recrystallization process, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
This first step exhibits remarkable regioselectivity, favoring the desired 5-bromo isomer over the 3-bromo isomer with a ratio of approximately 94:6, thereby minimizing the formation of difficult-to-separate impurities. The subsequent step employs a Lewis acid-catalyzed substitution where the hydroxyl group is replaced by a second bromine atom using phosphorus tribromide. This elegant two-step sequence not only simplifies the operational workflow by eliminating the need for intermediate purification but also ensures that the final product can be obtained with high purity (>99.5%) through a straightforward recrystallization process, making it an ideal solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Lewis Acid Catalyzed Hydroxyl Replacement
The core innovation of this synthesis lies in the second transformation, where the hydroxyl group of 2-hydroxy-5-bromopyridine is converted into a bromide. This reaction is facilitated by the synergistic action of phosphorus tribromide (PBr3) and a Lewis acid catalyst, such as tris(pentafluorophenyl)borane (B(C6F5)3) or triphenylborane (BPh3). Mechanistically, the Lewis acid coordinates with the oxygen atom of the pyridone tautomer or assists in activating the phosphorus reagent, thereby lowering the activation energy required for the nucleophilic substitution. The electron-deficient boron center enhances the electrophilicity of the brominating species, allowing the reaction to proceed efficiently under reflux conditions in solvents like acetonitrile or 1,2-dichloroethane. This catalytic cycle ensures that the stoichiometric usage of brominating reagents remains close to theoretical equivalents (1.10-1.30 molar ratio), preventing excessive halogenation or side reactions that could compromise the integrity of the pyridine ring. For process chemists, understanding this mechanism is crucial for optimizing reaction times and ensuring consistent batch-to-batch reproducibility during technology transfer.
Furthermore, the control of impurity profiles is intrinsically linked to the high regioselectivity observed in the initial bromination step. The hydroxyl group on the pyridine ring acts as a strong activating and ortho/para-directing group; however, steric and electronic factors in this specific system favor substitution at the 5-position (para to the nitrogen, meta to the OH in the pyridone form context, but effectively para to the OH in the enol form context relative to the ring numbering). The patent data indicates a product-to-isomer ratio of 94:6, meaning that the vast majority of the material proceeds down the desired pathway. By avoiding rigorous purification after the first step and carrying the crude mixture directly into the Lewis acid catalyzed step, the process capitalizes on the fact that the minor 3-bromo isomer likely reacts differently or is removed during the final recrystallization. This "telescoped" approach reduces solvent consumption and processing time, directly contributing to a leaner manufacturing process that aligns with modern green chemistry principles and supports the production of high-purity pharmaceutical intermediates required for GMP environments.
How to Synthesize 2,5-Dibromopyridine Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and safety. The process begins by dissolving 2-hydroxypyridine in a suitable organic solvent such as acetonitrile or 1,2-dichloroethane, followed by the controlled addition of a brominating agent like NBS at low temperatures (-10 to 0°C) to manage exotherms. After the initial bromination is complete, the reaction mixture is warmed to room temperature, quenched, and the intermediate 2-hydroxy-5-bromopyridine is isolated via filtration or phase separation. This intermediate is then subjected to the second bromination step in the presence of a Lewis acid catalyst and phosphorus tribromide under reflux. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures, are provided in the guide below to assist technical teams in replicating this efficient route.
- React 2-hydroxypyridine with NBS or dibromohydantoin in acetonitrile or DCE at -10 to 25°C to obtain 2-hydroxy-5-bromopyridine with high regioselectivity.
- Quench the first reaction, filter or separate layers to isolate the intermediate without rigorous purification.
- React the intermediate with phosphorus tribromide (PBr3) in the presence of a Lewis acid catalyst like B(C6F5)3 or BPh3 under reflux.
- Recrystallize the crude product from an isopropanol and water mixture to achieve purity greater than 99.5%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic advantages beyond mere technical feasibility. By eliminating the use of unstable diazonium salts, the process removes a major bottleneck related to safety compliance and insurance costs, allowing for more flexible and continuous manufacturing schedules. The reduction in wastewater generation and the avoidance of heavy metal catalysts (often used in cross-coupling alternatives) simplify waste disposal logistics, leading to significant operational cost savings. Moreover, the ability to achieve high purity through simple recrystallization rather than complex chromatography or distillation reduces the consumption of energy and solvents, further driving down the cost of goods sold (COGS). This efficiency makes the supply of 2,5-dibromopyridine more resilient against raw material price fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The elimination of the diazotization step removes the need for expensive safety infrastructure and specialized handling protocols required for explosive intermediates. Additionally, the high regioselectivity of the first step means that less material is lost to isomeric byproducts, improving overall atom economy. The process avoids the use of precious metal catalysts, relying instead on commercially available Lewis acids and phosphorus reagents, which significantly lowers raw material costs. The simplified purification strategy, requiring only a single recrystallization to reach >99.5% purity, drastically reduces solvent usage and processing time, resulting in substantial cost savings compared to multi-step purification sequences.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity; by removing the risk of diazonium salt decomposition and explosion, the manufacturing process becomes inherently more stable and predictable. This stability allows for larger batch sizes and longer campaign runs without the frequent shutdowns associated with high-risk chemical operations. The raw materials, including 2-hydroxypyridine and common brominating agents, are commodity chemicals with robust global supply chains, reducing the risk of shortages. Consequently, suppliers utilizing this technology can offer more reliable lead times and consistent availability of high-purity pharmaceutical intermediates to their downstream clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor materials and conditions (reflux in common solvents) that are easily transferable from kilogram to tonne scale. The significant reduction in wastewater volume and the absence of heavy metal contaminants simplify effluent treatment, ensuring compliance with increasingly stringent environmental regulations. The use of recyclable solvents like acetonitrile and 1,2-dichloroethane further enhances the sustainability profile of the manufacturing process. This alignment with green chemistry principles not only mitigates regulatory risk but also appeals to end-users who prioritize sustainable sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,5-dibromopyridine using this patented methodology. These answers are derived directly from the experimental data and advantageous effects reported in patent CN112679420B, providing clarity on safety, purity, and process parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines.
Q: Why is this new method safer than traditional diazotization routes?
A: Traditional methods rely on unstable diazonium salts which pose significant explosion risks under heat or light. This novel patent CN112679420B eliminates diazotization entirely, using stable brominating reagents and Lewis acid catalysis, drastically improving operational safety for industrial scale-up.
Q: What purity levels can be achieved with this synthesis route?
A: The process is designed for high efficiency, yielding a final product with purity exceeding 99.5% after a single recrystallization step. The first step also demonstrates high regioselectivity (94:6 ratio), minimizing difficult-to-remove isomers early in the process.
Q: Which Lewis acid catalysts are recommended for the second bromination step?
A: The patent specifies the use of tris(pentafluorophenyl)borane or triphenylborane as effective Lewis acid catalysts. These catalysts facilitate the replacement of the hydroxyl group with bromine using phosphorus tribromide under reflux conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Dibromopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics and agrochemicals. Our technical team has extensively analyzed the pathway described in CN112679420B and possesses the expertise to implement this safe, Lewis acid-catalyzed route on a commercial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, including the 3-bromo isomer, guaranteeing that every batch of 2,5-dibromopyridine delivered adheres to the highest industry standards for pharmaceutical applications.
We invite R&D directors and procurement specialists to collaborate with us to optimize their supply chains for pyridine derivatives. By leveraging our advanced manufacturing capabilities and commitment to process safety, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your projects proceed with the most reliable and cost-effective raw materials available in the market.
