Advanced Silver-Mediated Synthesis of 4-Substituted Thiophenyl Isoquinoline-1(2H)-ketone Intermediates
Advanced Silver-Mediated Synthesis of 4-Substituted Thiophenyl Isoquinoline-1(2H)-ketone Intermediates
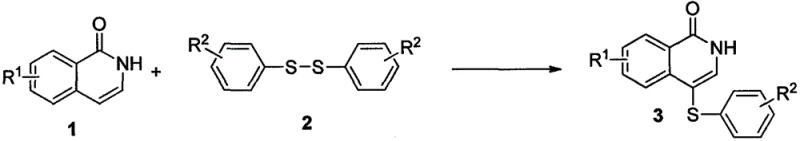
In the rapidly evolving landscape of pharmaceutical development, the efficient construction of nitrogen-containing heterocycles remains a critical bottleneck for R&D teams worldwide. Patent CN108822035B introduces a transformative approach to synthesizing 4-substituted thiophenyl isoquinoline-1(2H)-ketone compounds, a structural motif prevalent in bioactive molecules with anticancer and other therapeutic properties. This technology leverages a silver hexafluoroantimonate-mediated oxidative coupling strategy that fundamentally shifts the paradigm from multi-step, hazard-prone sequences to a streamlined, one-pot operation. By directly utilizing readily available isoquinoline-1(2H)-ketones and diaryl disulfides, this method addresses long-standing challenges in regioselectivity and operational complexity. For global procurement and supply chain leaders, this innovation represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-quality scaffolds with improved consistency and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-thioisoquinoline-1(2H)-ketone derivatives has relied on arduous multi-step pathways that pose significant safety and efficiency risks. The traditional route typically involves the initial bromination of the isoquinoline core to generate a 4-bromo intermediate, followed by low-temperature lithiation using highly reactive reagents such as methyllithium or butyllithium. These organometallic steps demand stringent anhydrous and oxygen-free conditions, often requiring cryogenic temperatures that escalate energy consumption and equipment costs. Furthermore, the subsequent reaction with disulfides to install the thioether group adds another layer of complexity, resulting in a four-step sequence with poor atom economy. This fragmented approach not only generates substantial chemical waste but also introduces multiple purification points where yield losses accumulate, making it economically unviable for cost reduction in API manufacturing on a commercial scale.
The Novel Approach
In stark contrast, the methodology disclosed in CN108822035B offers a direct, catalytic solution that bypasses the need for pre-functionalization or hazardous organometallics. By employing silver hexafluoroantimonate as a mediator, the process facilitates a direct C-H functionalization at the C4 position of the isoquinoline ring. This oxidative coupling occurs efficiently in a single pot using simple aprotic solvents like 1,2-dichloroethane, with reaction temperatures ranging moderately from 60°C to 140°C. The elimination of cryogenic steps and the tolerance for ambient atmospheric conditions drastically simplify the operational protocol. This streamlined workflow not only enhances the overall yield—reported to reach up to 98% in specific embodiments—but also significantly shortens the production cycle, thereby reducing lead time for high-purity pharmaceutical intermediates and enabling faster time-to-market for downstream drug candidates.
Mechanistic Insights into Silver-Mediated Oxidative Coupling
The core of this technological breakthrough lies in the unique ability of silver hexafluoroantimonate to activate the C-H bond at the C4 position of the isoquinoline-1(2H)-one scaffold. Unlike traditional electrophilic substitution which often lacks regiocontrol, the silver mediator likely coordinates with the nitrogen atom or the carbonyl oxygen, directing the activation specifically to the C4 site. This coordination creates a transient organosilver species that undergoes oxidative coupling with the diaryl disulfide. The disulfide bond is cleaved in situ, allowing the arylthio group to attach directly to the heterocyclic core while releasing the corresponding thiolate byproduct. This mechanism avoids the formation of unstable carbanions associated with lithiation, thereby minimizing side reactions such as over-lithiation or nucleophilic attack on the carbonyl group. The result is a highly selective transformation that preserves the integrity of sensitive functional groups on both the isoquinoline and the disulfide partners.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity isoquinoline derivatives. Conventional lithiation routes are prone to generating halogenated impurities from incomplete bromination or metal-contaminated byproducts from quenching steps. The silver-mediated pathway, operating under milder thermal conditions without strong bases, inherently suppresses these degradation pathways. The reaction profile is cleaner, with fewer side products formed, which simplifies the downstream purification process. Flash column chromatography using standard petroleum ether and ethyl acetate mixtures is often sufficient to isolate the target compound in high purity. This robustness in impurity management is critical for meeting the stringent quality specifications required by regulatory bodies for clinical-grade materials, ensuring that the final product is suitable for immediate use in medicinal chemistry campaigns.
How to Synthesize 4-Substituted Thiophenyl Isoquinoline-1(2H)-ketone Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The procedure involves simply mixing the isoquinolinone substrate, the chosen diaryl disulfide, and the silver catalyst in the solvent at room temperature before heating. This user-friendly protocol minimizes the need for specialized training or complex equipment setups. Detailed standardized synthesis steps, including precise molar ratios and workup procedures validated across multiple substrate examples, are outlined in the guide below to ensure reproducibility and optimal outcomes for your specific application.
- Mix isoquinoline-1(2H)-one, diaryl disulfide, and silver hexafluoroantimonate in an aprotic solvent like dichloroethane.
- Heat the reaction mixture to a temperature between 60°C and 140°C and maintain for 6 to 12 hours.
- Upon completion, concentrate the mixture under reduced pressure and purify the crude product via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silver-mediated technology translates into tangible strategic benefits beyond mere chemical efficiency. The shift from a four-step hazardous process to a one-pot reaction fundamentally alters the cost structure and risk profile of producing these valuable intermediates. By removing the dependency on cryogenic infrastructure and pyrophoric reagents, facilities can operate with greater flexibility and lower overhead. This operational simplicity directly contributes to cost reduction in API manufacturing by lowering utility costs, reducing waste disposal fees, and minimizing the capital expenditure required for specialized containment systems. Furthermore, the use of commodity chemicals like dichloroethane and commercially available disulfides ensures a stable raw material supply chain that is less susceptible to market volatility compared to custom-synthesized organolithium reagents.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences significantly lowers the cumulative cost of goods sold (COGS). By consolidating bromination, lithiation, and coupling into a single unit operation, manufacturers save on labor, solvent usage, and energy consumption associated with heating and cooling cycles. The high atom economy of the direct coupling means less raw material is wasted as byproduct, further enhancing the economic viability of the process. Additionally, the avoidance of expensive and hazardous reagents like butyllithium removes a major cost driver and safety liability from the production budget, allowing for more competitive pricing structures in the final supply agreement.
- Enhanced Supply Chain Reliability: Sourcing reliability is paramount for maintaining continuous production schedules. This method utilizes widely available starting materials that are not subject to the same supply constraints as specialized organometallics. The robustness of the reaction conditions, which do not require strict exclusion of air or moisture, reduces the risk of batch failures due to environmental factors. This resilience ensures consistent output and dependable delivery timelines, mitigating the risk of production stoppages that can ripple through the entire pharmaceutical supply network. Partners can rely on a steady flow of high-purity pharmaceutical intermediates without the bottlenecks typical of complex, multi-step synthetic routes.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this technology is inherently scalable due to its mild conditions. The absence of exothermic lithiation steps reduces the thermal load on reactors, making the transition from gram-scale to ton-scale production smoother and safer. From an environmental standpoint, the reduction in waste generation aligns with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. The simplified workup and purification processes also reduce the volume of solvent waste, contributing to a more sustainable manufacturing footprint that appeals to environmentally conscious stakeholders and regulatory agencies alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details helps R&D and procurement teams make informed decisions about integrating this technology into their existing workflows.
Q: What are the advantages of this silver-mediated method over traditional lithiation?
A: This method eliminates the need for hazardous organolithium reagents, cryogenic conditions, and multi-step bromination, offering a safer, one-pot alternative with higher atom economy.
Q: Does this process require strict anhydrous or oxygen-free conditions?
A: No, unlike conventional methods requiring rigorous exclusion of air and moisture, this protocol operates under simple conditions without the need for specialized anhydrous or oxygen-free environments.
Q: What is the substrate scope for this reaction?
A: The method demonstrates wide adaptability, successfully accommodating various substituents on both the isoquinoline ring (e.g., chloro, bromo, methyl) and the disulfide partner (e.g., fluoro, methoxy, cyano).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Substituted Thiophenyl Isoquinoline-1(2H)-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in accelerating drug discovery and development. Our team of expert chemists has extensively evaluated the silver-mediated oxidative coupling technology described in CN108822035B and possesses the technical expertise to implement it effectively. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-substituted thiophenyl isoquinoline-1(2H)-ketone meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis route for your next project. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to efficiency and quality. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your target molecules, ensuring a secure and cost-effective supply of these essential heterocyclic building blocks.
