Advanced Synthetic Route for Alpha-Diacetoxymethyl Nitrogen Heterocycles via Oxidative Ring Contraction
The pharmaceutical and fine chemical industries are constantly seeking efficient methodologies to construct complex nitrogen-containing heterocycles, which serve as pivotal scaffolds in drug discovery and material science. Patent CN107474001B introduces a groundbreaking synthetic strategy for preparing α-diacetoxymethyl-substituted nitrogen-containing heterocyclic compounds. This technology leverages a tandem sequence involving oxidation, ring contraction, and diacetate esterification, transforming readily available N-aryl-substituted nitrogen heterocycles into valuable protected aldehyde equivalents. The significance of this invention lies in its ability to streamline the synthesis of pyrrolidine and piperidine derivatives, which are ubiquitous in clinical drugs and natural products. By utilizing a copper-catalyzed system under an oxygen atmosphere, this method bypasses the need for harsh reagents and multi-step protection-deprotection sequences, offering a robust pathway for generating high-purity pharmaceutical intermediates.
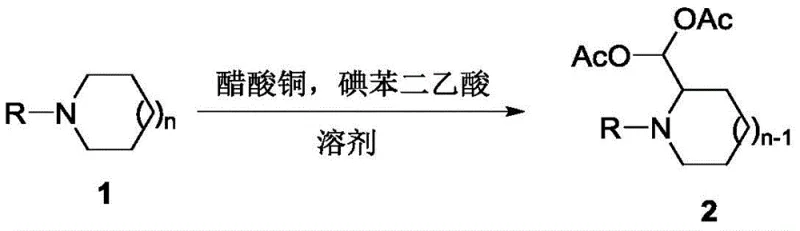
Traditionally, the synthesis of α-functionalized nitrogen heterocycles often involves laborious multi-step procedures that suffer from low atom economy and generate substantial waste. Conventional routes typically require the pre-functionalization of the heterocyclic ring followed by separate protection steps for aldehyde groups, often utilizing unstable intermediates that complicate purification and scale-up. These legacy methods frequently rely on stoichiometric amounts of expensive oxidants or toxic heavy metals, posing significant challenges for environmental compliance and cost management in large-scale manufacturing. Furthermore, the instability of free aldehyde forms in these structures necessitates additional protection steps, extending lead times and increasing the overall cost of goods sold. The limitations of these prior art methods create a bottleneck for R&D teams aiming to rapidly access diverse libraries of heterocyclic building blocks for structure-activity relationship studies.
In stark contrast, the novel approach detailed in CN107474001B revolutionizes this landscape by integrating oxidation and ring contraction into a single operational step. This method employs a catalytic system comprising copper acetate and iodobenzene diacetate (PhI(OAc)2) under a mild oxygen atmosphere, facilitating a direct transformation of N-aryl piperidines or azepanes into contracted pyrrolidine or cyclohexane derivatives bearing diacetoxymethyl groups. The reaction proceeds efficiently in common solvents like acetonitrile at temperatures ranging from 50°C to 100°C, with optimal results observed at 80°C. This one-pot protocol not only drastically reduces the number of unit operations but also enhances the overall yield and purity of the final product. The ability to directly install the diacetoxymethyl protecting group during the ring contraction process eliminates the need for separate aldehyde protection steps, representing a significant leap forward in process efficiency and sustainability for fine chemical manufacturing.
Mechanistic Insights into Copper-Catalyzed Oxidative Ring Contraction
The core of this transformative chemistry lies in the synergistic interaction between the copper catalyst and the hypervalent iodine oxidant. The mechanism initiates with the activation of the alpha-position of the nitrogen heterocycle by the copper species, likely forming a reactive radical or cationic intermediate. Subsequent oxidation by PhI(OAc)2 facilitates the cleavage of the carbon-carbon bond adjacent to the nitrogen, driving the thermodynamic favorability of the ring contraction from a six-membered piperidine to a five-membered pyrrolidine, or from a seven-membered azepane to a six-membered cyclohexane ring. This skeletal rearrangement is concomitant with the installation of two acetoxy groups, effectively trapping the transient aldehyde species as a stable diacetate. The presence of molecular oxygen serves as a terminal oxidant, regenerating the active copper species and ensuring the catalytic cycle continues efficiently without the accumulation of reduced metal byproducts. This elegant mechanistic pathway ensures high selectivity and minimizes the formation of over-oxidized side products.
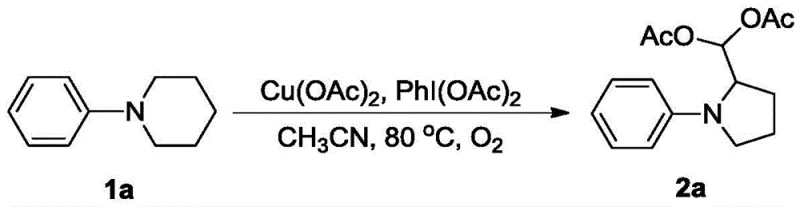
From an impurity control perspective, this mechanism offers distinct advantages due to the stability of the diacetate moiety under the reaction conditions. Unlike free aldehydes which are prone to polymerization or unwanted nucleophilic attacks, the diacetoxymethyl group remains inert throughout the reaction and workup, leading to cleaner crude profiles. The use of copper acetate, a relatively benign transition metal salt, avoids the introduction of difficult-to-remove heavy metal contaminants often associated with palladium or rhodium catalysis. Furthermore, the reaction tolerates a wide range of functional groups on the N-aryl ring, including halogens (fluoro, chloro, bromo) and electron-donating groups (methyl, methoxy), without significant interference in the oxidative cycle. This robustness ensures that the impurity profile remains manageable even with diverse substrate inputs, simplifying downstream purification processes such as silica gel chromatography and crystallization.
How to Synthesize Alpha-Diacetoxymethyl Heterocycles Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins by dissolving the N-aryl substituted nitrogen heterocycle starting material in a polar aprotic solvent, with acetonitrile being the preferred choice for optimal solubility and reaction kinetics. To this solution, stoichiometric or near-stoichiometric amounts of copper acetate and iodobenzene diacetate are added. The reaction vessel is then subjected to an oxygen atmosphere, typically maintained at 1 to 2 atmospheres of pressure, and heated to facilitate the transformation. Following the completion of the reaction, indicated by TLC or HPLC monitoring, the mixture is quenched with aqueous brine and extracted with organic solvents like ethyl acetate. The combined organic layers are dried and concentrated, yielding the crude product which can be purified via flash column chromatography to afford the target α-diacetoxymethyl substituted heterocycle in high purity. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve the N-aryl substituted nitrogen-containing heterocyclic compound (e.g., N-phenylpiperidine) in a suitable solvent such as acetonitrile.
- Add copper acetate and iodobenzene diacetate to the reaction mixture under an oxygen atmosphere (1-2 atm).
- Heat the mixture to 50-100°C (optimally 80°C) and stir for approximately 12 hours to complete the oxidation and ring contraction.
- Quench the reaction with saturated brine, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents compelling economic and logistical benefits. The primary advantage stems from the significant reduction in process complexity, which directly translates to lower manufacturing costs. By consolidating multiple synthetic steps into a single pot, the method reduces solvent consumption, energy usage, and labor hours associated with intermediate isolation and purification. The reliance on commodity chemicals such as copper acetate and iodobenzene diacetate, rather than exotic or proprietary catalysts, ensures a stable and cost-effective supply of raw materials. This accessibility mitigates the risk of supply chain disruptions often caused by the scarcity of specialized reagents, thereby enhancing the reliability of production schedules and delivery timelines for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of separate protection and deprotection steps for the aldehyde functionality results in substantial savings in reagent costs and waste disposal fees. Traditional methods often require expensive silyl or acetal protecting groups and additional acidic or basic hydrolysis steps, all of which add to the overall cost base. In this novel process, the diacetate group is installed concurrently with the ring contraction, effectively performing two chemical transformations for the price of one. Furthermore, the use of oxygen as a co-oxidant is inherently economical compared to stoichiometric organic oxidants, reducing the material cost per kilogram of product. The simplified workup procedure also minimizes the volume of organic waste generated, aligning with green chemistry principles and reducing environmental compliance costs.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, specifically N-aryl piperidines and azepanes, are widely available from global chemical suppliers and are produced at large scales for various industries. This abundance ensures that raw material sourcing is not a bottleneck, allowing for flexible procurement strategies and bulk purchasing opportunities. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality or environmental factors, leading to consistent batch-to-batch reproducibility. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts and ensuring that project timelines are met without delay due to synthetic failures or yield fluctuations.
- Scalability and Environmental Compliance: The reaction operates under mild thermal conditions (80°C) and moderate oxygen pressure, which are easily manageable in standard stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. This ease of scale-up facilitates the transition from gram-scale R&D to ton-scale commercial production with minimal process re-engineering. Additionally, the copper catalyst used is less toxic than many alternative transition metals, simplifying the regulatory burden associated with heavy metal residue limits in final drug substances. The overall greener profile of the synthesis, characterized by higher atom economy and reduced solvent intensity, supports corporate sustainability goals and helps meet increasingly stringent environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative ring contraction technology. These insights are derived directly from the experimental data and scope defined in patent CN107474001B, providing clarity on reaction parameters, substrate compatibility, and product isolation. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific target molecules and for procurement specialists assessing the long-term viability of the supply chain.
Q: What are the key advantages of this oxidative ring contraction method?
A: The method described in patent CN107474001B offers a one-pot synthesis that combines oxidation, ring contraction, and esterification. It utilizes readily available reagents like copper acetate and operates under mild conditions (80°C), significantly simplifying the process compared to traditional multi-step protections.
Q: What is the substrate scope for this reaction?
A: The reaction demonstrates broad substrate tolerance, accommodating various N-aryl substituents including electron-withdrawing groups (fluoro, chloro, bromo, nitro) and electron-donating groups (alkyl, methoxy). It is effective for both six-membered (piperidine) and seven-membered (azepane) starting rings.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process uses common solvents like acetonitrile and inexpensive copper catalysts. The reaction conditions are mild (80°C, 1 atm O2), and the workup involves standard extraction and column chromatography, making it highly scalable for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Diacetoxymethyl Compounds Supplier
The technological advancements presented in CN107474001B underscore the potential for more efficient and sustainable production of complex nitrogen heterocycles. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of alpha-diacetoxymethyl substituted compounds meets the exacting standards required by the global pharmaceutical industry. We understand the critical nature of these intermediates in drug development and are dedicated to providing a seamless supply experience.
We invite you to collaborate with us to leverage this advanced synthetic capability for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our reliable alpha-diacetoxymethyl compounds supply solutions.
