Scalable Synthesis of 2-Methyl-3-Trifluoromethyl Aniline for Global Pharmaceutical Supply Chains
The global demand for high-purity veterinary pharmaceutical intermediates continues to surge, driven by the expanding livestock healthcare market. At the forefront of this sector is Flunixin Meglumine, a potent non-steroidal anti-inflammatory drug (NSAID) widely utilized for its antipyretic and analgesic properties in large animals. The synthesis of its key precursor, 2-methyl-3-trifluoromethyl aniline, has historically presented significant challenges regarding safety, cost, and scalability. Recent intellectual property developments, specifically patent CN114890902B, have introduced a transformative two-step preparation method that addresses these longstanding industrial pain points. This innovative approach replaces hazardous lithiation chemistry with a safer electrophilic substitution followed by a robust reductive protocol, marking a substantial leap forward in process chemistry. For procurement leaders and R&D directors, understanding the nuances of this patented route is essential for securing a reliable supply chain for this critical building block.
Furthermore, the strategic implementation of this synthesis pathway offers a compelling value proposition for manufacturers seeking to optimize their production lines. By shifting away from expensive and dangerous reagents like butyl lithium and methyl iodide, the new method significantly lowers the barrier to entry for high-volume production. The utilization of readily available raw materials such as dimethyl sulfide and chlorine gas, coupled with a highly efficient activated zinc alloy reduction, ensures that the process is not only chemically elegant but also commercially viable. As we delve deeper into the technical specifics, it becomes clear that this methodology represents a paradigm shift in how complex fluorinated anilines are manufactured, promising enhanced stability and consistency for the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-3-trifluoromethyl aniline has been plagued by severe operational hazards and economic inefficiencies. Traditional routes, such as those disclosed in U.S. Pat. No. 6,596,573, heavily rely on the use of butyl lithium for ortho-lithiation. This reagent is pyrophoric, requiring stringent handling protocols and specialized equipment to prevent catastrophic fires. Moreover, the reaction must be conducted at cryogenic temperatures, often below -70°C, which imposes a massive energy burden on the manufacturing facility and limits the feasible batch size due to heat transfer constraints. Additionally, the subsequent methylation step typically employs toxic agents like methyl iodide or dimethyl sulfate, introducing significant environmental and occupational health risks that complicate waste disposal and regulatory compliance.
Alternative pathways, such as the four-step synthesis described in Chinese patent CN103570558, suffer from excessive linearity and poor atom economy. Each additional step in a synthetic sequence inherently accumulates yield losses and increases the accumulation of impurities, necessitating complex purification procedures that drive up costs. Other fluorination-based routes, like those in CN102491906, demand highly specialized reaction vessels capable of withstanding aggressive fluorinating conditions, leading to high capital expenditure. The resulting product mixtures from these older methods often require difficult separations, further eroding the overall process yield. Consequently, these conventional methods are ill-suited for modern, large-scale industrial production where safety, cost-efficiency, and environmental stewardship are paramount.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114890902B introduces a streamlined two-step sequence that elegantly circumvents these obstacles. The first step involves a controlled electrophilic substitution where chlorine gas is introduced into a system of dimethyl sulfide and solvent at a moderate low temperature range of -15°C to -30°C. This generates a reactive species in situ that functionalizes the aniline derivative without the need for cryogenic cooling or pyrophoric bases. The subsequent heating phase allows for the formation of the intermediate sulfide compound with high conversion rates. This shift from lithiation to electrophilic chloromethylation fundamentally alters the risk profile of the operation, making it accessible to standard chemical manufacturing infrastructure.
The second step leverages a specialized activated zinc alloy for reductive dehalogenation and desulfurization. Unlike traditional catalytic hydrogenation which may require high-pressure equipment, this zinc-mediated reduction proceeds under atmospheric pressure at reflux temperatures (70-90°C). The activation of the zinc surface, achieved through a specific pretreatment with metal ligands like copper sulfate, dramatically enhances its reactivity, allowing for the simultaneous removal of the chlorine atom and the reduction of the sulfide group to a methyl group. This tandem transformation is highly efficient, delivering the target 2-methyl-3-trifluoromethyl aniline in excellent yields exceeding 90% in optimized embodiments. The result is a process that is not only safer and cheaper but also remarkably robust, ensuring consistent quality output suitable for GMP manufacturing environments.
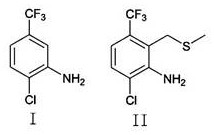
Mechanistic Insights into DMS-Chlorine Complex Functionalization and Zinc Reduction
The core innovation of this synthesis lies in the generation and utilization of a chloromethyl sulfonium species derived from dimethyl sulfide (DMS) and chlorine. In the first stage, the interaction between DMS and chlorine gas at low temperatures creates a potent electrophile. When the starting material, a chloro-trifluoromethyl aniline derivative (Compound I), is introduced along with an organic base like triethylamine, this electrophile selectively attacks the position ortho to the amino group. The amino group acts as a directing group, facilitating the installation of the -CH2-S-Me moiety. This step is critical because it installs the carbon framework necessary for the final methyl group while avoiding the harsh conditions associated with direct Friedel-Crafts alkylation on deactivated rings. The presence of the trifluoromethyl group, which is strongly electron-withdrawing, typically deactivates the ring towards electrophilic attack, making the mild yet effective nature of this DMS-Cl2 system particularly noteworthy for maintaining high reaction rates without degradation.
Following the isolation of the intermediate sulfide (Compound II), the process moves to the reductive phase. The use of activated zinc alloy in a protic solvent system (isopropanol) with acetic acid creates a dissolving metal reduction environment. The activation of the zinc powder, involving treatment with hydrochloric acid and a metal ligand such as copper sulfate, creates a galvanic cell effect on the particle surface. This significantly lowers the activation energy for electron transfer. In this environment, the zinc serves as a two-electron donor. It first facilitates the reductive dehalogenation, cleaving the carbon-chlorine bond to remove the halogen substituent. Simultaneously, it effects the desulfurization of the -CH2-S-Me group, cleaving the carbon-sulfur bonds and replacing them with carbon-hydrogen bonds to yield the final methyl group. This dual-reduction capability is mechanistically distinct from simple catalytic hydrogenation and offers superior chemoselectivity, preserving the sensitive trifluoromethyl group which might otherwise be susceptible to hydrodefluorination under more vigorous reducing conditions.
How to Synthesize 2-Methyl-3-Trifluoromethyl Aniline Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the careful generation of the reactive chlorinating species, followed by the controlled addition of the substrate to manage exotherms. The subsequent reduction step relies heavily on the quality of the activated zinc alloy, which must be prepared immediately prior to use to ensure maximum surface activity. Detailed operational procedures, including specific stoichiometry, temperature ramps, and workup protocols, are essential for reproducing the high yields reported in the patent literature. For a comprehensive guide on the exact laboratory and pilot-scale procedures, please refer to the standardized synthesis steps provided below.
- Prepare a reaction system with solvent and dimethyl sulfide, introduce chlorine gas at low temperature (-15°C to -30°C), then add the starting aniline derivative and organic base, heating to 40-80°C to form the intermediate sulfide.
- In a separate reactor, combine the intermediate sulfide with solvent and activated zinc alloy, heat to reflux (70-90°C), and slowly add acetic acid to effect reduction and obtain the final aniline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into tangible operational benefits and risk mitigation. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for butyl lithium, methyl iodide, and specialized fluorination reagents, the supply chain becomes far more resilient. These traditional reagents are often subject to volatile pricing and strict regulatory controls due to their toxicity and hazard profiles. In contrast, dimethyl sulfide, chlorine gas, and zinc powder are commodity chemicals with stable, global supply networks. This shift ensures a continuous flow of materials, reducing the likelihood of production stoppages caused by raw material shortages or logistics bottlenecks associated with hazardous cargo transport.
- Cost Reduction in Manufacturing: The economic implications of this process are profound. The elimination of cryogenic cooling requirements represents a massive saving in energy costs. Maintaining reactors at -70°C for extended periods is energy-intensive and requires specialized refrigeration infrastructure. By operating the first step at -15°C to -30°C and the second step at reflux (70-90°C), the process utilizes standard heating and cooling utilities, drastically lowering utility expenditures. Furthermore, the high yield of the reaction minimizes the loss of expensive fluorinated starting materials. The avoidance of precious metal catalysts or complex high-pressure hydrogenation equipment also reduces capital depreciation and maintenance costs, leading to a lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain reliability. Facilities that handle pyrophoric reagents like butyl lithium face higher insurance premiums and stricter regulatory scrutiny, which can lead to unplanned shutdowns during inspections or incidents. This new method operates with a significantly improved safety profile, utilizing reagents that are easier to store and handle. The reduced hazard level allows for larger batch sizes and more flexible scheduling, enabling manufacturers to respond more agilely to market demand fluctuations. The robustness of the zinc reduction step also means fewer failed batches due to catalyst poisoning or sensitivity, ensuring a steady and predictable output of high-purity product for downstream customers.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers a cleaner alternative to traditional methods. The waste stream primarily consists of zinc salts and organic solvents, which are easier to treat and dispose of compared to the lithium salts and toxic sulfur byproducts generated by other routes. The absence of toxic methylating agents like dimethyl sulfate removes a major carcinogenic hazard from the plant floor. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the sustainability profile of the supply chain. The process is inherently scalable, as demonstrated by the successful transition from gram-scale experiments to multi-kilogram batches in the patent examples, proving its readiness for commercial tonnage production without the need for exotic engineering solutions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-methyl-3-trifluoromethyl aniline. These answers are derived directly from the experimental data and technical disclosures found in the relevant patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their broader manufacturing strategies.
Q: Why is the activated zinc alloy method superior to traditional Raney Nickel reduction?
A: The patent data indicates that using activated zinc alloy results in significantly higher yields compared to Raney Nickel. Furthermore, the zinc-based protocol operates under milder conditions with shorter reaction times and reduced solvent usage, enhancing overall process efficiency and safety.
Q: What are the safety advantages of this new route over butyl lithium methods?
A: Traditional routes often rely on butyl lithium, which requires cryogenic temperatures and poses severe fire risks. This novel method utilizes dimethyl sulfide and chlorine gas at manageable low temperatures (-15°C to -30°C), eliminating the need for extreme cooling and hazardous pyrophoric reagents.
Q: How does this process impact the purity of the final Flunixin Meglumine intermediate?
A: The optimized reaction conditions, particularly the controlled temperature profile and the specificity of the activated zinc reduction, minimize side reactions. Experimental data from the patent demonstrates product purities exceeding 99.0%, which is critical for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-3-Trifluoromethyl Aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the availability of high-quality intermediates is the backbone of a successful pharmaceutical supply chain. Our technical team has thoroughly analyzed the advancements presented in patent CN114890902B and possesses the expertise to implement this superior synthesis route at scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your demand for Flunixin Meglumine precursors is met with unwavering consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-methyl-3-trifluoromethyl aniline meets the exacting standards required for veterinary drug formulation.
We invite you to collaborate with us to leverage these technological improvements for your business. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and reduce your overall procurement costs.
