Scalable Synthesis of N-Aryl-Heterocyclic Compounds Using Stable Supported Copper Catalysts
The landscape of C-N bond formation has long been dominated by palladium-catalyzed methodologies, yet the industry constantly seeks more economical and robust alternatives for large-scale manufacturing. Patent CN102382058A introduces a transformative approach to synthesizing N-aryl-heterocyclic nitrogen compounds by leveraging a loaded Cu(II) salt catalyst. This technology addresses critical pain points in fine chemical production by replacing expensive precious metals with stable, low-cost copper species supported on carriers like alumina or silica gel. The innovation lies not just in the catalyst composition, but in the operational simplicity, allowing reactions to proceed at temperatures between 110°C and 150°C with exceptional yields. For R&D directors and procurement specialists alike, this represents a pivotal shift towards sustainable and cost-efficient process chemistry that does not compromise on purity or conversion efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Ullmann-type coupling reactions often rely heavily on homogeneous copper salts or precious metal catalysts like palladium, which present significant logistical and financial hurdles for industrial application. Palladium catalysts, while effective, necessitate stringent anhydrous and oxygen-free conditions to maintain activity, thereby increasing the complexity of reactor setup and inert gas consumption. Furthermore, the high cost of palladium and the difficulty in removing trace metal residues from the final active pharmaceutical ingredient (API) create substantial downstream purification burdens. Even earlier generations of copper-catalyzed systems suffered from poor catalytic activity unless harsh conditions or complex ligand systems were employed, leading to inconsistent batch-to-batch reproducibility and lower overall throughput in commercial settings.
The Novel Approach
The methodology described in the patent circumvents these issues by utilizing a heterogeneous, loaded Cu(II) salt catalyst that combines high stability with ease of separation. By supporting copper salts such as cupric chloride or copper sulfate on carriers like aluminum oxide or natural manganese dioxide, the catalyst achieves a dispersed active phase that maximizes surface area and reactivity. This novel approach allows the reaction to tolerate a broader range of conditions without the extreme sensitivity associated with Pd-catalysis. The result is a streamlined process where the catalyst can be easily managed, and the reaction proceeds with high efficiency, as evidenced by the successful synthesis of diverse N-aryl derivatives including imidazoles and indoles with yields reaching up to 99%.
Mechanistic Insights into Supported Cu(II)-Catalyzed C-N Coupling
The core of this technological advancement is the interaction between the supported copper species and the nitrogen heterocycle under basic conditions. The loaded catalyst acts as a reservoir of active Cu(II) ions that likely undergo reduction to Cu(I) in situ, facilitating the oxidative addition and reductive elimination cycles typical of Ullmann couplings. The choice of carrier, such as alumina or attapulgite clay (zeyssatite), plays a crucial role in stabilizing the copper species against aggregation, which is a common deactivation pathway in homogeneous systems. This stabilization ensures that the catalytic cycle remains active over extended reaction times, typically ranging from 5 to 23 hours, allowing for complete conversion of even less reactive aryl chlorides or bromides when optimized.
Impurity control is inherently improved through the use of this heterogeneous system, as the solid support minimizes the leaching of copper into the solution phase compared to soluble salts. The reaction mechanism favors the formation of the desired C-N bond over side reactions such as homocoupling of the aryl halide, which is often observed with less selective catalysts. The presence of strong bases like potassium phosphate or potassium carbonate further drives the equilibrium towards product formation by neutralizing the hydrogen halide byproduct. This mechanistic robustness is demonstrated across a wide substrate scope, where electron-withdrawing and electron-donating groups on the aryl ring are well-tolerated, ensuring a clean impurity profile for downstream processing.
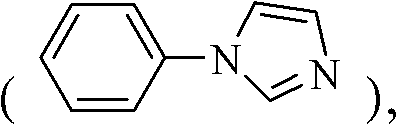
How to Synthesize N-Phenylimidazole Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-purity N-aryl heterocycles suitable for pharmaceutical applications. The process begins with the precise loading of the heterocyclic nitrogen compound, such as imidazole, and the aryl halide into a reactor containing the supported catalyst and solvent. The reaction is conducted under a nitrogen atmosphere to prevent oxidation of sensitive intermediates, although the catalyst itself is air-stable. Following the heating period, the workup involves simple aqueous washing and organic extraction, avoiding complex filtration steps required for removing fine homogeneous catalyst powders. The detailed standardized synthesis steps for replicating this high-yield process are provided in the guide below.
- Load the reactor with the nitrogen heterocycle (e.g., imidazole), alkali base (e.g., K3PO4), and the supported Cu(II) catalyst (e.g., CuCl2/Al2O3) in an organic solvent like DMSO.
- Add the aryl halide (e.g., iodobenzene) under nitrogen protection and heat the mixture to 110-150°C with stirring for 5 to 23 hours.
- Upon completion, cool the reaction, wash with water, extract with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this supported copper catalyst technology translates directly into enhanced margin protection and supply security. The elimination of precious metal palladium removes a major variable cost driver that is subject to volatile global market fluctuations. Additionally, the simplified reaction conditions reduce the energy burden associated with maintaining strict inert atmospheres and cryogenic temperatures, leading to lower utility costs per kilogram of product. The robustness of the catalyst also implies a longer shelf life and easier storage requirements, reducing waste associated with catalyst degradation prior to use.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with low-cost copper salts supported on abundant minerals like alumina drastically reduces the raw material cost basis. Since the catalyst is heterogeneous, it potentially allows for recovery and reuse strategies that are difficult to implement with homogeneous systems, further driving down the cost of goods sold (COGS). The high yields reported, such as 99% for N-phenylimidazole, minimize the loss of valuable starting materials, ensuring maximum atom economy and reducing the cost of waste disposal.
- Enhanced Supply Chain Reliability: The raw materials required for this process, including common aryl halides and nitrogen heterocycles, are commodity chemicals with established global supply chains, reducing the risk of shortages. The catalyst preparation itself uses readily available copper salts and carriers, meaning there is no dependency on specialized, single-source ligand suppliers that often bottleneck production. This decentralization of supply risk ensures consistent production schedules and reliable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (110-150°C) and uses standard solvents like DMSO or DMF, which are well-understood in terms of handling and recycling at scale. The absence of toxic heavy metals like palladium simplifies the environmental compliance landscape, as wastewater treatment does not require specialized heavy metal scavenging resins. This facilitates easier permitting for new production lines and aligns with increasingly stringent green chemistry mandates in the pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this Cu(II) catalyzed coupling technology in a commercial setting. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for process engineers. Understanding these nuances is critical for evaluating the feasibility of technology transfer and scale-up.
Q: What are the advantages of using supported Cu(II) catalysts over traditional Palladium systems?
A: Supported Cu(II) catalysts offer significantly lower material costs compared to precious metal palladium systems. Furthermore, they exhibit higher stability in air and do not require strictly anhydrous or oxygen-free environments, simplifying operational requirements and reducing production costs.
Q: What is the substrate scope for this C-N coupling method?
A: The method is highly versatile, accommodating various nitrogen heterocycles including imidazoles, indoles, carbazoles, and triazoles. It effectively couples with diverse aryl halides such as iodobenzene, bromobenzene, and substituted variants containing trifluoromethyl, nitro, or methoxy groups.
Q: Can this process be scaled for commercial manufacturing?
A: Yes, the process utilizes conventional heating units like oil baths or heating jackets and standard workup procedures (extraction, filtration). The high yields (up to 99%) and simple purification steps make it highly suitable for commercial scale-up from kilogram to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Aryl-Heterocyclic Nitrogen Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting efficient C-N coupling technologies like the one described in CN102382058A for the production of complex pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and risk-mitigated. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace metal residues, guaranteeing that every batch of N-aryl-heterocyclic compound meets the exacting standards required for API synthesis.
We invite you to collaborate with us to leverage this cost-effective synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis comparing this copper-catalyzed method against your current palladium-based processes. Please contact us to request specific COA data and route feasibility assessments tailored to your target molecule, and let us demonstrate how our engineering expertise can optimize your supply chain.
