Advanced Monochiral Ruthenium Catalysts for Scalable Synthesis of Chiral Intermediates
The landscape of asymmetric synthesis in the pharmaceutical industry is constantly evolving, driven by the relentless demand for high-purity chiral intermediates that serve as the backbone for modern drug discovery. A significant breakthrough in this domain is documented in Chinese Patent CN112675920B, which introduces a novel class of monochiral center catalysts designed to overcome the inherent limitations of traditional multi-chiral catalytic systems. This technology represents a paradigm shift towards more atom-economical and cost-effective methodologies for generating chiral secondary alcohols and alpha-allyl alcohols, which are critical building blocks in the production of bioactive molecules. By leveraging a unique ruthenium-based coordination environment that relies on a single point of chirality rather than complex matching of multiple stereogenic elements, this innovation offers a robust pathway for industrial scale-up. For R&D directors and process chemists, understanding the mechanistic nuances and operational advantages of this system is crucial for optimizing synthetic routes and ensuring supply chain resilience for high-value active pharmaceutical ingredients.
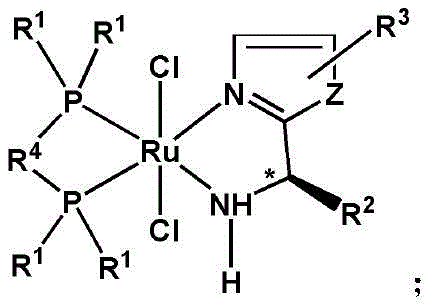
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional asymmetric hydrogenation methods, particularly those inspired by the Nobel-winning Noyori systems, have long been the gold standard for producing chiral alcohols. However, these established methodologies suffer from significant drawbacks that hinder their widespread adoption in cost-sensitive manufacturing environments. The primary limitation lies in the structural complexity of the requisite ligands, which typically demand the precise matching of three distinct chiral elements: axial chirality within the bisphosphine component and two point chiral centers within the diamine ligand. If these elements are not perfectly matched, the stereochemical outcome deteriorates rapidly, leading to poor enantiomeric excess (ee) values. Furthermore, the synthesis of these multifaceted chiral ligands is notoriously difficult, expensive, and time-consuming, creating a substantial bottleneck in the supply chain. The reliance on high-pressure hydrogenation equipment for some variants also introduces safety concerns and capital expenditure barriers, making the transition from laboratory bench to commercial production a risky and resource-intensive endeavor for many fine chemical manufacturers.
The Novel Approach
In stark contrast to the cumbersome requirements of legacy systems, the novel approach detailed in the patent utilizes a streamlined monochiral center catalyst architecture that drastically simplifies the stereochemical control mechanism. By employing a ruthenium center coordinated with a single chiral ligand and an achiral bidentate phosphine ligand, the system eliminates the need for complex chiral matching while maintaining exceptional stereoselectivity. This design philosophy not only reduces the synthetic steps required to prepare the catalyst itself but also enhances its stability and solubility in common organic solvents. The result is a highly efficient catalytic cycle capable of operating under mild conditions, specifically at temperatures ranging from 20°C to 40°C, with reaction times as short as 2 to 15 minutes. This efficiency translates directly into higher throughput and lower energy consumption, addressing the critical pain points of both process economics and operational safety. The ability to achieve high yields and ee values greater than 83% across a diverse range of sterically and electronically similar substrates underscores the versatility and robustness of this new catalytic platform.
Mechanistic Insights into Monochiral Ruthenium-Catalyzed Asymmetric Transfer Hydrogenation
The efficacy of this catalytic system stems from its unique coordination geometry, which facilitates an outer-sphere reaction mechanism similar to classic transfer hydrogenation but with enhanced stereocontrol derived from a single chiral source. The ruthenium metal center acts as the hub for hydride transfer, coordinated by a rigid bidentate phosphine ligand that provides stability and a chiral amine or heterocyclic ligand that dictates the facial selectivity of the substrate approach. Unlike traditional systems where mismatched chirality leads to catastrophic loss of selectivity, this monochiral design ensures that the steric environment around the metal center is sufficiently defined to differentiate between the prochiral faces of the ketone substrate. The presence of bulky substituents on the phosphine ligands, such as tert-butyl or trifluoromethyl groups, further tunes the electronic and steric properties of the catalyst, allowing for fine-tuning of reactivity towards challenging substrates like aryl-aryl ketones where steric differentiation is minimal. This mechanistic robustness allows the catalyst to maintain high turnover frequencies even at low loadings of 0.1 mol% to 1.0 mol%, making it an economically attractive option for large-scale synthesis.
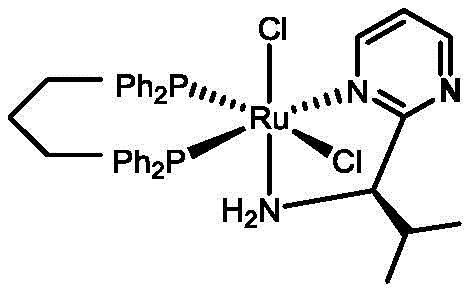
Impurity control is another critical aspect where this mechanism excels, particularly in the context of pharmaceutical manufacturing where regulatory standards are stringent. The high stereoselectivity inherent in the monochiral catalyst design minimizes the formation of the undesired enantiomer, thereby reducing the burden on downstream purification processes such as chiral chromatography or recrystallization. In traditional methods, the presence of closely related diastereomers or enantiomers often necessitates multiple purification steps, which erodes overall yield and increases waste generation. By achieving enantiomeric excess values consistently above 90% for many substrates directly from the reaction mixture, this technology significantly streamlines the purification workflow. Furthermore, the mild reaction conditions prevent the degradation of sensitive functional groups that might be present on complex drug intermediates, ensuring that the impurity profile remains clean and manageable. This level of control is essential for meeting the rigorous quality specifications required for clinical trial materials and commercial API production, providing a reliable foundation for process validation.
How to Synthesize Chiral Alcohols Efficiently
The practical implementation of this technology involves a straightforward two-stage process: catalyst preparation followed by the asymmetric transformation. The synthesis of the catalyst itself is designed for scalability, beginning with the reaction of a ruthenium precursor with a chiral ligand in a non-coordinating solvent like dichloromethane, followed by the introduction of the bidentate phosphine ligand in toluene at elevated temperatures. Once the catalyst is prepared, the asymmetric transfer hydrogenation is conducted by mixing the catalyst, a base such as potassium tert-butoxide, and the ketone substrate in a solvent system comprising isopropanol and dichloromethane. The detailed standardized synthesis steps for preparing the catalyst and executing the hydrogenation reaction are outlined in the guide below, providing a clear roadmap for laboratory and pilot plant execution.
- Prepare the catalyst by reacting tris(triphenylphosphine)ruthenium dichloride with a chiral ligand in dichloromethane, followed by reaction with a bidentate phosphine ligand in toluene.
- Dissolve the prepared catalyst and potassium tert-butoxide in a mixed solvent of isopropanol and dichloromethane under inert atmosphere.
- Add the ketone substrate and stir at 20°C to 40°C for 2 to 15 minutes, then purify the resulting chiral alcohol via filtration and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this monochiral catalyst technology offers compelling strategic advantages that extend beyond mere technical performance. The most significant benefit is the drastic simplification of the catalyst supply chain, as the ligands required are far easier to synthesize than the complex multi-chiral ligands used in conventional systems. This simplification translates into a more stable and predictable supply of catalytic materials, reducing the risk of production delays caused by ligand shortages. Additionally, the ability to operate at room temperature and atmospheric pressure eliminates the need for specialized high-pressure reactors, allowing existing manufacturing infrastructure to be utilized without significant capital investment. This flexibility enhances the agility of the supply chain, enabling faster response times to market demands and reducing the lead time for scaling up new processes from development to commercial production.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the reduced complexity of ligand synthesis and the high efficiency of the catalytic cycle. Since the catalyst requires only a single chiral center, the raw material costs and processing time associated with ligand production are significantly lower compared to traditional bisphosphine-diamine systems. Furthermore, the high turnover number and low catalyst loading mean that less precious metal is required per kilogram of product, directly lowering the bill of materials. The mild reaction conditions also contribute to cost savings by reducing energy consumption for heating and cooling, while the shortened reaction times increase reactor throughput. These factors combine to deliver substantial cost reductions in pharmaceutical intermediate manufacturing without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the robustness and broad substrate scope of the catalyst, which allows for the consolidation of multiple synthetic steps under a single technological platform. The ability to handle diverse substrates, including sterically hindered aryl-aryl ketones and heterocyclic compounds, means that fewer specialized catalysts need to be stocked and managed. This standardization simplifies inventory management and reduces the complexity of vendor qualification processes. Moreover, the stability of the catalyst under ambient conditions facilitates easier storage and transportation, minimizing the risks associated with degradation during logistics. By adopting a versatile catalyst system that performs consistently across a wide range of applications, organizations can build a more resilient and responsive supply chain capable of adapting to changing project requirements.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns perfectly with the principles of green chemistry and sustainable manufacturing. The atom economy of the transfer hydrogenation process is superior to stoichiometric reduction methods, generating less waste and reducing the environmental footprint of the synthesis. The use of isopropanol as a hydrogen source is safer and more environmentally benign than handling high-pressure hydrogen gas, simplifying safety protocols and regulatory compliance. Scalability is inherently supported by the simple operational parameters, which do not require exotic equipment or extreme conditions that are difficult to replicate on a large scale. This ease of scale-up ensures that processes developed in the lab can be transferred to commercial production facilities with minimal technical risk, accelerating the time to market for new drug candidates while maintaining strict environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and operational parameters of the monochiral ruthenium catalyst system. These insights are derived directly from the experimental data and embodiments described in the patent literature, providing a factual basis for evaluating the technology's suitability for specific applications. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: What is the primary advantage of the monochiral center catalyst over traditional Noyori-type systems?
A: The primary advantage lies in the simplified ligand architecture requiring only a single chiral center, which significantly reduces synthetic complexity and cost compared to traditional systems that require matching multiple chiral elements like axial and point chirality.
Q: What types of substrates are compatible with this catalytic system?
A: This system demonstrates broad substrate scope, effectively catalyzing aryl-aryl, heterocyclic aromatic-aryl, and heterocyclic-heterocyclic substituted ketones, as well as facilitating the kinetic resolution of racemic alpha-allyl alcohols.
Q: What are the typical reaction conditions for this asymmetric hydrogenation?
A: The reaction proceeds under mild conditions, typically at room temperature (20°C to 40°C) with short reaction times ranging from 2 to 15 minutes, using low catalyst loadings of 0.1 mol% to 1.0 mol%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ruthenium Catalyst Supplier
As the pharmaceutical industry continues to demand more efficient and sustainable solutions for chiral synthesis, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes a strategic imperative. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be seamlessly translated into reliable commercial supply. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and enantiomeric purity of every batch. By leveraging our expertise in organometallic catalysis and process optimization, we help clients navigate the complexities of chiral intermediate manufacturing, delivering cost-effective solutions that meet the highest industry standards.
We invite you to engage with our technical procurement team to discuss how this advanced monochiral catalyst technology can be integrated into your specific synthetic routes. Whether you require a Customized Cost-Saving Analysis for an existing process or need to evaluate the feasibility of a new route, our experts are ready to provide the support you need. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can drive value and efficiency in your supply chain.
