Advanced Synthesis of Dihydropyridino Spiro Indole Skeletons for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to access complex alkaloid scaffolds, particularly those with significant biological potential. Patent CN115093413B introduces a groundbreaking methodology for the construction of dihydropyridino spiro[3,4']indole and tetrahydropyridofuran[2,3-b]indol-5-one skeletons. These structural motifs are pervasive in natural products and serve as critical cores for various bioactive small molecules. The disclosed technology represents a paradigm shift from traditional transition metal-catalyzed processes to a more economical and environmentally benign approach utilizing hypervalent iodine reagents. By leveraging the unique oxidative capabilities of iodine(III) species in conjunction with Lewis acids, this invention enables the direct formation of these intricate spirocyclic systems under remarkably mild conditions. For R&D directors and process chemists, this patent offers a robust alternative that simplifies synthetic routes while maintaining high levels of structural diversity and yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiroindole skeletons has relied heavily on the use of precious transition metals such as gold, silver, palladium, iridium, and rhodium. As illustrated in prior art, these methods often involve the catalysis of unsaturated bonds within the substrate to generate pre-electrophilic centers, followed by aromatic electrophilic substitution reactions. While effective in academic settings, these conventional approaches present substantial hurdles for industrial application. The reliance on expensive noble metals significantly inflates the cost of goods, making the final intermediates less competitive in a price-sensitive market. Furthermore, these reactions frequently require harsh conditions, specialized ligands, and rigorous exclusion of air or moisture, which complicates process engineering. A major drawback is the necessity for pre-functionalization of substrates, adding extra synthetic steps and reducing overall atom economy. Additionally, the removal of trace heavy metal residues from the final product to meet stringent pharmaceutical purity standards adds further complexity and cost to the downstream processing.
The Novel Approach
In stark contrast, the methodology described in CN115093413B utilizes hypervalent iodine reagents and Lewis acids to mediate the cyclization at room temperature. This novel approach bypasses the need for expensive transition metal catalysts entirely. The reaction operates under mild conditions, typically between 20°C and 35°C, and tolerates a wide range of functional groups without the need for protective group manipulation or substrate pre-activation. The use of readily available Lewis acids such as Indium trifluoromethanesulfonate or Scandium trifluoromethanesulfonate ensures that the catalytic system is both accessible and cost-effective. This divergence from precious metal catalysis not only reduces the direct material costs but also streamlines the workflow by eliminating the need for specialized handling equipment required for air-sensitive metal complexes. The broad substrate scope demonstrated in the patent examples confirms that this method is versatile enough to accommodate various substituents on the indole ring and the amino acrylate chain, providing a powerful tool for medicinal chemists to explore structure-activity relationships efficiently.
Mechanistic Insights into Hypervalent Iodine-Mediated Spirocyclization
The core of this innovation lies in the intramolecular polar reverse spirocyclization reaction mediated by the hypervalent iodine and Lewis acid system. Unlike traditional electrophilic activations that rely on pi-acidic metals to activate alkynes or alkenes, this mechanism involves the oxidation of the electron-rich indole or enamine system by the hypervalent iodine species. The Lewis acid plays a crucial synergistic role by coordinating with the carbonyl or ester functionalities, thereby enhancing the electrophilicity of the reaction center and stabilizing the transition state. This dual activation strategy facilitates the formation of the quaternary spiro-center with high efficiency. The reaction proceeds through a concerted or stepwise pathway where the nucleophilic attack of the indole C3 position onto the activated beta-carbon of the acrylate moiety occurs smoothly. This mechanistic pathway is distinct from the 5-exo-dig cyclizations seen in gold catalysis, offering a different regiochemical outcome that is highly desirable for accessing specific alkaloid families.
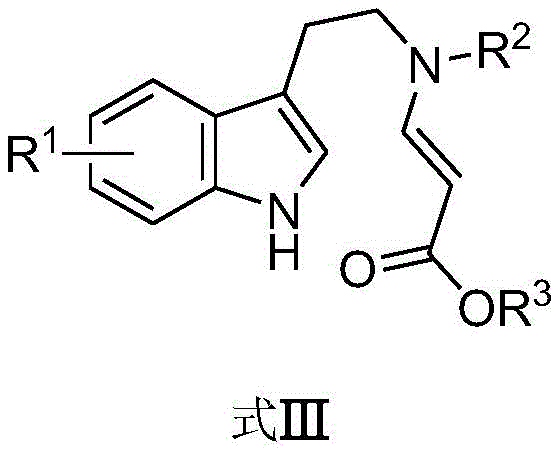
From an impurity control perspective, the mildness of the hypervalent iodine conditions is a significant advantage. Harsh acidic or basic conditions often lead to polymerization, decomposition, or the formation of regioisomeric byproducts. However, the neutral to mildly acidic environment provided by the Lewis acid and iodine reagent combination minimizes these side reactions. The high selectivity observed in the patent examples, with yields often exceeding 70-80% for diverse substrates, indicates a clean reaction profile. This cleanliness translates directly to easier purification, as the crude reaction mixtures contain fewer closely related impurities that are difficult to separate by chromatography. For quality control teams, this means a more robust process capable of consistently delivering high-purity intermediates that meet the rigorous specifications required for API synthesis.
How to Synthesize Dihydropyridino Spiro Indole Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and plant equipment. The general procedure involves dissolving the indole substituted beta-amino acrylate derivative in a suitable organic solvent such as ethyl acetate or dichloromethane. The hypervalent iodine reagent, such as 1-fluoro-3,3-dimethyl-1,3-dihydro-1-λ3-benzo[d][1,2]iodoxazole, is then added followed by the Lewis acid catalyst. The reaction is allowed to stir at room temperature until completion, which typically occurs within a few hours. Workup involves simple solvent removal and purification via silica gel column chromatography. The detailed standardized synthesis steps are outlined in the guide below.
- Prepare the reaction vessel by adding the indole substituted beta-amino acrylate derivative substrate along with the hypervalent iodine reagent and organic solvent.
- Introduce the Lewis acid catalyst to the mixture at room temperature or controlled low temperatures to initiate the cyclization.
- Upon completion, remove the solvent under reduced pressure and purify the crude product using column chromatography to isolate the target spiroindole skeleton.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hypervalent iodine-mediated technology offers compelling economic and logistical benefits. The most immediate impact is the drastic reduction in raw material costs associated with catalysts. By replacing precious metals like gold and palladium with inexpensive iodine reagents and earth-abundant Lewis acids, the direct cost of manufacturing is significantly lowered. This cost reduction in pharmaceutical intermediate manufacturing allows for more competitive pricing strategies and improved margin protection against fluctuations in the precious metal markets. Furthermore, the supply chain reliability is enhanced because hypervalent iodine reagents and common Lewis acids are commodity chemicals with stable global supply chains, unlike specialized organometallic catalysts which can suffer from availability issues and long lead times.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a major cost driver from the bill of materials. Additionally, the mild reaction conditions reduce energy consumption for heating or cooling, and the simplified workup procedures lower labor and solvent usage costs. The absence of heavy metals also negates the need for costly metal scavenging resins or specialized waste treatment processes, leading to substantial overall cost savings in the production lifecycle.
- Enhanced Supply Chain Reliability: Sourcing hypervalent iodine reagents and Lewis acids is far less risky than sourcing specialized chiral ligands or noble metal complexes. These reagents are produced at scale by multiple suppliers globally, ensuring continuity of supply even during market disruptions. The robustness of the reaction also means that raw material quality specifications can be slightly more relaxed without compromising the outcome, further widening the pool of eligible suppliers and reducing procurement lead times.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operational simplicity and safety profile. Running reactions at room temperature reduces the thermal load on reactors, allowing for larger batch sizes without extensive engineering modifications. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly strict regulatory requirements for green chemistry. This facilitates easier permitting and waste disposal, reducing the environmental compliance burden and associated costs for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for potential partners.
Q: What are the primary advantages of using hypervalent iodine over transition metals for spiroindole synthesis?
A: The use of hypervalent iodine reagents eliminates the need for expensive and toxic transition metals like gold, palladium, or iridium. This results in significantly lower raw material costs, reduced environmental impact due to the absence of heavy metal waste, and simpler purification processes that do not require rigorous metal scavenging steps.
Q: What are the typical reaction conditions for this synthesis method?
A: The reaction typically proceeds under mild conditions, often at room temperature (20-35°C) or slightly cooled conditions (0°C), using common organic solvents such as ethyl acetate or dichloromethane. The process utilizes Lewis acids like Indium triflate or Scandium triflate as catalysts.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the method is highly scalable due to its operational simplicity, use of readily available reagents, and mild reaction conditions. The avoidance of sensitive transition metal catalysts and the robustness of the hypervalent iodine system make it ideal for commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyridino Spiro Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the hypervalent iodine-mediated synthesis described in CN115093413B for the production of high-value alkaloid intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to verify that every batch meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cost-effective and sustainable synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can accelerate your development timeline while optimizing your overall production costs.
