Advanced Copper-Catalyzed Synthesis of Aryl Difluoromethylselenyl Ethers for Commercial Scale-up
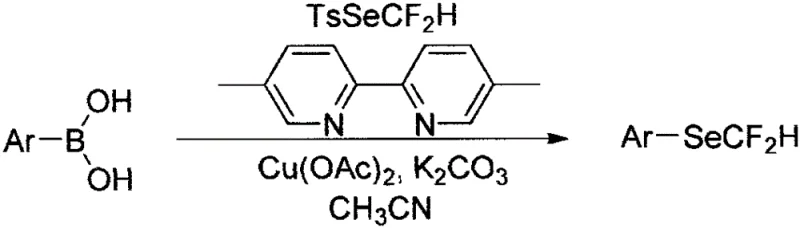
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to incorporate fluorine and selenium motifs into complex molecular architectures, driven by the profound impact these elements have on metabolic stability and lipophilicity. Patent CN111848480A introduces a groundbreaking approach for the synthesis of aryl difluoromethylselenyl ethers, utilizing readily available arylboronic acids as the primary substrate. This innovation addresses a critical gap in organoselenium chemistry by providing a direct, efficient, and scalable route to install the difluoromethylselenyl (SeCF2H) group onto aromatic rings. The methodology leverages a copper-catalyzed cross-coupling strategy that operates under remarkably mild conditions, specifically at room temperature, which is a significant departure from traditional methods that often require harsh thermal activation or cryogenic environments. By employing difluoromethylseleno-p-toluenesulfonate (TsSeCF2H) as the selenium source, the process ensures high reactivity while maintaining operational safety, making it an ideal candidate for the reliable production of high-purity pharmaceutical intermediates.
The strategic value of this technology lies in its ability to transform simple, commodity-grade starting materials into high-value functionalized building blocks. The SeCF2H moiety is increasingly recognized for its unique electronic properties, which can enhance membrane permeability and binding affinity in drug candidates. Consequently, mastering the synthesis of these derivatives is paramount for R&D teams aiming to optimize lead compounds. This patent not only delineates the specific reaction parameters but also demonstrates a broad substrate scope, encompassing various electronic and steric environments on the aryl ring. For procurement and supply chain leaders, this represents a tangible opportunity for cost reduction in pharmaceutical intermediates manufacturing, as it bypasses the need for exotic precursors and simplifies the overall synthetic sequence.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of selenium-containing functional groups into organic molecules has been fraught with significant challenges that hinder large-scale adoption. Traditional protocols often rely on the use of highly toxic and malodorous reagents such as hydrogen selenide or elemental selenium powder, which pose severe safety risks and require specialized containment infrastructure. Furthermore, many existing methods necessitate the use of expensive transition metal catalysts like palladium or rhodium, which not only inflate the raw material costs but also introduce complexities regarding residual metal removal—a critical quality attribute for API manufacturing. Another major drawback of conventional approaches is the instability of the selenium intermediates; many difluoromethyl selenium precursors are thermally unstable or prone to decomposition upon exposure to moisture or air, leading to inconsistent yields and difficult reproducibility. Additionally, older synthetic routes frequently demand extreme reaction conditions, such as very low temperatures or strong bases, which limit the functional group tolerance and complicate the commercial scale-up of complex polymer additives or drug substances.
The Novel Approach
In stark contrast, the methodology described in CN111848480A offers a streamlined and economically viable alternative that directly addresses these historical pain points. The core of this innovation is the utilization of arylboronic acids, which are ubiquitous, inexpensive, and commercially available in vast structural diversity, serving as the nucleophilic partner. The electrophilic selenium source, TsSeCF2H, is highlighted for its stability and ease of preparation in large quantities, effectively mitigating the supply chain risks associated with unstable reagents. The reaction proceeds efficiently at room temperature in acetonitrile, utilizing copper acetate as an earth-abundant catalyst and potassium carbonate as a mild base. This shift to mild conditions drastically reduces energy consumption and eliminates the need for cryogenic cooling systems, thereby simplifying the engineering requirements for plant operations. Moreover, the compatibility of this system with a wide range of functional groups—including esters, nitriles, and halogens—means that late-stage functionalization is feasible, reducing the number of synthetic steps required to reach the final target molecule.
Mechanistic Insights into Copper-Catalyzed Difluoromethylselenylation
The mechanistic pathway of this transformation is believed to proceed through a copper-mediated catalytic cycle that facilitates the transmetallation of the aryl group from boron to copper, followed by reductive elimination to form the C-Se bond. The use of 4,4'-dimethyl-2,2'-bipyridine as a ligand is crucial, as it stabilizes the copper center and modulates its electronic properties to favor the oxidative addition of the TsSeCF2H reagent. This ligand environment helps to lower the activation energy barrier for the key bond-forming step, allowing the reaction to proceed rapidly even at ambient temperatures. The role of potassium carbonate is twofold: it activates the boronic acid species to enhance transmetallation efficiency and neutralizes the sulfonic acid byproduct generated during the reaction, driving the equilibrium towards product formation. Understanding this mechanism is vital for process chemists, as it highlights the importance of maintaining anhydrous conditions to prevent hydrolysis of the boronic acid or the catalyst, ensuring consistent performance across different batches.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining product integrity. The Se-CF2 bond is susceptible to cleavage under strongly acidic or basic conditions, but the neutral to slightly basic environment provided by the potassium carbonate/acetonitrile system preserves this sensitive linkage. Furthermore, the selectivity of the copper catalyst minimizes homocoupling of the boronic acid, a common side reaction in cross-coupling chemistry that can generate difficult-to-remove biaryl impurities. The protocol specifies a straightforward workup involving aqueous extraction and silica gel chromatography, which effectively removes copper residues and unreacted starting materials. This simplicity in purification is a significant advantage for regulatory compliance, as it facilitates the generation of clean spectral data and ensures that the final material meets the stringent purity specifications required for clinical applications.
How to Synthesize Aryl Difluoromethylselenyl Ether Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize yield and minimize waste. The patent outlines a specific molar ratio of 1.2:1:0.1:1:0.1 for the arylboronic acid, TsSeCF2H, copper acetate, potassium carbonate, and ligand, respectively, which has been optimized to balance reaction rate with reagent cost. The process begins with the dissolution of the solid reagents in anhydrous acetonitrile under an inert atmosphere to prevent oxidation of the catalyst or the selenium species. Following the addition of the TsSeCF2H reagent, the mixture is stirred for a defined period of 12 to 14 hours, allowing sufficient time for the conversion to reach completion as monitored by thin-layer chromatography. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining arylboronic acid, copper acetate, potassium carbonate, and 4,4'-dimethyl-2,2'-bipyridine in anhydrous acetonitrile.
- Add the difluoromethylselenylation reagent TsSeCF2H dropwise to the stirred mixture at room temperature.
- Stir the reaction for 12-14 hours, monitor by TLC, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into immediate and tangible benefits regarding cost structure and operational reliability. The shift from precious metal catalysts to copper-based systems represents a fundamental change in the cost drivers of the synthesis, removing the volatility associated with palladium pricing. Additionally, the use of arylboronic acids as starting materials leverages a mature global supply chain, ensuring that raw materials are accessible from multiple vendors, which mitigates the risk of single-source dependency. The room temperature operation further contributes to cost reduction in pharmaceutical intermediates manufacturing by lowering utility costs related to heating and cooling, while also extending the lifespan of reactor equipment by reducing thermal stress. These factors combined create a more resilient and economical supply chain capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the use of commodity chemicals significantly lowers the bill of materials. By avoiding cryogenic conditions and complex purification techniques, the overall processing time and energy consumption are drastically reduced, leading to substantial cost savings per kilogram of produced material. The high atom economy of the reaction ensures that raw materials are converted efficiently into the desired product, minimizing waste disposal costs.
- Enhanced Supply Chain Reliability: Relying on widely available arylboronic acids and stable selenium reagents ensures a consistent supply of inputs, reducing the likelihood of production delays due to material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, resulting in more predictable batch outcomes and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving standard extraction and chromatography, facilitates easy scale-up from laboratory to pilot and commercial production scales without the need for specialized equipment. Furthermore, the use of less toxic reagents and the generation of manageable byproducts align with green chemistry principles, simplifying environmental permitting and waste management compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis method. These insights are derived directly from the experimental data and procedural details provided in the patent documentation, offering clarity on reaction scope and practical handling. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using TsSeCF2H over other selenium reagents?
A: TsSeCF2H (difluoromethylseleno-p-toluenesulfonate) offers superior stability and safety compared to volatile or highly toxic selenium precursors. It acts as a robust electrophilic source that reacts efficiently under mild conditions.
Q: Is this method compatible with sensitive functional groups?
A: Yes, the protocol operates at room temperature with a mild base (potassium carbonate), allowing tolerance for esters, nitriles, nitro groups, and hydroxyls without significant side reactions or decomposition.
Q: How is the product purified after the reaction?
A: The workup involves a standard aqueous extraction followed by drying and solvent removal. The final purification is achieved through conventional silica gel column chromatography, avoiding complex crystallization steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Difluoromethylselenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organoselenium chemistry in modern drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop concept to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aryl difluoromethylselenyl ether meets the highest international standards. We are committed to supporting your R&D efforts with high-purity intermediates that accelerate your timeline to market.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this copper-catalyzed method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering complex fine chemical solutions.
