Advanced One-Pot Synthesis of ATC: Enabling Scalable Production of High-Purity L-Cysteine Precursors
Introduction to Continuous ATC Manufacturing Technology
The global demand for L-cysteine, a critical amino acid utilized extensively in pharmaceutical formulations, food additives, and cosmetic applications, has driven intense research into efficient precursor synthesis. Patent CN102180842B introduces a groundbreaking methodology for the production of 2-amino-Δ2-thiazoline-4-carboxylic acid (ATC), the direct substrate for enzymatic L-cysteine production. Unlike conventional batch processes that suffer from low throughput and high waste generation, this invention discloses a sophisticated one-pot tandem reaction sequence. By integrating chlorination, dehydrochlorination, thiourea addition, and cyclization into a single continuous workflow, the technology eliminates the need for isolating unstable intermediates. This approach not only streamlines the operational workflow but also drastically reduces the capital expenditure required for separation equipment and solvent recovery units. For industrial stakeholders, this represents a paradigm shift towards greener, more atom-economical manufacturing of high-value sulfur-containing heterocycles.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ATC and its downstream derivatives has been plagued by inefficient process designs that rely heavily on the isolation of intermediates. Prior art, such as US Patent 4072687, describes a route starting from 2-chloroacrylic acid which necessitates the isolation of the S-(β-carboxy-β-vinyl) isothiourea intermediate. This isolation step is technically cumbersome due to the instability of the intermediate and adds significant time and cost to the overall production cycle. Furthermore, alternative methods utilizing acrylic acid as a starting material face severe physical limitations; acrylic acid is a solid at lower chlorination temperatures, requiring the addition of large volumes of inert organic solvents to maintain a liquid reaction phase. These solvents must subsequently be recovered and purified, leading to excessive energy consumption and increased environmental burden. The reliance on solid starting materials and multi-step isolation protocols inherently limits the scalability and economic viability of these traditional routes for commercial-scale operations.
The Novel Approach
The methodology outlined in CN102180842B overcomes these historical bottlenecks by utilizing methyl acrylate, a readily available liquid industrial chemical, as the primary feedstock. The innovation lies in the precise control of reaction conditions that allow four distinct chemical transformations to occur sequentially in the same reactor vessel. By employing a specific temperature ramping program and introducing minute quantities of cosolvents, the process maintains high reaction selectivity without the need for intermediate workups. This telescoping of reactions means that the crude product from the chlorination step is directly subjected to alkaline hydrolysis, followed immediately by thiourea addition and final cyclization. This continuous flow of chemistry minimizes material handling, reduces the footprint of the manufacturing facility, and significantly lowers the total cost of ownership for producing ATC. The result is a robust process capable of delivering high-purity products with a streamlined operational profile that is ideally suited for modern continuous manufacturing environments.
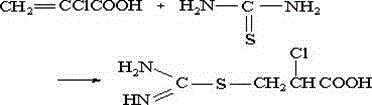
Mechanistic Insights into Cosolvent-Assisted Chlorination and Cyclization
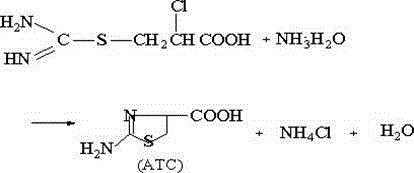
A critical component of this synthesis is the initial chlorination of methyl acrylate, a reaction that is notoriously difficult to control due to the propensity of acrylic double bonds to undergo radical polymerization. The patent reveals that the addition of polar aprotic cosolvents, specifically N,N-dimethylformamide (DMF) or triethanolamine, at concentrations as low as 1% to 5% by weight, plays a pivotal mechanistic role. These additives function not merely as solvents but as dispersion agents that enhance the solubility of chlorine gas within the organic phase. By improving gas-liquid mass transfer, the cosolvents prevent the local accumulation of high chlorine concentrations which typically initiate uncontrolled radical chain reactions leading to polymer formation. This ensures that the electrophilic addition of chlorine across the double bond proceeds selectively to form 2,3-dichloro methyl propionate with yields exceeding 97.5%, providing a clean substrate for subsequent transformations without the need for distillation purification.
Following the formation of the dichloro intermediate, the process relies on a carefully orchestrated sequence of base-mediated elimination and nucleophilic substitution. The conversion to 2-chloroacrylic acid is achieved under mild alkaline conditions, which then reacts in situ with thiourea. The final cyclization step is triggered by adjusting the pH of the reaction mixture to neutrality using ammonium hydroxide. This pH swing facilitates the intramolecular nucleophilic attack of the sulfur atom onto the activated carbon center, closing the thiazoline ring to form the final ATC structure. The ability to perform these pH-sensitive steps in a single pot without isolating the sensitive chloro-acid or isothiourea intermediates demonstrates a deep understanding of the kinetic and thermodynamic parameters governing the reaction pathway. This mechanistic control is what allows the process to achieve a final HPLC purity of 98.2%, ensuring the material is suitable for sensitive enzymatic downstream applications.
How to Synthesize 2-amino-Δ2-thiazoline-4-carboxylic acid Efficiently
The implementation of this synthesis route requires precise adherence to the temperature profiles and reagent addition rates specified in the patent to ensure safety and optimal yield. The process begins with the low-temperature chlorination of methyl acrylate, followed by a controlled warm-up to complete the reaction, and proceeds through alkaline treatment and thiourea addition without breaking the vacuum or transferring the bulk material. Detailed standard operating procedures regarding specific molar ratios, agitation speeds, and crystallization parameters are essential for replicating the high efficiency described in the intellectual property. For process engineers looking to adopt this technology, the following guide outlines the critical operational phases required to transition from laboratory scale to commercial production effectively.
- Chlorinate methyl acrylate with chlorine gas at -5°C to 10°C using DMF or triethanolamine as a cosolvent to prevent polymerization.
- React the resulting 2,3-dichloro methyl propionate with aqueous sodium hydroxide at 30°C to 40°C to form 2-chloroacrylic acid in situ.
- Add thiourea and hydrochloric acid, heat to 75°C-85°C for addition, then adjust pH to 7.0-7.5 with ammonia water for cyclization and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot synthesis technology offers substantial strategic advantages beyond simple yield improvements. The elimination of intermediate isolation steps translates directly into a reduction in unit operations, which lowers both the fixed capital investment for plant infrastructure and the variable costs associated with labor and utilities. By removing the need for extensive solvent recovery systems typically required when using solid acrylic acid or inert diluents, the process significantly reduces energy consumption and waste disposal costs. This lean manufacturing approach enhances the overall cost competitiveness of the final ATC product, allowing suppliers to offer more aggressive pricing structures while maintaining healthy margins. Furthermore, the use of methyl acrylate, a commodity chemical with a stable and abundant global supply chain, mitigates the risk of raw material shortages that can plague specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the drastic simplification of the process flow. By telescoping four reaction steps into a single vessel, the requirement for multiple reactors, filtration units, and drying ovens is eliminated. This reduction in equipment intensity leads to lower depreciation costs and reduced maintenance overheads. Additionally, the avoidance of large volumes of inert organic solvents removes the costly and energy-intensive step of solvent distillation and recycling. The high selectivity of the chlorination step, achieved through the use of minimal cosolvents, ensures that raw material utilization is maximized, further driving down the cost of goods sold (COGS) for the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: Relying on methyl acrylate as the primary feedstock provides a robust foundation for supply chain stability. Unlike specialized starting materials that may have limited suppliers or long lead times, methyl acrylate is produced on a massive industrial scale for the polymer industry, ensuring consistent availability and price stability. The simplified process also reduces the complexity of the manufacturing schedule, allowing for faster batch turnover times and improved responsiveness to market demand fluctuations. This agility is crucial for pharmaceutical supply chains where Just-In-Time delivery and consistent quality are paramount. The ability to produce high-purity ATC without complex purification trains ensures that supply disruptions due to equipment failure or purification bottlenecks are minimized.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with modern green chemistry principles. The reduction in solvent usage significantly lowers the volume of volatile organic compounds (VOCs) emitted during production, simplifying compliance with stringent environmental regulations. The aqueous nature of the later reaction steps and the generation of benign byproducts like ammonium chloride facilitate easier wastewater treatment compared to processes laden with organic waste streams. This environmental efficiency not only reduces the cost of waste management but also future-proofs the manufacturing site against tightening ecological standards. The gentle reaction conditions and lack of hazardous intermediate handling make the process inherently safer and easier to scale from pilot plants to multi-ton commercial facilities without encountering the heat transfer or mixing limitations often seen in batch chlorinations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ATC synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the operational realities and benefits. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the addition of DMF or triethanolamine critical in the chlorination step?
A: These cosolvents act as dispersing agents that improve the solubility of chlorine gas in methyl acrylate. This prevents local accumulation of chlorine which can cause radical reactions and unwanted polymerization of the acrylic double bond, thereby significantly improving the yield of the dichloro intermediate.
Q: How does this method compare to traditional acrylic acid chlorination routes?
A: Traditional methods often use solid acrylic acid which requires large amounts of inert organic solvents to maintain a liquid state for reaction. This new method utilizes liquid methyl acrylate directly, eliminating the need for bulky solvent recovery systems and reducing energy consumption associated with distillation.
Q: What is the purity profile of the ATC produced via this continuous route?
A: The patent data indicates that the final product achieves a high-pressure liquid chromatography (HPLC) purity of approximately 98.2%. The controlled temperature profile and avoidance of intermediate isolation help minimize the formation of complex byproducts often seen in multi-step batch processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-amino-Δ2-thiazoline-4-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality precursors play in the efficient production of downstream amino acids like L-cysteine. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of ATC performs consistently in your enzymatic or chemical synthesis processes. Our facility is equipped to handle the specific temperature control and safety requirements of chlorination chemistry, providing a secure and reliable source for your supply chain.
We invite you to collaborate with us to optimize your amino acid manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can enhance your production efficiency and reduce your overall operational expenses.
