Scalable Ligand-Free Synthesis of Beta-Silylated Aromatic Nitro Intermediates for Pharmaceutical Manufacturing
Introduction to Advanced Beta-Silylation Technology
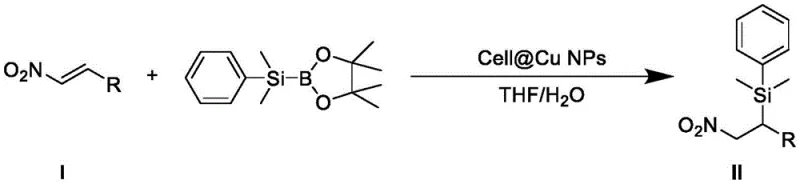
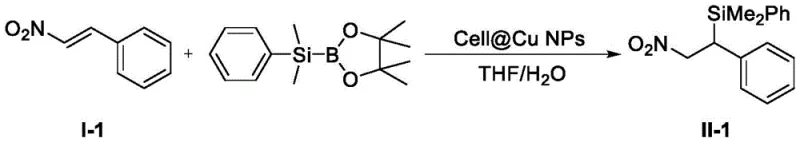
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the urgent need for greener, more cost-effective methodologies that do not compromise on yield or purity. A significant breakthrough in this domain is documented in Chinese Patent CN112778351B, which introduces a novel preparation method for beta-dimethylphenyl silicon substituted aromatic nitro compounds. This technology represents a paradigm shift from traditional homogeneous catalysis systems that rely on expensive, non-commercialized ligands. By leveraging cellulose-supported copper nanoparticles (Cell@Cu NPs), this invention achieves efficient beta-silylation of alpha,beta-unsaturated carbonyl compounds under remarkably mild conditions. For R&D directors and procurement specialists alike, this patent offers a compelling value proposition: a ligand-free, heterogeneous catalytic system that operates at room temperature in a THF/water mixture. The implications for scaling up the production of high-purity pharmaceutical intermediates are profound, as it directly addresses critical pain points regarding catalyst recovery, environmental compliance, and overall process economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C(sp3)-Si bonds in beta-silicon substituted aromatic nitro compounds has been fraught with significant technical and economic hurdles. Prior art, such as the methods reported in the Journal of the American Chemical Society (2015), relied heavily on the complexation of divalent copper salts like Cu(acac)2 with specialized chiral bipyridine ligands. These ligands are not only synthetically challenging to prepare but are also uncommercialized, leading to exorbitant raw material costs that render the process economically unviable for large-scale manufacturing. Furthermore, alternative metal-free approaches described in literature often necessitate the use of substantial quantities of strong acids and toxic bases, such as pyridine, creating severe environmental hazards and complicating downstream purification. The reliance on toxic solvents and the generation of hazardous waste streams in these conventional routes pose unacceptable risks for modern supply chains that prioritize sustainability and regulatory compliance. Additionally, these older methods frequently suffer from prolonged reaction times, sometimes extending up to 24 hours, and inconsistent yields, which undermines production efficiency and predictability.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112778351B utilizes a robust, heterogeneous catalyst system based on cellulose-supported copper nanoparticles. This innovative approach completely eliminates the need for any external ligands, particularly the costly and hard-to-source chiral ligands that plagued previous iterations. The reaction proceeds efficiently at room temperature using a benign THF/water solvent system, drastically reducing energy consumption compared to methods requiring elevated temperatures. The heterogeneous nature of the Cell@Cu NPs catalyst allows for straightforward separation via simple filtration, enabling the catalyst to be washed and reused multiple times without significant loss of catalytic activity. This not only simplifies the operational workflow but also significantly reduces the total cost of ownership for the catalyst system. By avoiding toxic additives and harsh conditions, this novel route aligns perfectly with green chemistry principles, offering a safer, more sustainable pathway for the commercial scale-up of complex pharmaceutical intermediates while maintaining high conversion rates and excellent product purity.
Mechanistic Insights into Cell@Cu NPs Catalyzed Beta-Silylation
The core of this technological advancement lies in the unique interaction between the cellulose-supported copper nanoparticles and the silylating agent, (dimethylphenylsilyl)boronic acid pinacol ester. Under the catalytic influence of Cell@Cu NPs, the supported copper nanoparticles form a transient complex metal species with the boron-silicon reagent. This activation step is crucial as it facilitates the transfer of the silyl group to the substrate through a highly organized six-membered ring transition state. Unlike traditional 1,2-additions, this mechanism selectively favors a 1,4-addition pathway, ensuring the silicon moiety is installed precisely at the beta-position relative to the nitro group. This regioselectivity is vital for the subsequent utility of these intermediates in downstream synthetic transformations, such as the preparation of aromatic amines or heterocyclic structures. The cellulose support plays a dual role: it stabilizes the copper nanoparticles preventing aggregation, thereby maintaining high surface area and catalytic activity, and it provides a solid matrix that renders the entire system heterogeneous. This structural integrity ensures that the active copper species remain accessible to the reactants in the liquid phase while being easily retrievable post-reaction.
From an impurity control perspective, this mechanism offers distinct advantages over homogeneous counterparts. The absence of free ligands in the reaction mixture means there are no ligand-derived byproducts or residual ligand contaminants to remove during purification, which is a common challenge in metal-ligand catalyzed processes. The mild reaction conditions at room temperature further suppress thermal degradation pathways and side reactions that often lead to complex impurity profiles in high-temperature processes. The selectivity of the 1,4-addition minimizes the formation of regioisomers, simplifying the chromatographic separation steps required to achieve high-purity specifications. For quality control teams, this translates to a cleaner crude reaction profile and higher overall recovery of the target molecule. The robustness of the cellulose support also prevents copper leaching into the product stream, ensuring that the final API intermediate meets stringent heavy metal limits without requiring aggressive metal scavenging treatments, thus preserving the integrity of the sensitive nitro functionality.
How to Synthesize Beta-Dimethylphenyl Silicon Substituted Aromatic Nitro Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting is designed to be operationally simple and scalable. The process begins with the preparation of the catalyst suspension, followed by the addition of substrates under ambient conditions, eliminating the need for specialized heating or cooling equipment. The standard protocol involves dispersing the Cell@Cu NPs in a THF/water mixture, stirring to ensure homogeneity, and then introducing the nitro-alkene and silyl-boron reagent. The reaction progress is monitored until completion, typically within 6 to 12 hours, after which the solid catalyst is filtered off. This filtrate contains the crude product, which is then concentrated and purified via standard column chromatography using ethyl acetate and petroleum ether mixtures. The simplicity of this workflow makes it highly attractive for rapid process development and tech transfer. For detailed operational parameters and specific stoichiometric ratios validated by experimental data, please refer to the standardized synthesis guide below.
- Disperse cellulose-supported copper nanoparticles (Cell@Cu NPs) in a tetrahydrofuran aqueous solution and stir at room temperature to form a homogeneous suspension.
- Add the nitro-substituted alkenyl compound and (dimethylphenylsilyl)boronic acid pinacol ester to the mixture, maintaining a molar ratio of 1: 1.0-2.0, and stir until reaction completion.
- Filter the reaction mixture to recover the solid catalyst for reuse, then concentrate the filtrate and purify the residue via column chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand-free catalytic technology offers transformative benefits that extend far beyond the laboratory bench. The primary driver for cost optimization here is the complete elimination of expensive, custom-synthesized chiral ligands, which historically accounted for a significant portion of the raw material bill in similar silylation reactions. By substituting these proprietary reagents with a reusable, cellulose-based copper catalyst, the direct material costs are drastically simplified and reduced. Furthermore, the ability to recover and recycle the catalyst multiple times without regeneration losses creates a circular economy within the manufacturing process, effectively amortizing the catalyst cost over tons of production rather than single batches. This stability in supply and cost structure provides a hedge against market volatility for precious metal catalysts and specialty ligands, ensuring long-term budget predictability for large-scale campaigns.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the removal of high-cost ligand inputs and the reduction in energy expenditure associated with room temperature operations. Traditional methods often require heating to 50°C or higher and extended reaction times, whereas this method proceeds efficiently at ambient temperature, lowering utility costs. The simplified workup procedure, which avoids complex extractions to remove toxic bases or acids, reduces solvent consumption and labor hours. Consequently, the overall cost of goods sold (COGS) for these beta-silylated intermediates is significantly lowered, enhancing margin potential for downstream API manufacturers. The avoidance of toxic reagents also minimizes waste disposal fees, contributing further to the bottom line.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commercially available and stable reagents. Unlike specialized chiral ligands that may have long lead times or single-source dependencies, the components of this system—cellulose, copper salts, and common boron esters—are commodity chemicals with robust global supply networks. The robustness of the catalyst allows for stockpiling without significant degradation concerns, ensuring continuity of supply even during market disruptions. Additionally, the high yield and consistency demonstrated across various substrates reduce the risk of batch failures, which can cause costly delays in multi-step synthesis sequences. This reliability is critical for maintaining just-in-time inventory levels and meeting tight delivery schedules for pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from grams to metric tons is facilitated by the heterogeneous nature of the catalyst, which is compatible with standard fixed-bed or slurry reactor configurations used in fine chemical plants. The green chemistry credentials of the process, utilizing water as a co-solvent and avoiding toxic additives, streamline regulatory approvals and environmental permitting. Reduced hazardous waste generation simplifies effluent treatment requirements, lowering the environmental footprint of the manufacturing site. This alignment with ESG (Environmental, Social, and Governance) goals is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies, making this technology a strategic asset for future-proofing the supply chain.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries based on the patent data and our internal process knowledge. These questions address critical aspects such as catalyst longevity, substrate compatibility, and purification protocols. Understanding these details is essential for accurate process modeling and risk assessment. The following responses provide a clear picture of what to expect when transitioning this chemistry from paper to production, ensuring that all stakeholders have the necessary information to make informed decisions regarding technology adoption and resource allocation.
Q: Can the cellulose-supported copper catalyst be recycled without loss of activity?
A: Yes, the patent data confirms that the heterogeneous Cell@Cu NPs catalyst can be recovered by simple filtration, washed with ethyl acetate and acetone, and reused for at least five consecutive cycles with yields remaining consistently high (above 90%).
Q: What are the specific reaction conditions required for this beta-silylation?
A: The process operates under mild conditions, specifically at room temperature using a THF/water solvent system. It eliminates the need for expensive chiral ligands or toxic bases, typically requiring 6 to 12 hours for completion depending on the substrate.
Q: Is this method suitable for substrates with electron-withdrawing groups?
A: Absolutely. The methodology demonstrates broad substrate tolerance, successfully processing various aromatic nitro alkenes including those with phenyl, p-methyl, p-fluoro, and heteroaromatic furan groups, delivering high conversion rates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Dimethylphenyl Silicon Substituted Aromatic Nitro Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the Cell@Cu NPs catalyzed beta-silylation route and is fully prepared to implement this technology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of beta-dimethylphenyl silicon substituted aromatic nitro compounds delivered meets the highest industry standards for impurity profiles and physical properties.
We invite you to leverage our technical expertise to optimize your supply chain for these critical intermediates. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive quotation. Let us demonstrate how our commitment to innovation and quality can drive value and efficiency in your drug development pipeline, ensuring a reliable supply of high-performance chemical building blocks for your most challenging projects.
