Advanced Catalytic Synthesis of Chiral Amines for Commercial Pharmaceutical Manufacturing
Advanced Catalytic Synthesis of Chiral Amines for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously demands more efficient routes to access complex chiral building blocks, particularly those containing nitrogen heterocycles which are ubiquitous in bioactive molecules. Patent CN112608274A discloses a groundbreaking catalytic synthesis method for chiral amine compounds that addresses critical bottlenecks in traditional manufacturing. This technology leverages a dual-catalytic system involving palladium and a specific chiral aldehyde to facilitate the direct coupling of methylamine compounds with cinnamyl alcohol derivative esters. The resulting intermediates are subsequently protected to yield high-value chiral amines with excellent enantiomeric excess. This innovation represents a significant leap forward for reliable pharmaceutical intermediate suppliers seeking to optimize their production portfolios with robust, scalable chemistry.
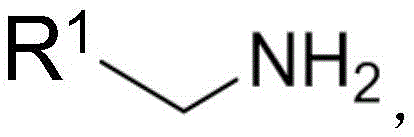
Traditionally, the synthesis of chiral amines bearing unsaturated side chains has been fraught with challenges, often requiring harsh conditions that compromise sensitive functional groups. Conventional methods typically rely on the catalytic hydrogenation of imines using chiral metal catalysts, such as nickel-based systems. However, these hydrogenation protocols frequently fail when the target molecule contains unsaturated chain hydrocarbon groups, as the double bonds are inadvertently reduced, destroying the desired structural motif. Alternative approaches involving acrylation of imines or decarboxylative allylation of amino acid derivatives often suffer from excessively long reaction routes, multiple synthetic steps, and the necessity for additional protective group manipulations that increase waste and cost. These limitations severely hinder the commercial scale-up of complex pharmaceutical intermediates, forcing manufacturers to seek more direct and atom-economical alternatives.
In stark contrast, the novel approach detailed in the patent utilizes a streamlined palladium-catalyzed allylic alkylation strategy that operates under remarkably mild alkaline conditions. By reacting a methylamine compound directly with a cinnamyl alcohol derivative ester in the presence of a chiral aldehyde catalyst, the method constructs the chiral center and the carbon-nitrogen bond simultaneously without the need for pre-functionalized imines. This one-pot-like efficiency eliminates the requirement for external protective groups during the initial C-N bond formation, significantly simplifying the workflow. The subsequent in-situ protection with Boc2O ensures the stability of the chiral amine for downstream processing. This methodology not only preserves the valuable alkene functionality found in cinnamyl derivatives but also accommodates a wide range of nitrogen-containing heterocycles, including quinolines and pyridines, thereby expanding the chemical space accessible for drug discovery.
Mechanistic Insights into Pd-Catalyzed Asymmetric Allylic Alkylation
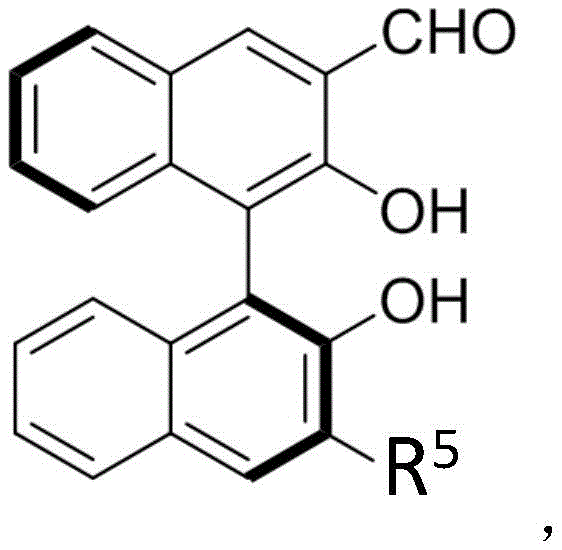
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst and the chiral aldehyde organocatalyst. The reaction initiates with the oxidative addition of the palladium(0) species to the cinnamyl alcohol derivative ester, generating a cationic pi-allyl palladium complex. This electrophilic intermediate is then intercepted by the nucleophilic methylamine compound. Crucially, the chiral aldehyde, such as the binaphthyl-derived structures shown in the patent data, interacts with the reaction milieu to create a highly defined chiral pocket. This chiral environment dictates the facial selectivity of the nucleophilic attack, ensuring that the amine adds to the allyl system with high stereocontrol. The use of Lewis acids like ZnF2 and Lewis bases like TMG further modulates the reactivity, stabilizing transition states and suppressing racemic background reactions.
Impurity control is another critical aspect where this mechanism excels, particularly for R&D directors focused on purity profiles. The mild reaction temperatures, typically ranging from -40°C to 80°C with an optimal window around 0°C, prevent thermal degradation of sensitive heterocyclic rings and minimize the formation of regioisomers. Furthermore, the specific choice of ligands, such as dppp or dppf, coordinated with the palladium center enhances the stability of the catalytic cycle, reducing the likelihood of palladium black precipitation which can act as a source of metal contamination. The protocol specifies a straightforward workup involving filtration through diatomite and silica gel treatment, effectively removing residual metal catalysts and polar byproducts. This rigorous control over the reaction pathway ensures that the final high-purity chiral amine compounds meet stringent regulatory standards required for API manufacturing, minimizing the burden on downstream purification processes.
How to Synthesize Chiral Amine Compounds Efficiently
Implementing this synthesis route requires precise control over stoichiometry and atmospheric conditions to maximize yield and enantioselectivity. The patent outlines a standardized procedure where the palladium catalyst and solvent are pre-mixed under an inert nitrogen atmosphere to prevent oxidation of the active Pd(0) species. Subsequent addition of the substrates and auxiliaries must be sequenced carefully to maintain the integrity of the catalytic cycle. While the general procedure is robust, optimization of the chiral aldehyde loading (typically 10-20 mol%) is essential for balancing cost and performance. For detailed operational parameters and safety considerations regarding the handling of palladium catalysts and organic bases, please refer to the specific guidelines below.
- Prepare the reaction vessel under inert atmosphere with palladium catalyst and solvent, then add methylamine compound, cinnamyl ester, chiral aldehyde, and auxiliaries.
- React at controlled temperatures (e.g., 0°C) until consumption of the cinnamyl derivative, then isolate the intermediate product via chromatography.
- Treat the intermediate with Boc2O and base (Na2CO3) at room temperature to install the protecting group and finalize the chiral amine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic method offers substantial strategic benefits beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment and the avoidance of cryogenic conditions drastically simplify the infrastructure requirements for production. This translates to lower capital expenditure (CAPEX) for manufacturing facilities and reduced operational risks associated with handling hazardous gases. Moreover, the use of commodity chemicals like toluene as the primary solvent ensures that raw material sourcing remains stable and cost-effective, shielding the supply chain from volatility associated with exotic or regulated solvents. The streamlined nature of the process inherently supports cost reduction in chiral amine manufacturing by reducing the total number of unit operations and minimizing solvent consumption per kilogram of product.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by bypassing the need for multiple protection and deprotection steps that are characteristic of older synthetic routes. By directly synthesizing the chiral amine scaffold without additional protective groups in the first step, the method reduces the consumption of reagents and the generation of chemical waste. The high turnover frequency of the palladium catalyst allows for lower catalyst loading relative to the substrate, further driving down the bill of materials. Additionally, the simplified purification protocol, which relies on standard column chromatography or crystallization rather than complex distillation or preparative HPLC, lowers energy costs and labor hours, resulting in a more competitive cost structure for bulk procurement.
- Enhanced Supply Chain Reliability: The starting materials, specifically cinnamyl alcohol derivative esters and various methylamine heterocycles, are widely available from global chemical suppliers, ensuring a resilient supply chain. The robustness of the reaction against minor variations in temperature and moisture content means that batch-to-batch consistency is easier to maintain, reducing the risk of production delays due to failed quality control tests. This reliability is crucial for reducing lead time for high-purity chiral amines, allowing pharmaceutical companies to accelerate their clinical trial timelines. The ability to source diverse R2 and R3 substituents from a common platform technology also enables rapid analog synthesis without needing to requalify entirely new manufacturing processes for each derivative.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns well with green chemistry principles. The reaction operates at near-ambient pressures and moderate temperatures, significantly lowering the energy footprint compared to high-temperature reflux or high-pressure hydrogenation processes. The absence of stoichiometric heavy metal reagents (using catalytic amounts instead) and the potential for solvent recycling make the process more sustainable. Scaling this reaction from gram to ton scale is facilitated by the homogeneous nature of the catalytic system, which ensures efficient heat and mass transfer in large reactors. This scalability ensures that supply can meet demand surges without compromising the environmental compliance standards increasingly demanded by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these nuances is vital for integrating this synthesis route into existing manufacturing pipelines. The answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What are the key advantages of this Pd-catalyzed method over traditional hydrogenation?
A: Unlike catalytic hydrogenation which struggles with unsaturated chain hydrocarbon groups, this method utilizes allylic alkylation to preserve double bonds while establishing chirality, allowing for diverse functionalization in downstream API synthesis.
Q: How does the chiral aldehyde catalyst influence enantioselectivity?
A: The chiral aldehyde acts as an organocatalyst that forms a transient chiral environment around the palladium center, directing the nucleophilic attack of the amine to specific faces of the pi-allyl palladium complex, achieving ee values up to 94%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates under mild conditions (0°C to room temperature) using common solvents like toluene and avoids cryogenic temperatures or high-pressure hydrogenation, making it highly adaptable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed synthesis technology for the next generation of therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee enantiomeric excess levels that match or exceed patent benchmarks. We are committed to delivering high-quality intermediates that empower your drug development programs.
We invite you to collaborate with our technical team to evaluate the feasibility of this route for your specific target molecules. By engaging with us, you can request a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this catalytic method for your supply chain. Please contact our technical procurement team today to obtain specific COA data for our catalog compounds and to discuss route feasibility assessments tailored to your project's unique requirements. Let us be your partner in turning complex chemical challenges into commercial successes.
