Scalable One-Pot Synthesis of Complex Spiro Heterocycles for Advanced Drug Discovery
Introduction to Patent CN111825681A
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN111825681A, published on October 27, 2020, introduces a groundbreaking methodology for the synthesis of (E)-benzylidene spiropyrazole pyrrolizinone compounds. These spiro-fused heterocycles are of immense interest in medicinal chemistry due to their rigid three-dimensional structures, which often confer improved binding affinity and metabolic stability in drug candidates. The disclosed technology represents a significant leap forward by enabling the construction of these intricate molecular architectures through a streamlined, one-pot multi-component reaction strategy. By leveraging the unique reactivity of azomethine ylides generated in situ, this invention provides a robust platform for accessing diverse chemical space without the operational complexities associated with traditional multi-step syntheses.
For research and development teams focused on expanding their library of bioactive molecules, this patent offers a compelling solution to the challenges of synthesizing spiro-compounds. The method circumvents the need for harsh reaction conditions or sensitive reagents, instead utilizing stable and commercially accessible starting materials such as tetrahydropyrrole, various substituted aromatic aldehydes, and pyrazolone acetals. This accessibility is crucial for accelerating the hit-to-lead optimization phase in drug discovery programs. Furthermore, the versatility of the reaction allows for the introduction of a wide array of functional groups on the aromatic rings, facilitating the rapid generation of analogues for structure-activity relationship (SAR) studies. As a reliable pharmaceutical intermediate supplier, understanding such innovative synthetic routes is essential for maintaining a competitive edge in providing high-value building blocks to the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazolone-based spiro compounds has been fraught with significant technical hurdles that impede efficient production. Prior art, such as the work by Wang Rui's group in 2012 and 2013, relied heavily on organocatalysts like thiourea derivatives to facilitate asymmetric Michael addition and cycloaddition reactions. While these methods demonstrated stereocontrol, they often suffered from limited substrate scope and moderate yields, restricting their utility in large-scale applications. Additionally, many conventional routes require the pre-synthesis of specialized dipolarophiles or the use of expensive chiral catalysts that are difficult to recover and reuse. The reliance on multi-step sequences increases the overall process mass intensity (PMI), leading to higher solvent consumption and waste generation. For procurement managers, these inefficiencies translate directly into elevated raw material costs and longer lead times, making the sourcing of such complex intermediates a logistical challenge.
The Novel Approach
In stark contrast, the methodology described in CN111825681A revolutionizes the synthesis landscape by employing a direct, one-pot three-component coupling reaction. This novel approach utilizes a simple acid catalyst, such as acetic acid or p-toluenesulfonic acid, to drive the formation of the key azomethine ylide intermediate directly from tetrahydropyrrole and aromatic aldehydes. This eliminates the necessity for isolating unstable intermediates or employing transition metal catalysts, thereby simplifying the workflow significantly. The reaction proceeds efficiently in common organic solvents like toluene, acetonitrile, or ethanol at temperatures between 80°C and 130°C. 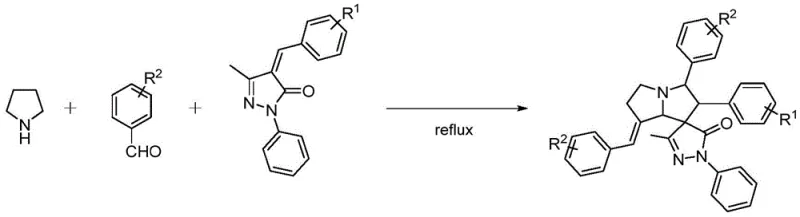 This streamlined process not only enhances the overall atom economy but also drastically reduces the operational burden on laboratory personnel. By consolidating multiple bond-forming events into a single vessel, the new method achieves cost reduction in API manufacturing by minimizing unit operations and purification steps. The broad tolerance for various substituents, including methyl, methoxy, bromo, and chloro groups, ensures that this protocol is adaptable for synthesizing a wide variety of derivatives, making it an ideal candidate for commercial scale-up of complex heterocycles.
This streamlined process not only enhances the overall atom economy but also drastically reduces the operational burden on laboratory personnel. By consolidating multiple bond-forming events into a single vessel, the new method achieves cost reduction in API manufacturing by minimizing unit operations and purification steps. The broad tolerance for various substituents, including methyl, methoxy, bromo, and chloro groups, ensures that this protocol is adaptable for synthesizing a wide variety of derivatives, making it an ideal candidate for commercial scale-up of complex heterocycles.
Mechanistic Insights into Azomethine Ylide Mediated Cycloaddition
The core of this synthetic innovation lies in the elegant generation and utilization of azomethine ylides as 1,3-dipoles. The mechanism initiates with the condensation of tetrahydropyrrole and an aromatic aldehyde to form an iminium ion intermediate. This species undergoes a crucial [1,3]-hydrogen shift to generate the reactive azomethine ylide. Uniquely, in the presence of excess aldehyde, this ylide can further react to form a benzylidene-substituted variant, which is pivotal for the final structural outcome. This in situ generation avoids the handling of hazardous or unstable ylide precursors, enhancing the safety profile of the process. The resulting 1,3-dipole then engages in a [3+2] cycloaddition with the pyrazolone derivative, which acts as the dipolarophile. This concerted pericyclic reaction constructs two new sigma bonds and a new ring system simultaneously, establishing the spiro-center with high regioselectivity.
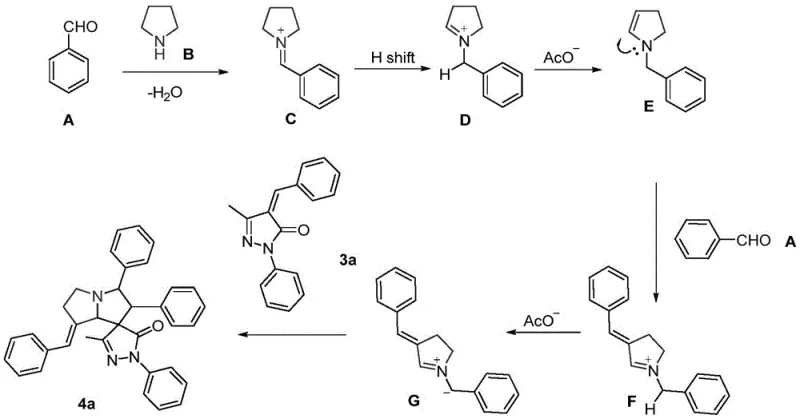 Understanding this mechanistic pathway is vital for R&D directors aiming to optimize purity and control the impurity profile. The use of mild acid catalysis ensures that side reactions, such as polymerization of the aldehyde or decomposition of the pyrazolone, are minimized. The specific [1,3]-hydrogen migration phenomenon described in the patent allows for the formation of the (E)-benzylidene configuration, which is thermodynamically favored and structurally distinct from other isomers. This level of control over the stereochemical outcome is essential for producing high-purity spiro compounds that meet the stringent quality standards required for pharmaceutical applications. The mechanism underscores the efficiency of the process, as it leverages the intrinsic reactivity of simple starting materials to build complexity rapidly, avoiding the need for protecting groups or activation steps that typically complicate synthesis.
Understanding this mechanistic pathway is vital for R&D directors aiming to optimize purity and control the impurity profile. The use of mild acid catalysis ensures that side reactions, such as polymerization of the aldehyde or decomposition of the pyrazolone, are minimized. The specific [1,3]-hydrogen migration phenomenon described in the patent allows for the formation of the (E)-benzylidene configuration, which is thermodynamically favored and structurally distinct from other isomers. This level of control over the stereochemical outcome is essential for producing high-purity spiro compounds that meet the stringent quality standards required for pharmaceutical applications. The mechanism underscores the efficiency of the process, as it leverages the intrinsic reactivity of simple starting materials to build complexity rapidly, avoiding the need for protecting groups or activation steps that typically complicate synthesis.
How to Synthesize (E)-Benzylidene Spiropyrazole Pyrrolizinones Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The protocol is designed to be user-friendly, utilizing standard equipment such as round-bottom flasks and heating mantles. The choice of solvent plays a critical role, with toluene being the preferred medium for achieving optimal results, although acetonitrile and ethanol are viable alternatives depending on solubility requirements. The molar ratio of reactants is also a key variable; typically, a slight excess of the aromatic aldehyde and tetrahydropyrrole relative to the pyrazolone derivative drives the equilibrium towards the desired spiro product. Following the reaction, standard workup procedures involving solvent evaporation and silica gel chromatography are sufficient to isolate the target compound in high purity.
- Mix tetrahydropyrrole, substituted aromatic aldehydes, and pyrazolone acetal derivatives in an organic solvent such as toluene.
- Add an organic acid catalyst like acetic acid and heat the mixture to reflux temperatures between 80°C and 130°C.
- Maintain the reaction for 12 to 16 hours, then concentrate the solvent and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic benefits for supply chain stability and cost management. The elimination of transition metal catalysts is a primary driver for cost efficiency, as it removes the need for expensive metal salts and the associated downstream processing required to reduce metal residues to ppm levels. This simplification of the purification train directly contributes to cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the use of commodity chemicals like benzaldehyde derivatives and pyrrolidine ensures a robust supply chain, mitigating the risk of shortages associated with specialized reagents. The operational simplicity of the one-pot process reduces labor hours and energy consumption, enhancing the overall economic viability of the production process.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive organic acids like acetic acid significantly lowers the direct material costs. Additionally, the one-pot nature of the reaction reduces solvent usage and waste disposal fees, leading to substantial cost savings over the lifecycle of the product. The avoidance of complex purification steps for metal removal further streamlines the manufacturing process, reducing both time and resource expenditure.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals ensures that production schedules are not disrupted by the scarcity of exotic reagents. This stability is crucial for maintaining consistent delivery timelines to downstream customers. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing the supply chain against potential bottlenecks.
- Scalability and Environmental Compliance: The method's compatibility with standard reflux conditions and common solvents facilitates easy scale-up from gram to kilogram scales without requiring specialized high-pressure or cryogenic equipment. The reduced generation of hazardous waste aligns with green chemistry principles, simplifying regulatory compliance and environmental reporting. This scalability ensures that the technology can meet increasing market demand efficiently.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this synthesis route. Understanding these details helps in assessing the feasibility of integrating this method into existing production workflows.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes inexpensive organic acids like acetic acid or p-toluenesulfonic acid, eliminating the need for costly transition metals and subsequent heavy metal removal steps.
Q: What is the typical reaction temperature and time for this process?
A: The reaction is typically conducted under reflux conditions at temperatures ranging from 80°C to 130°C, with an optimal duration of 12 to 16 hours depending on the specific substrate substituents.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the one-pot nature of the reaction, combined with the use of readily available raw materials and simple workup procedures, makes it highly amenable to commercial scale-up and industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)-Benzylidene Spiropyrazole Pyrrolizinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic methodologies outlined in CN111825681A for advancing drug discovery pipelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity spiro compounds that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex heterocyclic synthesis positions us as a strategic partner for pharmaceutical companies seeking to secure their supply of critical intermediates.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can optimize your budget. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a reliable supply chain dedicated to quality, efficiency, and innovation in fine chemical manufacturing.
